Surface Plasmon Resonance (SPR): A Comprehensive Guide to Label-Free Biomolecular Interaction Analysis
This article provides a thorough exploration of Surface Plasmon Resonance (SPR) as a powerful, label-free technology for real-time biomolecular interaction analysis.
Surface Plasmon Resonance (SPR): A Comprehensive Guide to Label-Free Biomolecular Interaction Analysis
Abstract
This article provides a thorough exploration of Surface Plasmon Resonance (SPR) as a powerful, label-free technology for real-time biomolecular interaction analysis. Tailored for researchers, scientists, and drug development professionals, it covers foundational principles from the plasmonic effect to sensorgram interpretation. The scope extends to practical methodologies across drug discovery, diagnostics, and biomedical research, alongside advanced optimization strategies and signal enhancement techniques. A comparative analysis with other label-free methods like Biolayer Interferometry (BLI) offers guidance for platform selection. The content synthesizes the latest advancements, including algorithm-assisted optimization and novel biosensor designs, to empower robust experimental design and data interpretation in kinetic and affinity studies.
The Principles of SPR: Unlocking Label-Free, Real-Time Detection
Surface Plasmon Resonance (SPR) has established itself as a cornerstone technology for real-time, label-free biomolecular interaction analysis, revolutionizing fields from drug discovery to diagnostics [1]. The core of this technology hinges on a fascinating optical phenomenon: the excitation of plasmons, which are coherent oscillations of free electrons at the interface of a metal and a dielectric material [2] [1]. When incident light couples with these electron oscillations under specific conditions, it generates a surface-bound electromagnetic wave, leading to a sharp drop in reflected light intensity at a characteristic resonance angle or wavelength [1]. This resonance is exquisitely sensitive to minute changes in the refractive index at the metal surface, a property that forms the physical basis for label-free detection. When a biomolecule such as a protein or DNA binds to a ligand immobilized on the sensor surface, it alters the local refractive index, causing a measurable shift in the resonance condition [1]. This allows researchers to monitor binding events in real-time without the need for fluorescent or radioactive labels, thereby preserving the native state of the molecules under study [2].
The significance of SPR in modern biosensing is profound. Its label-free nature circumvents the drawbacks of label-based methods, such as potential perturbation of molecular function, and does not suffer from photobleaching, enabling prolonged observations [2]. Furthermore, SPR provides direct access to quantitative kinetic parameters—association (k_on) and dissociation (k_off) rate constants—and equilibrium affinity constants (K_D), which are crucial for understanding interaction mechanisms and for the efficient development of therapeutic candidates [3] [1]. The technology's versatility allows it to be applied to a wide range of analytes, including proteins, nucleic acids, small molecules, and even whole cells [4] [1].
Fundamental Principles of Plasmons and Resonance
The Optical Phenomenon of Surface Plasmons
The term "plasmon" refers to the quanta of plasma oscillations. In the context of SPR, surface plasmons (SPs) are electromagnetic waves that propagate along the interface between a metal (typically gold or silver) and a dielectric (e.g., a buffer solution or glass) [1]. These waves are transverse magnetic (TM-polarized), meaning the magnetic field is parallel to the interface while the electric field is perpendicular and decays exponentially into both media, creating an evanescent field [1]. This field typically extends 200-300 nanometers from the metal surface, defining the sensitive volume of the sensor [1].
The excitation of surface plasmons requires momentum matching between the incident photons and the plasmons. For metals like gold, this condition cannot be met by direct illumination. The most common method to overcome this momentum mismatch is the Kretschmann configuration, which uses a prism coupler [1]. In this setup, a thin metal film (∼50 nm) is deposited on the prism base. A beam of p-polarized light is directed through the prism onto the metal film. At a specific angle of incidence, the component of the light's wave-vector parallel to the interface matches that of the surface plasmon, leading to a resonant transfer of energy and a sharp minimum in the intensity of the reflected light [1]. This is the fundamental "resonance" in Surface Plasmon Resonance.
From Resonance to Label-Free Detection
The working principle of SPR-based biodetection is directly tied to the sensitivity of the surface plasmon's propagation constant to the refractive index (RI) of the dielectric medium adjacent to the metal surface. The resonance condition is given by:
k_SP = (ω/c) * √(ε_m * n² / (ε_m + n²))
where k_SP is the surface plasmon wave-vector, ω is the angular frequency of light, c is the speed of light, ε_m is the dielectric constant of the metal, and n is the refractive index of the dielectric.
Any change in the refractive index within the evanescent field, such as the binding of an analyte molecule (with a higher RI than the buffer) to an immobilized ligand, alters n. This shifts the resonance condition, observable as a change in the resonance angle (Δθ), wavelength (Δλ), or intensity (ΔI) [1]. This shift, often measured in Response Units (RU), is directly proportional to the mass concentration of the bound analyte, enabling real-time monitoring of binding events [4] [1]. The following diagram illustrates this core configuration and detection principle.
Advancements in SPR Sensing Platforms
The field of SPR sensing has evolved significantly beyond the traditional prism-coupled configuration, leading to enhanced performance and new applications.
Key SPR Configurations and Their Performance
The following table summarizes and compares the core principles, advantages, and typical performance metrics of the most prominent SPR and related plasmonic sensing platforms.
| Sensing Platform | Core Principle | Key Advantage(s) | Typical Limit of Detection (LOD) | Throughput & Multiplexing |
|---|---|---|---|---|
| Traditional SPR [1] | Prism-coupled excitation of propagating surface plasmons on a thin metal film. | Established golden standard for kinetic analysis; high sensitivity. | Picomolar (pM) to nanomolar (nM) range. | Low to moderate; requires complex optics for multiplexing. |
| Localized SPR (LSPR) [2] [1] | Resonant light scattering & absorption by confined plasmons on individual metal nanoparticles. | Miniaturization; simpler optics; no prism required; higher spatial resolution. | Comparable or slightly lower than SPR for single particles. | High potential via nanoparticle arrays. |
| MetaSPR [4] | Integration of SPR/LSPR with engineered metasurfaces (periodic nanoarrays). | Direct light-plasmon coupling without prisms; high-throughput; portable systems; cost-effective. | High sensitivity suitable for cell-based assays and biomarker detection. | Very High (e.g., 96-well plate format). |
Plasmonic Energy Transfer Mechanisms
Beyond refractometric sensing, plasmons can participate in resonant energy transfer processes. A notable example is Plasmon-Induced Resonance Energy Transfer (PIRET), which differs from traditional Förster Resonance Energy Transfer (FRET) [5]. In PIRET, energy is transferred non-radiatively from a plasmonic nanostructure to a semiconductor or acceptor molecule, potentially in a direction opposite to the Stokes shift, without a required spectral overlap between donor emission and acceptor absorption [5]. This mechanism is particularly explored for solar energy conversion, as it can harvest a broader spectrum of sunlight and induce charge separation in semiconductors with energies below their band gap [5]. While distinct from the direct refractometric sensing of conventional SPR, PIRET exemplifies the broader utility of plasmonic excitations in optical sensing and energy harvesting.
Experimental Protocols for Biomolecular Interaction Analysis
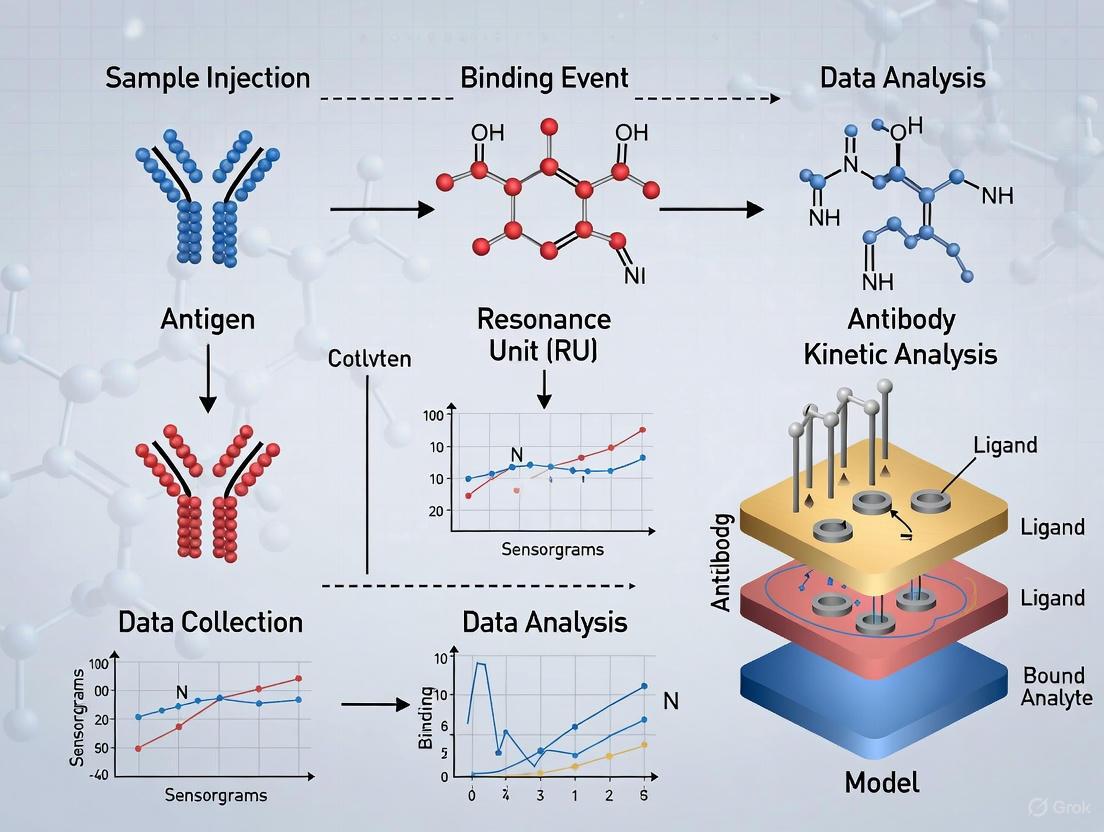
A typical SPR experiment involves immobilizing one interaction partner (the ligand) on the sensor surface and flowing the other (the analyte) over it. The following workflow details a standard protocol for kinetic characterization.
Detailed Experimental Workflow
The following diagram outlines the key stages of an SPR experiment, from surface preparation to data analysis.
Phase 1: Surface Preparation and Ligand Immobilization The sensor surface must be functionalized to allow for specific and stable immobilization of the ligand while minimizing non-specific binding. A common strategy is amine coupling [3].
- Procedure:
- Activation: The gold sensor chip, typically pre-coated with a carboxymethylated dextran matrix, is activated with a mixture of N-hydroxysuccinimide (NHS) and N-ethyl-N'-(3-dimethylaminopropyl)carbodiimide (EDC) [3].
- Immobilization: The ligand (e.g., a protein, antibody, or DNA oligonucleotide) in a low-salt buffer at a pH slightly below its isoelectric point (pI) is injected over the activated surface. The positively charged amine groups on the ligand react with the NHS esters on the surface, forming stable amide bonds [3].
- Blocking: Excess reactive esters are deactivated by injecting a blocking agent, such as ethanolamine [3].
- Critical Note: The immobilization level must be optimized. Too high a density can cause mass transport limitation, where the rate of analyte binding is limited by its diffusion to the surface rather than the intrinsic interaction kinetics. This can be checked by running the analyte at different flow rates; a change in observed rate constants indicates mass transport effects [3].
Phase 2: Baseline Acquisition The system is flushed with a suitable running buffer (e.g., PBS, HEPES) until a stable baseline is achieved. This establishes the reference refractive index signal [3].
Phase 3: Association Phase
- The analyte is injected over the ligand-functionalized surface at a constant flow rate (e.g., 30-50 µL/min) for a set period (typically 1-5 minutes) [3].
- The binding event causes an increase in the refractive index at the surface, leading to a positive shift in the SPR signal (the "sensogram"). The rate of this increase is governed by the association rate constant (
k_on) and the analyte concentration.
Phase 4: Dissociation Phase
- The flow is switched back to the running buffer without analyte.
- The decrease in the SPR signal as the analyte dissociates from the ligand is monitored. The rate of this decrease is governed by the dissociation rate constant (
k_off).
Phase 5: Surface Regeneration
- To reuse the sensor surface, the bound analyte is removed by injecting a regeneration solution (e.g., Glycine-HCl pH 1.5-2.5, or Guanidine HCl) that disrupts the interaction without denaturing the immobilized ligand [3].
- The surface must be tested for stability over multiple regeneration cycles.
Phase 6: Data Analysis and Kinetic Modeling
The sensogram (SPR signal vs. time) is fit to a kinetic model to extract k_on and k_off. The simplest and most common model is the 1:1 Langmuir binding model.
- The analysis involves globally fitting the association and dissociation data for multiple analyte concentrations simultaneously [3].
- The equilibrium dissociation constant (
K_D) is calculated from the ratio of the rate constants:K_D = k_off / k_on[3] [1]. - It is critical to use appropriate software (commercial or open-source) and to understand the underlying model assumptions to avoid erroneous results from "black box" fitting [3].
The Scientist's Toolkit: Essential Research Reagents and Materials
A successful SPR experiment relies on a suite of specialized materials and reagents. The following table details the key components of the researcher's toolkit.
| Item | Function / Description | Key Considerations |
|---|---|---|
| SPR Instrument [1] | Core system for generating and detecting the plasmon resonance. | Choices include traditional prism-based (Biacore), high-throughput (MetaSPR 96-well), or single-particle LSPR systems. |
| Sensor Chip (Gold) [1] | The substrate where the metal-dielectric interface is established. | A high-quality gold film with uniform thickness and smoothness is critical for consistent plasmon resonance and sensitivity [1]. |
| Dextran Matrix [3] | A hydrogel layer on the chip surface that provides a hydrophilic environment for immobilization and reduces non-specific binding. | Commonly used in commercial chips (e.g., CM5 from Biacore). |
| Coupling Reagents (NHS/EDC) [3] | Chemicals used to activate carboxyl groups on the sensor surface for covalent amine coupling of ligands. | EDC promotes the formation of an reactive intermediate, which NHS stabilizes as an NHS ester. |
| Ligand | The biomolecule that is immobilized on the sensor surface to capture the analyte. | Purity and activity are paramount. Must be in a compatible buffer for immobilization. |
| Analyte | The molecule in solution that binds to the immobilized ligand. | Should be highly pure and prepared in a series of concentrations for kinetic analysis. |
| Running Buffer [3] | The solution used to establish baseline, dilute analytes, and dissociate complexes. | Must be optimized to maintain ligand and analyte stability and minimize non-specific binding (e.g., PBS with added surfactants). |
| Regeneration Solution [3] | A solution that removes bound analyte without damaging the immobilized ligand, allowing for surface reuse. | Must be empirically determined for each interaction (e.g., low pH, high salt, chaotropic agents). |
Surface Plasmon Resonance stands as a powerful and versatile technology rooted in the fundamental optical principles of plasmons and resonant energy transfer. Its capacity for real-time, label-free detection of biomolecular interactions provides researchers and drug development professionals with unparalleled insights into kinetics and affinity. The ongoing evolution of the technology—from traditional prism-coupled SPR to miniaturized LSPR and high-throughput MetaSPR platforms—continues to expand its application horizons into cell biology, point-of-care diagnostics, and environmental monitoring. By adhering to robust experimental protocols and understanding the core physical phenomena, scientists can fully leverage SPR to drive discovery and innovation in the life sciences.
Surface Plasmon Resonance (SPR) is a powerful, label-free detection technique that has revolutionized the study of biomolecular interactions in real-time. Unlike traditional methods that require fluorescent or radioactive labeling of molecules, SPR measures binding events directly by detecting changes in the refractive index at a sensor surface, thereby avoiding potential alterations to the molecules' natural activity [6]. First demonstrated in 1983, this technology has become indispensable in pharmaceutical engineering, food sample analysis, antigen-antibody characterization, and basic science research [7]. The core principle hinges on exciting surface plasmons—coherent electron oscillations at the interface between a metal and a dielectric medium—using light, and precisely monitoring the conditions under which this resonance occurs [8]. The Kretschmann configuration is the most widely adopted and efficient method for achieving this excitation, forming the foundation for a vast range of biosensing applications [9] [8].
Fundamental Principles of the Kretschmann Configuration
Optical Setup and Critical Angle Phenomenon
The Kretschmann configuration, named after the German physicist Erich Kretschmann, employs a high-refractive-index prism as an optical coupler [6]. In this setup, a thin metal film (typically gold or silver) is deposited directly onto the base of the prism. Monochromatic, polarized light is then directed through the prism to strike the metal film at an angle greater than the critical angle for total internal reflection (TIR) [8]. Under TIR, an evanescent wave penetrates a short distance (typically hundreds of nanometers) beyond the glass-metal interface into the metal layer. When the momentum of this evanescent wave matches that of the surface plasmons at the outer metal-dielectric (e.g., sample solution) interface, a resonance condition is met [6]. This coupling results in a transfer of energy from the incident photon to the surface plasmon, manifesting as a sharp dip in the intensity of the reflected light at a specific angle of incidence—the SPR angle [6] [7].
The Role of the Metal Film and Plasmon Generation
The thin metal film, usually 50 nm gold, serves a dual purpose: it enables the evanescent wave to tunnel through and excite surface plasmons on its outer surface [9] [8]. Gold is favored for its high plasmonic activity and chemical inertness [10]. During resonance, the oscillating surface plasmons generate an enhanced electromagnetic field that extends approximately 300 nm from the metal surface into the adjacent medium [6]. Any change in the properties of this adjacent medium, such as the binding of biomolecules to probes immobilized on the metal film, alters the local refractive index. This change shifts the SPR angle, and tracking this shift in real-time allows for the quantitative assessment of binding kinetics—including association (k_on) and dissociation (k_off) rates—and affinity (K_D) without any labels [6].
Experimental Protocols for Kretschmann-Based SPR
Standardized Sensor Chip Preparation
A foundational experiment in SPR involves characterizing the well-established interaction between biotin and streptavidin to validate sensor surface functionality [10].
- Objective: To functionalize a gold sensor chip and verify its performance by detecting the specific binding of streptavidin to immobilized biotin.
- Materials:
- SPR instrument with Kretschmann configuration prism.
- Bare gold sensor chip.
- Biotinylated Bovine Serum Albumin (biotin-BSA).
- Non-biotinylated BSA (for negative control).
- Streptavidin solution in a suitable buffer (e.g., PBS).
- Procedure:
- Surface Cleaning: The gold sensor chip is first cleaned, often with a piranha solution, to remove organic contaminants and ensure a uniform surface.
- Ligand Immobilization: A solution of biotin-BSA is incubated over the gold surface, allowing the protein to adsorb. A separate flow cell or spot is incubated with non-biotinylated BSA as a negative control.
- Blocking: Remaining active sites on the gold surface are blocked with a non-reactive protein (e.g., casein) or ethanolamine to minimize non-specific binding in future steps.
- Analyte Binding: Streptavidin solution is injected and flowed across both the experimental and control surfaces.
- Dissociation & Regeneration: The flow is switched to buffer to monitor the dissociation of streptavidin. The surface can often be regenerated for subsequent experiments using a mild acidic or basic solution that breaks the biotin-streptavidin bond without damaging the immobilized biotin-BSA.
- Expected Outcome: A significant binding signal is observed on the biotin-BSA surface upon streptavidin injection, which remains stable after rinsing. The control surface should show little to no binding, confirming the specificity of the interaction [10].
Advanced Protocol: Quantifying mAb Glycosylation in Crude Samples
A more complex, integrated assay demonstrates the power of SPR for analyzing therapeutic monoclonal antibodies (mAbs) directly in crude, heterogeneous mixtures [11].
- Objective: To simultaneously quantify and characterize the glycosylation (terminal galactosylation and core fucosylation) of mAbs from bioreactor cell cultures without prior purification.
- Materials:
- SPR instrument.
- Sensor chip pre-immobilized with Protein A (binds the Fc region of IgG).
- Crude mAb sample from a bioreactor.
- Running buffer.
- Recombinant FcγRII receptor.
- Procedure:
- In-Situ Capture and Purification: The crude mAb sample is injected over the Protein A surface. Protein A specifically captures mAbs from the mixture via their Fc region, effectively purifying them on the sensor surface. Impurities are washed away with running buffer.
- Quantification: Under conditions of mass transport limitation, the initial binding response can be correlated to the concentration of mAbs in the sample.
- Glycosylation Characterization: Following the wash, the kinetics of the interaction between the captured mAbs and an injected FcγRII receptor are recorded. The affinity and kinetics of this interaction are highly sensitive to the mAb's glycosylation pattern, particularly the levels of terminal galactose and core fucose.
- Expected Outcome: This protocol provides two critical quality attributes from a single, crude sample: mAb concentration and a detailed glycosylation profile, enabling real-time, at-line monitoring of biopharmaceutical production [11].
Performance Optimization and Material Innovations
Quantitative Comparison of Sensing Materials
The performance of an SPR sensor is quantified by its Sensitivity (response per refractive index unit, RIU) and Figure of Merit (FoM), which is the ratio of sensitivity to the resonance dip's full width at half minimum (FWHM) [9]. Research continues to optimize the metal film and explore hybrid structures.
Table 1: Performance Comparison of Different Plasmonic Sensing Films
| Sensing Structure | Metal/Thickness | Dielectric Layer | Sensitivity | Figure of Merit (FoM) | Key Characteristics |
|---|---|---|---|---|---|
| Conventional SPR [9] | Au (50 nm) | None | Baseline | Baseline | Standard, widely used, chemically inert. |
| Hyperbolic Mode Resonance (HMR) [9] | Au (36 nm) | SnO₂ (70 nm) | ~2x increase vs. 36nm Au alone | 16% improvement vs. 36nm Au alone | Combines metallic and dielectric films; enhanced visibility. |
| Aluminum Thin Film [10] | Al/Al₂O₃ (12/3 nm) | Native Al₂O₃ | 59.25°/RIU (angular)70041 IU/RIU (intensity) | N/A | Low-cost, naturally anti-fouling, sharp plasmonic dip suited for fixed-angle sensing. |
Emerging Materials: Aluminum as a Plasmic Substrate
While gold is the traditional choice, aluminum is emerging as a promising alternative. When deposited via electron-beam physical vapor deposition to a thickness of ~15 nm, it forms a stable, native Al₂O₃ layer upon air exposure [10]. This Al/Al₂O₃ structure exhibits a steeper plasmonic dip than gold, making it exceptionally well-suited for fixed-angle intensity measurement, where it demonstrated a 13.9% higher sensitivity than gold [10]. A significant advantage is its inherent resistance to non-specific protein adsorption; when exposed to undiluted human blood serum, it showed a reduction of over 75% in non-specific binding compared to gold, making it ideal for sensing in complex media like blood without additional surface modifications [10].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful SPR experiments rely on a suite of specialized reagents and materials designed for the Kretschmann configuration.
Table 2: Key Research Reagent Solutions for SPR Experiments
| Item | Function in Experiment | Example Use Case |
|---|---|---|
| Gold Sensor Chip | The core plasmonic substrate; often pre-functionalized with chemical groups (e.g., carboxymethyl dextran) for ligand immobilization. | General protein-protein interaction studies [7]. |
| Protein A Sensor Chip | Pre-immobilized with Protein A for specific capture of antibodies via their Fc region. | Quantification and characterization of monoclonal antibodies [11]. |
| Biotinylated BSA | Used to immobilize a biotin ligand on the surface, which then captures streptavidin-coated or biotin-labeled molecules. | Surface validation and assay development [10]. |
| Streptavidin | High-affinity binding partner for biotin; often conjugated to molecules to capture them onto a biotinylated surface. | Amplified detection of DNA or other biotinylated analytes. |
| Running Buffer (e.g., PBS, HBS-EP) | The solution that carries the analyte; it maintains a constant pH and ionic strength and can include surfactants to minimize non-specific binding. | All SPR experiments require a stable, matched running buffer [7]. |
| Regeneration Solution (e.g., Glycine pH 2.0) | A solution that disrupts the ligand-analyte bond without damaging the immobilized ligand, allowing for chip re-use. | Regenerating a Protein A or antibody surface for multiple analysis cycles. |
Visualizing the Kretschmann Configuration and Workflow
The following diagrams illustrate the core optical setup and a generalized experimental workflow for an SPR binding assay.
Optical Path and SPR Dip Formation
SPR Binding Assay Workflow
The Kretschmann configuration remains the cornerstone of modern SPR technology, providing a robust and versatile platform for label-free, real-time biomolecular interaction analysis. Its principle—utilizing a prism to couple light to a surface plasmon at a specific critical angle—enables the sensitive detection of refractive index changes corresponding to mass binding on the sensor surface. Ongoing innovations, such as the development of hyperbolic mode resonances and the introduction of low-cost, anti-fouling aluminum films, continue to push the boundaries of sensitivity and application. As the demand for sophisticated analytical techniques grows in drug discovery and diagnostics, the Kretschmann configuration's role as an invaluable tool for researchers and scientists is firmly cemented, with its future prospects brightened by continuous material and methodological advancements.
Surface Plasmon Resonance (SPR) is a powerful optical biosensing technology that has revolutionized the study of molecular interactions in fields ranging from biochemistry to drug development. Its significance lies in its ability to provide real-time, label-free, and noninvasive analysis of binding events occurring at a sensor surface [12]. Since its first application for real-time biological analysis in the 1990s, SPR has become an indispensable tool for quantifying molecular interactions without requiring fluorescent or radioactive labels that could potentially alter molecular behavior [12] [13].
At the heart of SPR technology is a simple yet powerful principle: when molecules bind to a functionalized sensor surface, they alter the local refractive index in the immediate vicinity of the surface. This change in refractive index directly influences the behavior of surface plasmons—collective oscillations of free electrons at a metal-dielectric interface [14]. By monitoring these changes in real-time, researchers can obtain rich information about binding kinetics, affinity, and concentration. This technical guide explores the fundamental relationship between refractive index changes and mass concentration, providing researchers with the theoretical and practical knowledge to implement SPR effectively in their experimental workflows.
Theoretical Foundations: From Binding to Signal
The SPR Phenomenon and Evanescent Field
The most common configuration for SPR sensing is the Kretschmann geometry, where a thin metal film (typically gold) is positioned at the interface between a glass prism and the sample medium [12] [14]. When plane-polarized light hits this metal film under conditions of total internal reflection, an evanescent wave penetrates into the metal layer. Under specific resonance conditions, this evanescent wave can couple with the free electrons in the metal, generating surface plasmons [14].
The evanescent field generated during SPR extends approximately 100-300 nm from the sensor surface into the adjacent medium, making SPR exceptionally sensitive to changes occurring within this limited region [13] [14] [15]. This property makes SPR ideal for monitoring binding events between molecules immobilized on the sensor surface and their partners in solution. When binding occurs, the accumulation of mass within the evanescent field alters the local refractive index, which in turn modifies the conditions required for resonance [12].
The Quantitative Relationship: Refractive Index to Mass Concentration
The fundamental relationship that enables SPR to function as a quantitative tool is the linear correlation between the change in refractive index (n) at the sensor surface and the mass concentration (C) of molecules that have bound to that surface [14]. This relationship can be expressed as:
Δn = (dn/dC) × ΔC
Where Δn represents the change in refractive index, ΔC is the change in mass concentration at the surface, and dn/dC is the refractive index increment (RII), a property specific to each type of biomolecule [14]. For most proteins, the RII falls within a relatively narrow range of approximately 0.18-0.19 mL/g, which simplifies quantitative applications [14].
The SPR response (measured in resonance units or RU) is directly proportional to this change in refractive index. For many commercial SPR instruments, 1000 RU corresponds to a shift of 0.1° in resonance angle, and this change typically equates to an increase in surface concentration of approximately 1 ng/mm² [14]. This well-characterized relationship forms the basis for quantifying binding events in SPR experiments.
Experimental Methodology and Protocols
Core SPR Experimental Setup
The basic components required for SPR measurements include a light source, prism, gold film, and detector [12]. A typical experimental setup based on the Kretschmann configuration positions a gold-coated glass coverslip against a prism using immersion oil to ensure optimal optical contact [12]. A sample cell is then attached to the gold film, allowing controlled introduction of analytes.
To measure the SPR angle, researchers monitor the intensity of reflected light while varying the angle of incidence using a rotating turntable or other mechanical means [12]. The resonance angle is identified as the point of minimum reflectivity, where light energy is most efficiently transferred to surface plasmons [12] [14]. Modern SPR instruments automate this process, continuously tracking the resonance angle and converting it to resonance units that are displayed in a sensorgram—a real-time plot of response versus time [14].
Figure 1: Standard SPR experimental workflow showing key phases from surface preparation to data analysis.
Critical Parameters for Quantitative Measurements
Several parameters must be carefully controlled to ensure accurate correlation between SPR response and mass concentration:
- Metal film properties: Gold is typically used due to its chemical inertness and favorable SPR properties. The optimal thickness is approximately 50 nm [14].
- Temperature stability: Refractive index is sensitive to temperature fluctuations, necessitating precise temperature control during measurements [14].
- Light source characteristics: Monochromatic, p-polarized light is essential for obtaining a sharp resonance dip [14].
- Flow rate consistency: In flow-based systems, maintaining consistent flow rates ensures reproducible mass transport conditions [15].
Calibration and Reference Techniques
To account for nonspecific binding and bulk refractive index changes, SPR experiments typically incorporate reference channels [14]. These channels consist of functionalized surfaces that should not interact specifically with the analyte, allowing researchers to subtract background effects from the specific binding signal. For absolute quantification, systems must be calibrated using standards with known concentrations and properties [14].
Advanced SPR systems now incorporate multi-parametric detection that monitors not only the resonance angle but also the shape and intensity of the SPR curve [14]. This approach enables more sophisticated analysis and can account for confounding factors such as changes in bulk refractive index without requiring a separate reference channel.
Quantitative Data and Correlations
Refractive Index Increments for Common Biomolecules
Table 1: Typical refractive index increments (RII) for various biomolecule classes
| Biomolecule Class | Refractive Index Increment (dn/dc, mL/g) | Notes |
|---|---|---|
| Proteins | 0.18-0.19 | Relatively consistent across different proteins |
| Antibodies | ~0.185 | Slight variations between subtypes |
| Nucleic Acids | ~0.17 | Varies with base composition |
| Small Molecules | Variable | Highly dependent on chemical structure |
SPR Response Correlations and Conversions
Table 2: SPR response conversions and their practical significance
| Parameter | Value | Experimental Significance |
|---|---|---|
| SPR Response | 1000 RU | Typically corresponds to 0.1° resonance angle shift |
| Surface Coverage | 1 ng/mm² | Approximately equivalent to 1000 RU response |
| Protein Layer | 1 ng/mm² | Represents ~1 pg protein per sensor spot (1 mm²) |
| Detection Limit | ~0.1-1 RU | Enables detection of small molecules and low-abundance interactions |
The maximum expected response (Rmax) for a 1:1 binding interaction can be predicted using the equation:
Rmax = (MWanalyte / MWligand) × R_ligand × (Stoichiometry) [14]
Where MW represents molecular weight, R_ligand is the immobilization level of the ligand, and stoichiometry accounts for the number of binding sites. This calculation is essential for experimental design and data interpretation.
Research Reagent Solutions and Materials
Table 3: Essential reagents and materials for SPR experiments
| Item | Function | Specifications |
|---|---|---|
| Gold Sensor Chips | SPR-active surface | 50 nm gold thickness on glass substrate [12] [14] |
| Carboxymethylated Dextran | Hydrogel matrix for ligand immobilization | Provides 3D structure for increased binding capacity |
| Coupling Reagents | Covalent immobilization of ligands | EDC/NHS chemistry for amine coupling |
| Regeneration Solutions | Surface regeneration between cycles | Glycine pH 2.0-3.0 or other mild denaturants |
| HBS-EP Buffer | Standard running buffer | 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% surfactant P20 |
| Immersion Oil | Optical coupling between prism and sensor chip | Matches refractive index of prism (n ≈ 1.515) [12] |
Advanced Applications and Techniques
SPR Imaging (SPRi) for Enhanced Throughput
SPR imaging extends conventional SPR by enabling simultaneous monitoring of multiple interaction spots on the sensor surface [13]. This is achieved by using a broad, collimated light beam and detecting the reflected light with a CCD or CMOS camera [13]. SPRi is particularly valuable for screening applications where multiple ligand-analyte combinations must be tested in parallel, such as in epitope mapping, antibody characterization, and biomarker discovery [13].
Recent advances in SPRi have focused on improving spatial resolution through techniques such as surface plasmon scattering microscopy (SPSM), which can achieve diffraction-limited resolution free from the parabolic tails that traditionally limit conventional SPR microscopy [13]. These improvements have opened new possibilities for studying heterogeneous cellular responses and single-molecule interactions.
Integration with Electrochemistry and Other Modalities
The combination of SPR with electrochemical techniques creates a powerful hybrid approach for studying complex biological processes [12]. This integration allows researchers to simultaneously monitor binding events (via SPR) and redox reactions or electron transfer (via electrochemistry) [12]. Applications include investigating the mechanism of electrochemical reactions, studying potential-induced conformational changes in proteins, and developing sophisticated biosensing platforms.
Figure 2: SPR working principle showing key components and the evanescent field that enables detection.
Cellular Applications: SPR Cytometry
SPR cytometry represents an emerging application where SPR is used to measure parameters from intact cells [15]. This approach enables label-free detection of cell membrane antigens, monitoring of cellular morphology changes, and analysis of secreted molecules from single cells [15]. Special considerations for cellular applications include managing the large size of cells relative to the evanescent field depth and addressing the challenges of maintaining cell viability during measurements [15].
A typical cellular experiment involves immobilizing cells on the sensor surface, often through specific antibody-antigen interactions, and then monitoring their response to various stimuli [15]. The multiparametric nature of newer SPR systems is particularly advantageous for cellular studies, as it can help deconvolve complex responses involving multiple simultaneous processes [15].
The correlation between refractive index change and mass concentration forms the fundamental basis for SPR's utility in label-free detection. This relationship, characterized by well-defined refractive index increments for most biomolecules, enables researchers to extract quantitative information about binding kinetics, affinity, and concentration from SPR measurements. As SPR technology continues to evolve, with improvements in spatial resolution, integration with complementary techniques, and expanded applications to cellular systems, its value as a tool for basic research and drug development continues to grow. The protocols and principles outlined in this guide provide a foundation for researchers to implement SPR technology effectively and interpret resulting data within the broader context of molecular interactions and label-free detection research.
Surface Plasmon Resonance (SPR) stands as a cornerstone technology for label-free, real-time analysis of biomolecular interactions. This technical guide deconstructs the SPR sensorgram, the primary data output of this technology, within the broader context of label-free detection research. Aimed at researchers and drug development professionals, this whitepay provides a comprehensive, step-by-step interpretation of the association, steady-state, and dissociation phases. It further offers detailed methodologies for robust experimental design and data analysis, supported by structured data presentation and visualization, to empower the accurate determination of binding kinetics and affinity.
Surface Plasmon Resonance (SPR) is a label-free optical biosensing technique that enables the real-time monitoring of molecular interactions, such as those between an antibody and antigen or a small molecule drug and its protein target [16]. The technology operates on the principle of detecting changes in the refractive index at a sensor surface, which are proportional to the mass of molecules bound [17]. When polarized light hits a sensor chip coated with a thin metal layer (typically gold), it generates an electromagnetic field at the surface. As molecules bind to this surface, the refractive index shifts, altering the resonance condition, which is detected as a change in the angle or wavelength of the reflected light [17] [16].
The primary data output from an SPR instrument is a sensorgram, a plot of the SPR response (in Resonance Units, RU) against time [17]. This real-time binding curve visually encapsulates the entire interaction lifecycle, from initial binding to final dissociation, and contains a wealth of quantitative information on the kinetics, affinity, and specificity of the interaction [18]. The following workflow illustrates the typical process of an SPR experiment leading to sensorgram generation:
The Core Phases of a Sensorgram
A sensorgram is composed of several distinct phases, each revealing specific aspects of the molecular interaction. A typical sensorgram, detailing these phases and their corresponding events on the sensor surface, is illustrated below:
Baseline Phase
The baseline phase represents the starting point of the sensorgram, where only the running buffer flows across the sensor surface, conditioning it and establishing a stable starting signal [17] [18]. A flat, stable baseline is crucial, as drift, injection spikes, or a high buffer response can indicate system instability, requiring inspection or cleaning before proceeding [17] [18]. Common running buffers include phosphate-buffered saline (PBS) and HEPES-NaCl, chosen for their compatibility with the biological interaction under study [17] [19].
Association Phase
The association phase begins at time t=0 with the injection of the analyte over the ligand-immobilized surface [17]. The binding of analyte to ligand causes an increase in mass at the sensor surface, leading to a sharp rise in the SPR signal [18]. This phase is kinetically controlled by the association rate constant (kₐₙ or kₐ) and the concentration of the analyte [C] [17] [20]. The initial part of the curve should show curvature; a linear increase often indicates that the interaction is limited by mass transport, where the diffusion of the analyte to the surface is slower than the binding reaction itself [21]. Ideally, the association profile follows a single exponential curve [17].
Steady-State Phase
The steady-state phase, also referred to as equilibrium, is reached when the rate of analyte association equals the rate of dissociation, resulting in a flat, horizontal response in the sensorgram [22]. At this point, the net rate of complex formation is zero [17]. The response level at steady-state (Rₑq) is dependent on the analyte concentration and the equilibrium dissociation constant (KD) [22]. It is critical to distinguish between steady-state and full saturation; a system can be at equilibrium without all ligand binding sites being occupied [22]. Reaching steady-state is essential for accurate equilibrium analysis.
Dissociation Phase
The dissociation phase is initiated when the analyte solution is replaced by the wash buffer, causing the specific bonds between the analytes and ligands to break [17]. This is represented by a downward slope in the sensorgram as the signal decreases [18]. The rate of this decay is governed solely by the dissociation rate constant (kₒff or k𝒹) [23]. The dissociation should ideally follow a single exponential decay [18]. A slow dissociation rate (low k𝒹) indicates a stable complex with a long half-life, while a fast dissociation suggests a transient interaction [23]. For reliable analysis, the dissociation curve should decrease by at least 5% [23].
Regeneration Phase
The final phase, regeneration, involves flowing a solution (often low pH, like glycine, or high salt) over the sensor surface to disrupt the ligand-analyte interaction completely, removing any remaining bound analyte and restoring the SPR signal to the original baseline [17] [18]. This prepares the surface for a new analysis cycle. The regeneration conditions must be strong enough to remove the analyte but not so harsh as to damage the immobilized ligand's functionality [19]. Successful regeneration is indicated by a return to the pre-injection baseline and is key to reusing sensor surfaces efficiently [17].
Quantitative Analysis of Binding
The sensorgram is not merely a qualitative picture of binding; it is a source of precise kinetic and affinity parameters.
Kinetic Analysis
Kinetic analysis involves fitting the sensorgram data to a binding model, most commonly the Langmuir 1:1 model, which assumes a single analyte binding to a single ligand site with equivalent and independent binding sites [20]. The model provides the following key constants:
- Association rate constant (kₐ): Measured in M⁻¹s⁻¹, it describes how quickly the complex forms.
- Dissociation rate constant (k𝒹): Measured in s⁻¹, it describes how quickly the complex falls apart.
The change in response during the association and dissociation phases can be described by the following equations derived from the Langmuir model [20]:
- Association: ( \frac{dR}{dt} = ka C (R{max} - Rt) - kd R_t )
- Dissociation: ( Rt = R0 e^{-kd(t-t0)} )
Where ( Rt ) is the response at time ( t ), ( C ) is the analyte concentration, and ( R{max} ) is the maximum binding capacity.
Affinity Analysis
The equilibrium dissociation constant (KD) is a measure of binding affinity and can be determined in two primary ways:
- From kinetics: ( KD = \frac{kd}{k_a} ) (units: M) [17] [20].
- From steady-state (equilibrium) analysis: The response at equilibrium (Rₑq) is plotted against the analyte concentration [C], and the data is fit to the equation ( R{eq} = \frac{R{max} \cdot C}{K_D + C} ) to derive the KD [20]. This method requires that the injection time is long enough for all analyte concentrations to reach a clear steady-state level [22].
The following table summarizes the key parameters obtained from sensorgram analysis:
Table 1: Key Quantitative Parameters Derived from Sensorgram Analysis
| Parameter | Symbol | Units | Description | Derivation |
|---|---|---|---|---|
| Association Rate Constant | kₐ, kₒₙ | M⁻¹s⁻¹ | Speed of complex formation | From the curvature of the association phase [20] |
| Dissociation Rate Constant | k𝒹, kₒff | s⁻¹ | Speed of complex breakdown | From the exponential decay of the dissociation phase [23] [20] |
| Equilibrium Dissociation Constant | KD | M | Analyte concentration at half-maximal saturation; measure of affinity | KD = k𝒹 / kₐ (Kinetic) or from steady-state response (Equilibrium) [17] [20] |
| Maximum Response | Rₘₐₓ | RU | Theoretical response at full ligand saturation | Determined by curve fitting or by injecting a saturating analyte concentration [22] |
| Complex Half-Life | t₁/₂ | s, min, h | Time for half the complexes to dissociate | t₁/₂ = ln(2) / k𝒹 [23] |
The Scientist's Toolkit: Essential Reagents and Materials
A successful SPR experiment relies on careful selection of reagents and materials. The following table details key components and their functions.
Table 2: Essential Research Reagent Solutions for SPR Experiments
| Item | Function & Importance | Examples & Notes |
|---|---|---|
| Sensor Chips | Provides the surface for ligand immobilization. Various types exist for different coupling chemistries. | CM5: Carboxymethylated dextran for covalent coupling via amine chemistry. SA: Streptavidin-coated for capturing biotinylated ligands. NTA: Pre-immobilized with NTA for capturing His-tagged ligands [24]. |
| Running Buffer | The solution that flows continuously, establishing the chemical environment for the interaction. | PBS, HEPES-NaCl. Must have appropriate pH and ion composition to maintain biological activity. DMSO concentration must be matched if used to dissolve analytes [17] [19]. |
| Regeneration Buffer | A solution that removes bound analyte without damaging the ligand, enabling surface reuse. | Low pH (e.g., Glycine pH 2.0), High Salt (e.g., 2 M NaCl). Must be optimized for each specific ligand-analyte pair [17] [19] [18]. |
| Ligand | The molecule immobilized on the sensor surface, serving as the binding target. | Protein, antibody, DNA, etc. Can be covalently coupled or captured via tags (His, biotin) for a uniform orientation [19]. |
| Analyte | The molecule in solution that binds to the immobilized ligand. | Small molecule, protein, antibody, etc. Should be prepared in running buffer to avoid bulk refractive index shifts [19] [18]. |
Experimental Protocol: A Methodological Framework
This section outlines a general protocol for a kinetic characterization experiment using SPR.
Pre-Experimental Considerations
- Surface Selection: Choose a sensor chip and immobilization chemistry (covalent vs. capture) that ensures the ligand is biologically active and presents a uniform orientation. Capture methods (e.g., anti-His, streptavidin) often yield more reproducible data [19].
- Ligand Density: The amount of immobilized ligand is critical. Too high a density can cause mass transport limitation or steric hindrance, while too low a density may yield a weak signal. Aim for an Rₘₐₓ appropriate for your analyte size; for kinetic measurements, ~100 RU is often preferred [19].
- Analyte Concentration Series: Design a dilution series that brackets the expected KD. A range from 0.1 to 10 times the KD is optimal, spacing the sensorgrams evenly for reliable fitting [22]. Use at least five different analyte concentrations.
Step-by-Step Procedure
- System Preparation: Prime the SPR instrument microfluidic system with degassed, filtered running buffer until a stable baseline is achieved [18].
- Ligand Immobilization: Immobilize the ligand on one flow cell of the sensor chip using the chosen chemistry (e.g., amine coupling for covalent attachment or capture for tagged ligands). A reference flow cell (with no ligand or a non-interacting molecule) should be prepared for subtraction of non-specific binding and bulk effects [19].
- Analyte Injection Series: Using the automated fluidics, inject the series of analyte concentrations over both the ligand and reference surfaces. Use a sufficiently long association time to allow the higher concentrations to approach steady-state and a long enough dissociation time to reliably measure k𝒹 [19] [23].
- Surface Regeneration: After each analyte injection cycle, inject the optimized regeneration solution to remove all bound analyte and reset the surface for the next injection [17].
- Data Referencing: Subtract the sensorgram from the reference flow cell from the ligand flow cell sensorgram to correct for bulk refractive index changes and non-specific binding.
Data Analysis Workflow
- Quality Control: Visually inspect all sensorgrams for expected shapes (exponential association and dissociation), a stable baseline, and minimal noise or drift [21].
- Model Selection: Begin data fitting with the simplest model, the Langmuir 1:1 interaction model [20].
- Global Fitting: Perform a global fitting analysis across all analyte concentrations in the series, where kₐ and k𝒹 are shared (global) parameters for all curves, while Rₘₐₓ can be shared or local. This constrains the parameters and provides more reliable results [20].
- Validation: Assess the goodness-of-fit using statistical parameters like χ² (Chi-squared) and visually inspect the alignment of the fitted curves (red lines in software) with the experimental data [20].
Advanced Concepts and Troubleshooting
Even well-designed experiments can encounter issues. The table below outlines common problems and their solutions.
Table 3: Common SPR Experimental Issues and Recommended Solutions
| Problem | Possible Cause | Solution |
|---|---|---|
| Baseline Drift | Contamination on sensor chip or in fluidics; buffer instability; temperature fluctuations [18]. | Clean fluidic system and sensor chip; prepare fresh, degassed buffers; ensure instrument temperature is stable [18]. |
| Low Binding Signal | Analyte concentration too low; insufficient active ligand immobilized; low affinity interaction [18]. | Increase analyte concentration; optimize ligand immobilization level to increase Rₘₐₓ; verify ligand activity [18]. |
| Non-Specific Binding | Analyte interacting with the sensor matrix rather than the specific ligand [18]. | Include a proper reference surface; use a different sensor chip with a more inert surface (e.g., hydrophilic); add a non-ionic surfactant to the buffer; adjust buffer pH or ionic strength [18]. |
| Mass Transport Limitation | Ligand density too high, causing binding to be limited by analyte diffusion to the surface [21]. | Lower the immobilization level of the ligand; increase the flow rate during analyte injection [21] [20]. |
| Biphasic or "Bumpy" Curves | Heterogeneity in the ligand or analyte (e.g., multiple binding modes, impure sample) [21]. | Improve sample purity; use a capture-based immobilization for uniform ligand orientation; avoid over-interpreting complex models [21]. |
| Incomplete Regeneration | Regeneration buffer is too weak for the ligand-analyte complex. | Test a series of increasingly harsh regeneration buffers (e.g., lower pH, higher salt, with additives). Ensure the ligand remains active after regeneration [19]. |
The SPR sensorgram is a powerful, information-rich data stream that, when decoded, provides unparalleled insight into the dynamics of molecular interactions. A rigorous, step-by-step understanding of its phases—baseline, association, steady-state, dissociation, and regeneration—is fundamental to leveraging SPR technology effectively within label-free detection research. By adhering to robust experimental protocols, carefully selecting reagents, and applying critical data analysis and troubleshooting practices, researchers can reliably extract high-quality kinetic and affinity parameters. These parameters are indispensable for advancing fundamental biological understanding and for streamlining the development of new therapeutic agents.
SPR in Action: Methodologies and Cutting-Edge Applications in Biomedicine
Surface Plasmon Resonance (SPR) biosensors have established themselves as powerful analytical tools for label-free, real-time monitoring of biomolecular interactions, revolutionizing pharmaceutical research and diagnostic applications [25]. The core principle of SPR detection hinges on monitoring changes in the refractive index at the surface of a sensor chip, which occur when a target analyte binds to an immobilized ligand [1]. This label-free nature provides significant advantages over traditional methods, including the avoidance of fluorescent or radioactive labels that can alter molecular behavior and interfere with native conformational dynamics [2]. The technological leap to single-molecule sensitivity has further opened new frontiers in biotechnological applications, pushing the requirements for surface functionalization to unprecedented levels of precision [2].
The performance of an SPR biosensor critically depends on the meticulous design of the sensor chip surface [25]. This surface must accomplish two primary objectives: immobilize an adequate density of bio-recognition molecules to generate a detectable signal while concurrently minimizing non-specific interactions that compromise data reliability [25]. The sensor chip, often regarded as the heart of the SPR instrument, requires sophisticated functionalization strategies to transform a pristine gold film into a biologically active interface capable of specific molecular capture. Advances in surface chemistry have yielded numerous techniques for fabricating SPR sensor chips, each with distinct advantages in sensitivity, specificity, reusability, and applicability to different biological systems [25]. This guide provides a comprehensive technical overview of these strategies, focusing on their implementation, performance characteristics, and role in advancing label-free detection research.
Fundamental Immobilization Strategies
The immobilization of bioreceptors onto SPR sensor chips can be broadly categorized into two strategies: covalent coupling and affinity-based immobilization. Selecting the appropriate method involves balancing factors such as binding capacity, orientation control, stability, and regeneration potential.
Covalent Coupling involves forming stable chemical bonds between functional groups on the sensor surface and complementary groups on the ligand. This approach typically utilizes carboxyl or thiol chemistry to create robust linkages that withstand the flow conditions and regeneration steps common in SPR experiments [25]. A common implementation involves forming a self-assembled monolayer (SAM) of alkanethiols like 11-mercaptoundecanoic acid (11-MUA) on a gold sensor chip [26]. The carboxyl terminals of the SAM are then activated by reagents such as N-hydroxysuccinimide (NHS) and N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride (EDC), creating reactive esters that form stable amide bonds with primary amines (e.g., lysine residues) on proteins [26]. While this method provides high stability, its primary limitation is the random orientation of immobilized ligands, as reactive groups are distributed throughout the protein structure. This can potentially block active sites and reduce binding capacity.
Affinity-Based Immobilization leverages specific biological interactions to capture and orient ligands on the sensor surface. This strategy includes the use of Protein G, avidin-biotin, and nitrilotriacetic acid (NTA) systems [25]. For antibody ligands, Protein G is first covalently immobilized to the surface, providing a high-affinity binding site for the Fc region of antibodies [26]. This directional approach ensures that antigen-binding sites (paratopes) face the solution, maximizing their accessibility to analytes. Similarly, the strong interaction between avidin (or streptavidin) and biotin (dissociation constant KD ~10−15 M) provides a versatile platform for immobilizing biotinylated ligands with controlled density and orientation. These affinity methods generally yield surfaces with enhanced binding activity and consistency compared to random covalent attachment.
Table 1: Comparison of Fundamental Immobilization Strategies
| Strategy | Chemistry/Mechanism | Advantages | Disadvantages | Typical Applications |
|---|---|---|---|---|
| Covalent Coupling (Amine) | NHS/EDC activation of carboxyls to form amide bonds with primary amines | High stability, permanent attachment, applicable to most proteins | Random orientation, potential loss of activity, requires accessible amines | General protein immobilization |
| Covalent Coupling (Thiol) | Maleimide or pyridyldithiol reaction with cysteine thiol groups | Directed orientation if using site-specific cysteines | Requires engineered cysteine residues, lower stability than amide bonds | Site-specifically modified proteins |
| Protein G Mediation | Fc-binding Protein G captures antibodies | Optimal antibody orientation, high binding activity | Specific to antibodies (mainly IgG), requires pre-immobilization of Protein G | Antibody-antigen studies |
| Biotin-Avidin | Biotinylated ligand binds to avidin/streptavidin surface | Very strong binding, controlled density, versatile | Requires biotinylation of ligand, potential non-specific binding | DNA, carbohydrates, engineered proteins |
Advanced Functional Matrices and Materials
Beyond simple monolayers, advanced functional matrices provide three-dimensional architectures that increase ligand loading capacity and can enhance detection sensitivity. The most prevalent among these is the carboxymethylated dextran (CMD) hydrogel matrix, a polymer layer covalently attached to the gold sensor chip [25]. This hydrogel creates a hydrophilic environment that mimics physiological conditions and reduces non-specific binding of analytes. The porous structure of CMD allows for a significant increase in ligand density compared to two-dimensional surfaces, thereby amplifying the signal upon analyte binding. However, the hydrogel matrix can introduce mass transport limitations where diffusion of analyte into the matrix becomes rate-limiting, potentially affecting kinetic measurements. Furthermore, the depth of the matrix may render some binding events inaccessible to the evanescent field, which typically penetrates 200-300 nm into the solution [1].
Recent innovations in surface chemistry have introduced nitrilotriacetic acid (NTA)-functionalized platforms and zwitterionic coatings that outperform traditional CMD-based systems, particularly in complex biological matrices [25]. NTA surfaces chelate nickel or other transition metals to capture histidine-tagged recombinant proteins, providing a uniform orientation and gentle yet reversible immobilization. Zwitterionic coatings, composed of molecules with both positive and negative charges, create a super-hydrophilic surface that effectively resists protein fouling from complex samples like serum or cell lysates. This antifouling property is crucial for applications in clinical diagnostics where specific signals must be distinguished from substantial background noise. The integration of nanomaterials such as gold nanoparticles, graphene, and metal-organic frameworks (MOFs) has further advanced sensor capabilities by increasing the surface area for immobilization and enhancing the local plasmonic effect, leading to improved sensitivity [27] [28].
Quantitative Comparison of Immobilization Performance
The choice of immobilization strategy profoundly impacts key performance metrics in SPR biosensing, including binding affinity (KD), limit of detection (LOD), and regeneration capability. A comparative study on the detection of Shiga toxin provides a compelling quantitative illustration of these differences [26].
In this study, a covalent, non-oriented approach using an 11-MUA-modified chip yielded a dissociation constant (KD) of 37 nM and a limit of detection (LOD) of 28 ng/mL [26]. In contrast, a Protein G-assisted oriented immobilization strategy dramatically improved performance, achieving a 2.3-fold higher binding affinity (KD = 16 nM) and a 2.9-fold lower detection limit (LOD = 9.8 ng/mL) [26]. Control measurements of free antibody-antigen interactions in solution established a baseline affinity (KD = 10 nM), demonstrating that the oriented method preserved 63% of the native binding efficiency, compared to only 27% preservation in the covalent approach [26]. This significant enhancement is attributed to Protein G's ability to maintain optimal antibody orientation, thereby maximizing paratope accessibility, minimizing steric interference, and preserving binding site functionality.
Table 2: Quantitative Performance of Immobilization Strategies for Shiga Toxin Detection [26]
| Immobilization Method | Dissociation Constant (KD) | Limit of Detection (LOD) | Preserved Binding Efficiency | Key Characteristics |
|---|---|---|---|---|
| Solution Baseline (Control) | 10 nM | Not Applicable | 100% (Reference) | Represents ideal, unhindered interaction |
| Protein G-Oriented | 16 nM | 9.8 ng/mL | 63% | Optimal orientation, high accessibility |
| Covalent (Non-Oriented) | 37 nM | 28 ng/mL | 27% | Random orientation, steric hindrance possible |
These findings underscore a critical principle in sensor surface functionalization: orientation is paramount for ligand performance. The random nature of covalent immobilization often blocks a significant proportion of active sites or induces structural strain, while affinity-based orientation presents ligands in their natural, receptive conformation. This principle extends beyond antibodies to other bioreceptors, including enzymes, receptors, and nucleic acids, where controlled presentation enhances binding capacity and assay sensitivity.
Detailed Experimental Protocol: Protein G-Mediated Antibody Immobilization
The following section provides a detailed, citable protocol for implementing the high-performance Protein G-mediated antibody orientation strategy, as utilized in the Shiga toxin detection study [26]. This protocol can be adapted for various antibody-antigen systems.
Reagents and Materials
- SPR sensor chip (e.g., gold disk with titanium adhesion layer)
- 11-mercaptoundecanoic acid (11-MUA)
- Absolute ethanol
- Protein G
- NHS (N-hydroxysuccinimide)
- EDC (N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide hydrochloride)
- Ethanolamine hydrochloride
- Antibody solution (e.g., anti-target IgG)
- Coupling buffer: 10 mM acetate buffer, pH 4.5
- Running buffer: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% (v/v) Tween 20, pH 7.4
- Regeneration buffer: 15 mM NaOH with 0.2% (w/v) SDS
Step-by-Step Procedure
Sensor Chip Cleaning: Thoroughly clean the SPR gold chip surface using a freshly prepared piranha solution (3:1 v/v H₂SO₄:30% H₂O₂; Caution: highly corrosive). Rise extensively with deionized water and absolute ethanol, then dry under a stream of nitrogen.
Self-Assembled Monolayer (SAM) Formation: Immerse the cleaned chip in a 1 mM solution of 11-MUA in absolute ethanol. Allow the SAM to form overnight at room temperature. Wash the chip three times with absolute ethanol and three times with deionized water to remove unbound thiols, then dry under nitrogen.
SPR Instrument Priming: Insert the functionalized chip into the SPR instrument. Perform optical alignment and verify no leakage between fluidic channels. Stabilize the surface by flowing acetate coupling buffer for 45 minutes.
Surface Activation: Inject a freshly prepared mixture of 400 mM EDC and 100 mM NHS over the SAM surface for 300 seconds (5 minutes). This step activates the carboxyl groups of 11-MUA to form NHS esters.
Protein G Immobilization: Inject a solution of Protein G (25 µg/mL in acetate buffer) over the activated surface for 900 seconds (15 minutes). The NHS esters will form stable amide bonds with primary amines on Protein G.
Blocking Residual Esters: Inject 1 M ethanolamine (pH 8.5) over the surface for 600 seconds (10 minutes) to deactivate and block any remaining active esters.
Antibody Capture: Introduce the antibody solution (e.g., 40 µg/mL in running buffer) as the secondary ligand. This allows the formation of oriented antibody/Protein G complexes through specific Fc-region binding. No covalent bond is formed between the antibody and the surface at this stage.
Regeneration (Optional): After a complete binding analysis cycle, the antibody layer can often be regenerated using a brief injection (e.g., 120 seconds) of regeneration buffer to remove the bound antigen, leaving the Protein G and antibody layer intact for subsequent analysis cycles.
The workflow for this protocol is visualized in the following diagram:
Diagram 1: Protein G-mediated antibody immobilization workflow. The process begins with a clean gold chip, proceeds through surface functionalization with a self-assembled monolayer (SAM), activation, Protein G immobilization, blocking, and final antibody capture, resulting in a ready-to-use SPR sensor surface with optimally oriented antibodies.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of SPR surface functionalization requires a set of core chemical reagents and materials. The following table details these essential components and their specific functions in the immobilization process.
Table 3: Essential Research Reagents for SPR Surface Functionalization
| Reagent/Material | Function in Functionalization | Key Considerations |
|---|---|---|
| 11-Mercaptoundecanoic Acid (11-MUA) | Forms a carboxyl-terminated self-assembled monolayer (SAM) on gold surfaces, providing functional groups for subsequent coupling. | Creates a stable, ordered monolayer. Ethanol is the preferred solvent for SAM formation [26]. |
| NHS (N-hydroxysuccinimide) | Activates carboxyl groups to form amine-reactive NHS esters, enabling efficient formation of amide bonds with primary amines. | Typically used in combination with EDC. Fresh preparation is critical for high activation efficiency [26]. |
| EDC (N-(3-dimethylaminopropyl)-N'-ethylcarbodiimide) | A carbodiimide crosslinker that catalyzes the formation of amide bonds between carboxyl and amine groups. | Unstable in aqueous solution; must be prepared fresh immediately before use [26]. |
| Protein G | An immunoglobulin-binding protein that captures antibodies via their Fc region, ensuring proper orientation for antigen binding. | Superior for orienting a wide range of IgG antibody subtypes. Pre-immobilization is required [26]. |
| Ethanolamine | A small amine-containing molecule used to block unreacted NHS esters after ligand immobilization, preventing non-specific binding. | Effective at high concentration (e.g., 1 M, pH 8.5) for complete deactivation of active esters [26]. |
| Carboxymethylated Dextran (CMD) | A hydrogel polymer that creates a three-dimensional matrix on the sensor surface, increasing ligand loading capacity. | Can introduce mass transport limitations for kinetic analysis but amplifies signal response [25]. |
Strategic Selection and Future Perspectives
Choosing the optimal immobilization strategy is a critical decision point in SPR assay development. The following decision pathway visualizes the key considerations for selecting a functionalization strategy based on the nature of the ligand and the assay objectives:
Diagram 2: A strategic decision pathway for selecting an immobilization chemistry. The choice is guided by the properties of the ligand (e.g., antibody, biotin tag, His-tag) and the requirement for binding orientation versus maximum stability.
The future of SPR surface functionalization is being shaped by several emerging trends. The integration of artificial intelligence (AI) and machine learning for real-time data interpretation and predictive modeling of drug-target interactions is on the rise [25]. Miniaturized SPR chips and lab-on-a-chip systems are being developed for in situ analysis, potentially enabling in vivo real-time monitoring of pharmacokinetics [25]. Furthermore, the development of multifunctional biochips that combine SPR with complementary techniques such as electrochemistry and mass spectrometry provides richer, multi-parametric data from a single experiment [25]. As these technologies mature, the demand for robust, highly specific, and reproducible surface functionalization strategies will only intensify, solidifying their role as a cornerstone of label-free biosensing research.
In conclusion, the strategic functionalization of SPR sensor surfaces is a precise science that directly determines the success of label-free biomolecular interaction analysis. By understanding the principles, trade-offs, and protocols outlined in this guide, researchers can make informed decisions to design surfaces that maximize data quality and drive scientific discovery in drug development and diagnostic applications.
Surface Plasmon Resonance (SPR) has established itself as a cornerstone technology for label-free, real-time analysis of biomolecular interactions. This technical guide delves into the application of SPR for determining the critical kinetic parameters—association rate (kon), dissociation rate (koff), and the equilibrium dissociation constant (KD)—that define the dynamics and affinity of these interactions. Framed within the context of advancing label-free detection research, this whitepaper provides researchers and drug development professionals with a comprehensive overview of the fundamental principles, detailed experimental protocols, and robust data analysis methods required to generate high-quality, reproducible kinetic data. By moving beyond ensemble averaging and observing interactions as they happen, SPR offers unparalleled insights into the mechanisms of biological binding events, making it an indispensable tool in modern biophysics and drug discovery.
Surface Plasmon Resonance (SPR) is an optical technique that enables the real-time, label-free monitoring of biomolecular interactions [19] [29]. The phenomenon was first successfully applied in a biosensor in 1983, and the first commercial SPR instrument was launched by Biacore, paving the way for its widespread adoption [29]. The core principle involves the generation of surface plasmons—collective oscillations of free electrons—at the interface between a metal (typically gold) and a dielectric medium (e.g., a buffer solution) when illuminated by polarized light under specific conditions [29]. The resonance angle or wavelength at which this phenomenon occurs is exquisitely sensitive to changes in the refractive index within the immediate vicinity of the sensor surface. When a biomolecule (the "analyte") binds to its interaction partner (the "ligand") immobilized on the sensor chip, it causes a change in the local refractive index, leading to a shift in the resonance signal. This shift is measured in Resonance Units (RU) and plotted in real-time to generate a sensorgram, a kinetic trace of the entire binding event [19] [30].
The label-free nature of SPR is its most significant advantage. Unlike fluorescent or radioactive labeling, which can potentially alter the conformation, activity, or binding properties of biomolecules—particularly critical for small molecules—SPR allows for the observation of interactions in their native state [2]. This provides a more biologically relevant context and eliminates the time-consuming and often costly labeling steps. Furthermore, SPR's capacity for real-time monitoring reveals the kinetics of the interaction—the rates of association (kon) and dissociation (koff)—in addition to the binding affinity (KD), offering a deeper understanding of the interaction mechanism that is invisible to endpoint binding assays [19] [31].
Theoretical Foundations of Kinetic Analysis
The determination of kinetic parameters via SPR is grounded in the principles of biomolecular interaction kinetics. The most fundamental model for a 1:1 interaction is described by the Langmuir binding model:
[ L + A \mathrel{\mathop{\rightleftharpoons}^{k{on}}{k_{off}}} LA ]
Where ( L ) represents the immobilized ligand, ( A ) is the flowed analyte, ( LA ) is the formed complex, ( k{on} ) is the association rate constant (M-1s-1), and ( k{off} ) is the dissociation rate constant (s-1}) [32].
From the sensorgram, two primary phases are analyzed:
- Association Phase: The rate of complex formation during analyte injection is governed by the equation: [ \frac{dR}{dt} = k{on} \cdot C \cdot (R{max} - R) - k{off} \cdot R ] where ( R ) is the response at time ( t ), ( C ) is the analyte concentration, and ( R{max} ) is the maximum binding response at saturation [32].
- Dissociation Phase: The rate of complex breakdown after analyte injection stops is given by: [ \frac{dR}{dt} = - k_{off} \cdot R ]
The ratio of the kinetic rate constants directly yields the equilibrium dissociation constant, a measure of binding affinity: [ KD = \frac{k{off}}{k_{on}} ] A lower KD indicates a higher affinity interaction [19] [30]. Separating KD into its kinetic components provides crucial mechanistic insights; for instance, a long-lasting interaction (high affinity) can be due to fast association (high kon) or slow dissociation (low koff), with the latter often being more desirable for drug candidates as it correlates with longer target residence time [31].
Experimental Design and Methodologies
A successful SPR kinetic experiment requires meticulous planning and optimization at every stage.
Immobilization Strategies
The first critical step is the stable and functional immobilization of the ligand onto the sensor chip surface. The chosen method can significantly impact the quality and interpretability of the data.
- Covalent Coupling: A standard approach uses a CM5 dextran chip with amine coupling via NHS/EDC chemistry. While robust, this can lead to heterogeneous attachment if the ligand has multiple amine groups, potentially masking binding sites or affecting activity [19].
- Capture Methods: To ensure a uniform, oriented presentation, capture techniques are often preferred. These utilize high-affinity tags fused to the ligand:
- His-Tag: Captured on NTA chips via nickel chelation.
- Biotin: Captured on streptavidin (SA) chips.
- Antibody Fc: Captured on protein A or protein G chips. These methods generally preserve a higher ligand activity and allow for more controlled surface regeneration [19].
Experimental Kinetics Formats
There are two primary experimental formats for collecting kinetic data, each with distinct advantages and applications.
Table 1: Comparison of Multi-Cycle Kinetics (MCK) and Single-Cycle Kinetics (SCK)
| Feature | Multi-Cycle Kinetics (MCK) | Single-Cycle Kinetics (SCK) |
|---|---|---|
| Workflow | Each analyte concentration is injected in a separate cycle, followed by surface regeneration. | Increasing analyte concentrations are injected sequentially in a single cycle without regeneration between steps. |
| Regeneration | Required after every injection, which can damage the ligand over time. | Only one regeneration step is needed at the very end, or none if a new chip is used. |
| Throughput | Lower, due to multiple regeneration and re-equilibration steps. | Higher analysis speed for a given interaction. |
| Ligand Stability | Less suitable for ligands sensitive to regeneration conditions. | Ideal for ligands that are difficult to regenerate or inactivate. |
| Data Quality | Provides multiple, independent binding curves for each concentration, aiding in diagnosis. | Offers a single, continuous dataset but is more susceptible to artifacts from a single poor injection [31]. |
Critical Buffer Considerations
The composition of the running buffer is paramount for obtaining biologically relevant data.
- pH and Ions: The buffer must maintain a pH and ionic strength that preserves the native conformation and activity of the interactants [19].
- Additives: For proteins requiring co-factors (e.g., ATP, magnesium), these must be included to ensure the correct oligomeric state and functionality [19].
- Solvent Matching: When analyzing small molecules dissolved in organic solvents like DMSO, the percentage of solvent must be perfectly matched in all analyte samples and the running buffer to avoid significant bulk refractive index shifts that can obscure the binding signal [19].
The diagram below illustrates the core workflow of an SPR experiment for kinetic analysis.
Data Fitting and Kinetic Modeling
Extracting kinetic parameters from sensorgrams requires fitting the data to an appropriate interaction model. The 1:1 Langmuir binding model is the simplest and should be the starting point [32]. However, the data must be of high quality to yield a reliable fit.
Pre-Fitting Data Processing
Before fitting, the raw sensorgram data must be processed:
- Reference Subtraction: The signal from a reference flow cell (with no ligand or an irrelevant ligand) is subtracted to correct for non-specific binding and bulk refractive index effects.
- Double Referencing: Further subtraction of the signal from a blank (buffer) injection helps eliminate systematic noise and instrument drift [32].
Global Fitting and Parameter Interpretation
In global fitting, the sensorgrams from all analyte concentrations are fitted simultaneously to a single model, with kon and koff shared (fitted globally) across all curves. This provides a more robust and reliable determination of kinetic constants compared to local fitting of individual curves [32].
Table 2: Key Parameters in SPR Kinetic Fitting
| Parameter | Symbol | Description | Fitting Method |
|---|---|---|---|
| Association Rate Constant | kon | Rate of complex formation. Depends on ligand/analyte properties and buffer conditions. | Global |
| Dissociation Rate Constant | koff | Rate of complex breakdown. Depends on ligand/analyte properties and buffer conditions. | Global |
| Maximum Response | Rmax | Theoretical response at saturation, proportional to the molecular weight ratio of analyte and ligand and the amount of immobilized ligand. | Global for a single analyte |
| Bulk Refractive Index | RI | Artifact from buffer mismatch between sample and running buffer. Should be small. | Local |
| Drift | - | Slow baseline shift, often from poor surface equilibration. Contribution should be minimal. | Local |
| Chi-Squared | χ² | Measure of the goodness-of-fit. Lower values indicate a better fit. | - |
Addressing Complex Binding Models
If the 1:1 model fails to fit the data well (evidenced by high χ² values and systematic patterns in the residuals), more complex models may be considered, but only after exhaustive optimization of experimental conditions [32]. Potential complex models include:
- Heterogeneity Model: Accounts for a population of ligands with different binding affinities.
- Bivalent Analyte Model: For analytes with two binding sites.
- Conformational Change Model: For interactions where binding induces a structural change in the ligand or analyte.
It is critical to avoid "model shopping"; the best practice is to optimize the experiment to achieve a 1:1 binding profile whenever possible [32].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for SPR Kinetic Studies
| Item | Function in SPR Experiment |
|---|---|
| Sensor Chips (e.g., CM5, NTA, SA) | The solid support with a gold film and specialized coating for ligand immobilization. |
| Running Buffer (e.g., HEPES, PBS) | The continuous phase that carries the analyte; maintains pH and ionic strength to preserve biomolecule activity. |
| Regeneration Solution (e.g., Glycine pH 2.0, 2 M NaCl) | A solution that breaks the ligand-analyte complex without damaging the ligand, allowing for surface re-use. |
| Capture Ligands (e.g., His-tagged Protein, Biotinylated Antibody) | The molecule of interest immobilized on the sensor chip surface. |
| Analytes | The binding partner flowed over the immobilized ligand in a concentration series. |
| Coupling Reagents (e.g., NHS/EDC) | Chemicals used to activate the chip surface for covalent ligand immobilization. |
| Membrane Scaffold Protein (MSP) & Lipids | For creating lipid nanodiscs, which allow for the study of membrane-protein interactions in a more native environment [19]. |
Advanced Applications and Emerging Trends
The principles of SPR kinetic analysis are being applied and enhanced through new technological developments. Localized Surface Plasmon Resonance (LSPR) utilizes metal nanoparticles instead of a continuous metal film. The resonant oscillation, or LSPR, is highly sensitive to local refractive index changes and can be tuned by the nanoparticle's size, shape, and composition [2] [33] [29]. LSPR-based biosensors are a cornerstone of the growing field of nanoplasmonic biosensors, which leverage nanomaterials to achieve ultra-sensitive detection, portability, and potential for high-throughput analysis [29].
These advanced platforms are pushing the boundaries of label-free research. For example, LSPR biosensors have been developed for the sensitive detection of small molecules like Ochratoxin-A in complex matrices such as wine, showcasing their applicability in food safety [33]. Furthermore, techniques like interference microscopy (iSCAT) are now achieving true label-free single-molecule sensitivity, allowing for the detection and mass measurement of individual proteins, acting as an "optical analog of mass spectrometry" [2]. The integration of SPR with microscopy (SPRM) further enables the visualization of binding events on whole cells with spatial resolution, allowing for the study of membrane protein kinetics in their native cellular environment [34]. These advancements collectively highlight the dynamic and evolving role of plasmonic technologies in deepening our understanding of biomolecular kinetics.
Surface Plasmon Resonance (SPR) has established itself as a cornerstone technology in modern drug discovery by enabling real-time, label-free analysis of biomolecular interactions [35]. This technique operates by detecting minute changes in the refractive index at a sensor surface, allowing researchers to monitor binding events as they happen without the need for fluorescent or radioactive labels that can potentially interfere with molecular interactions [2] [36]. The absence of labeling not only preserves the native state of the molecules under study but also significantly reduces assay development time and eliminates artifacts that often complicate data interpretation from label-based methods [37].
The pharmaceutical and biotechnology industries are increasingly adopting SPR technology throughout the drug discovery pipeline, driven by its unparalleled ability to provide detailed kinetic and affinity data [38]. As of 2024, SPR accounted for approximately 46.59% of the label-free detection market revenue, maintaining its leadership position based on superior sensitivity and regulatory familiarity [37]. The global label-free detection market, valued at $2.14 billion in 2024, is projected to reach $4.48 billion by 2032, reflecting a compound annual growth rate of 9.8% [38]. This growth is largely fueled by rising investments in biologics development, the need for high-throughput screening to eliminate label-based assay artifacts, and the increasing adoption of fragment-based drug discovery approaches that heavily rely on sensitive detection methods like SPR [37].
Technical Foundation of SPR Technology
Fundamental Principles of SPR
Surface Plasmon Resonance functions through the generation of surface plasmon waves at a metal-dielectric interface, typically a gold-coated glass sensor chip [4]. When polarized light strikes this interface under total internal reflection conditions, it excites surface plasmons—collective oscillations of free electrons at the metal surface—resulting in a characteristic drop in reflected light intensity at a specific angle known as the resonance angle [2] [4]. This resonance angle is exquisitely sensitive to changes in the refractive index within the immediate vicinity of the sensor surface (approximately within 300 nm) [2].
When biomolecular binding occurs on the sensor surface, the accumulated mass alters the local refractive index, causing a proportional shift in the resonance angle that can be measured in real-time [36]. This shift is recorded in resonance units (RU) over time, generating a sensorgram that provides a complete profile of the molecular interaction, including association, steady-state equilibrium, and dissociation phases [36]. The technology can detect interactions across an extensive affinity range from femtomolar (fM) to millimolar (mM) concentrations, with dissociation rate constants (kₐ) ranging from 10⁻⁵ to 1 s⁻¹ [36].
Comparative Advantages in Fragment Screening
The application of SPR in fragment-based drug discovery is particularly valuable due to its ability to detect weak interactions with low molecular weight compounds [39]. Traditional screening methods often struggle with fragments that typically bind with millimolar affinities, but SPR's sensitivity enables researchers to identify these weak binders that serve as critical starting points for drug development [39] [40]. The technology's capacity to analyze crude reaction mixtures without purification further accelerates the hit-to-lead optimization process, significantly reducing the time and resources required for traditional compound synthesis and purification [40].
Recent advancements in SPR instrumentation have substantially improved throughput capabilities. Modern systems can screen libraries of thousands of fragments while consuming minimal sample material—as little as 75μg of each protein—making the technology accessible even for targets that are difficult to express or purify in large quantities [36]. The implementation of SPR imaging (SPRi) has further enhanced throughput by enabling simultaneous analysis of thousands of immobilized fragments on microarray formats, with some systems capable of screening up to 9,216 compounds per array [39].
Fragment Screening Applications
Experimental Design and Library Considerations
The successful implementation of SPR biosensor-based fragment screening requires careful consideration of library design and experimental validation. In a landmark study documented in SLAS Discovery, researchers designed and validated a fragment library comprising 930 compounds selected from 4.6 million commercially available molecules using rigorous physicochemical and medicinal chemistry filters [41]. This library was systematically screened against three prototypical drug targets—HIV-1 protease, thrombin, and carbonic anhydrase—along with a non-target protein, human serum albumin, to assess specificity [41].
Under optimized screening conditions, compound solubility presented no significant challenges, and the high sensitivity of the SPR sensor surfaces enabled detection of interactions for 35% to 97% of the fragments, depending on the specific target protein [41]. This comprehensive validation approach proved invaluable for identifying promiscuous binders that could potentially generate false positives in downstream assays. Notably, none of the fragments demonstrated promiscuous binding behavior (defined as interacting with a stoichiometry ≥5:1 with all four proteins), and only two compounds exhibited slow dissociation from all tested proteins [41].
Protocol for SPR-Based Fragment Screening
Step 1: Surface Preparation
- Immobilize the target protein on a CM5 sensor chip using standard amine coupling chemistry
- Achieve immobilization levels between 5,000-15,000 RU for optimal detection
- Prepare a reference surface for background subtraction
Step 2: Sample Preparation
- Prepare fragment solutions at 0.1-1 mM concentration in running buffer
- Include 1-5% DMSO to maintain solubility and match running buffer conditions
- Centrifuge samples prior to injection to remove particulate matter
Step 3: Screening Cycle
- Inject fragments for 30-60 seconds at a flow rate of 30 μL/min
- Monitor association phase for 60-120 seconds
- Monitor dissociation phase for 60-180 seconds
- Regenerate surface with a 30-second pulse of regeneration solution (e.g., 10 mM glycine, pH 2.5)
- Allow adequate stabilization time between cycles
Step 4: Data Analysis
- Reference subtract all sensorgrams
- Identify hits based on significant response exceeding background threshold
- Classify hits according to binding profile and stoichiometry
Table 1: Key Performance Metrics for SPR Fragment Screening
| Parameter | Typical Range | Optimal Conditions |
|---|---|---|
| Fragment MW | 150-300 Da | <350 Da |
| Affinity Range | μM-mM | 100 μM - 10 mM |
| DMSO Tolerance | 1-5% | ≤2% |
| Immobilization Level | 5,000-15,000 RU | 10,000 RU |
| Hit Rate | 0.1-5% | 0.5-3% |
Data Interpretation and Hit Validation
Primary screening data from SPR fragment studies requires careful interpretation to distinguish genuine binding events from non-specific interactions. Dose-response experiments should be conducted for confirmed hits to determine steady-state affinity (KD) values. Additionally, kinetic analysis provides valuable information about binding mechanisms, though the fast dissociation rates of many fragments may limit accurate determination of individual rate constants [40].
The integration of crude reaction mixture (CRM) analysis represents a significant advancement in fragment-to-hit optimization. This approach allows for rapid evaluation of analogue compounds without time-consuming purification steps, dramatically accelerating structure-activity relationship (SAR) exploration [40]. By measuring the off-rate (kₒff) of compounds from CRMs, researchers can efficiently prioritize synthetic efforts toward derivatives with improved binding characteristics, particularly those exhibiting slower dissociation rates that often correlate with enhanced affinity [40].
Antibody Characterization Applications
Comprehensive Kinetic Profiling
SPR technology has become an indispensable tool for antibody characterization, providing complete kinetic profiles that include association rate (kₐ), dissociation rate (k𝒅), and overall binding affinity (KD) [36]. These parameters are critical for evaluating therapeutic antibody candidates, as they directly influence in vivo efficacy, dosing regimens, and potential immunogenicity [37]. The ability to monitor interactions in real-time using label-free detection allows researchers to observe complex binding mechanisms that might be obscured in endpoint assays, including conformational changes and multivalent interactions [36].
For bispecific antibodies and antibody-drug conjugates (ADCs), which represent a rapidly growing segment of biologics with a pipeline valued at USD 150 billion, SPR provides essential characterization data to confirm correct chain pairing and mitigate off-target effects [37]. The technology can quantify binding stoichiometry and cooperative interactions that fluorescent tags might distort, making it particularly valuable for characterizing complex biologics [37]. Contract research organizations (CROs) have reported a 40% rise in biologics-related kinetic characterization requests, underscoring the growing importance of detailed antibody profiling in therapeutic development [37].
Epitope Binning and Mapping
A critical application of SPR in antibody development is epitope binning, which groups monoclonal antibodies based on their binding to similar regions (epitopes) on the target antigen [36]. This information is invaluable for selecting complementary antibody pairs for diagnostic assays or identifying diverse therapeutic candidates with potentially synergistic mechanisms of action.
The standard sandwich assay format for epitope binning involves:
- Immobilizing the antigen to the sensor surface
- Saturating with a first monoclonal antibody
- Assessing binding of a second monoclonal antibody without regeneration
- Classifying antibodies into bins based on their ability to bind simultaneously or competitively
Advanced SPR systems can perform high-throughput epitope binning studies on hundreds of antibodies in a single automated run, significantly accelerating candidate selection processes [36].
Protocol for Antibody Affinity and Kinetic Characterization
Step 1: Surface Preparation
- Immobilize antigen using amine coupling (approximately 5,000 RU)
- Alternatively, capture antibody using anti-Fc specific surface for more oriented immobilization
- Prepare reference surface with irrelevant protein or blank immobilization
Step 2: Concentration Series Design
- Prepare 3-5-fold serial dilutions of antibody covering range above and below expected KD
- Include a zero concentration (buffer only) for double referencing
- Maintain consistent DMSO concentration across all dilutions if applicable
Step 3: Binding Analysis
- Inject antibody concentrations in random order to minimize systematic bias
- Use association time of 2-5 minutes depending on observed kinetics
- Monitor dissociation for 10-30 minutes to accurately determine k𝒅
- Regenerate surface with appropriate conditions between cycles
Step 4: Data Processing
- Reference subtract all sensorgrams
- Perform double referencing by subtracting buffer injection
- Fit processed data to 1:1 binding model or more complex models as needed
- Evaluate goodness of fit using residual analysis and chi-squared values
Table 2: Key Assay Parameters for Antibody Characterization
| Parameter | Typical Range | Impact on Assay Quality |
|---|---|---|
| Immobilization Level | 50-100 nM | Higher density may cause mass transport limitation |
| Flow Rate | 30-100 μL/min | Lower flow rates enhance mass transport limited conditions |
| Contact Time | 2-5 minutes | Longer injection for slow kₐ |
| Dissociation Time | 10-60 minutes | Longer dissociation for accurate k𝒅 for slow dissociating binders |
| Regeneration | pH 1.5-3.0 or high salt | Must fully regenerate without damaging the surface |
Lead Optimization Applications
Kinetic Parameter-Driven Optimization
The lead optimization phase benefits tremendously from SPR-derived kinetic parameters that guide medicinal chemistry efforts [40]. Unlike affinity measurements alone, which provide a composite view of binding strength, the individual association and dissociation rates offer insights into the structural determinants of the interaction that can be exploited for compound improvement [40]. The dissociation rate (kₒff) has emerged as a particularly valuable parameter during hit optimization, as improvements in affinity are frequently driven by a slower kₒff rather than a faster association rate [40].
A significant advantage of SPR in lead optimization is the ability to analyze crude reaction mixtures (CRMs) without purification, dramatically accelerating the exploration of structure-activity relationships [40]. This approach was successfully demonstrated in a study targeting Hsp90 and PDHK2, where 83 different CRMs were evaluated without purification of the reaction products [40]. The research confirmed that the kₒff rate, being concentration-independent, serves as a valid surrogate for affinity measurements in these unpurified mixtures, enabling rapid prioritization of synthetic targets [40].
Protocol for CRM Analysis in Lead Optimization
Step 1: Library Design
- Select starting material (SM) with confirmed binding to target
- Design CRMs through one-step synthesis with diverse building blocks
- Determine product yield in CRMs by LCMS prior to SPR analysis
Step 2: SPR Screening of CRMs
- Immobilize target protein on sensor chip
- Inject CRMs without purification at single concentration (e.g., 50-100 μM)
- Focus on dissociation phase to determine kₒff
- Include positive (SM) and negative controls in each run
Step 3: Data Interpretation
- Compare kₒff of CRM to starting material
- Prioritize CRMs with significantly slower kₒff
- Correlate structural modifications with kinetic improvements
Step 4: Hit Confirmation
- Resynthesize and purify selected hits from prioritized CRMs
- Determine full kinetic profile (kₐ, k𝒅, KD) for purified compounds
- Validate binding mode through structural methods (X-ray crystallography)
This integrated approach has demonstrated the potential to shorten hit-to-lead timelines from the conventional 18-24 months to just 6-12 months, enabling more efficient progression to first-in-class drug candidates [37] [40].
Emerging Technologies and Future Directions
Advanced SPR Platforms
The field of label-free detection continues to evolve with several emerging technologies enhancing the capabilities of traditional SPR systems. Localized Surface Plasmon Resonance (LSPR) based on metallic nanoparticles offers significantly reduced probing volumes and enhanced sensitivity for detecting small molecules [2] [33]. These systems have demonstrated remarkable performance in practical applications, such as a recently developed LSPR biosensor for detecting Ochratoxin-A in wine with a linear dynamic response from 0.05 to 2 ng mL⁻¹ and a detection limit of 7 pg mL⁻¹ [33].
Metasurface Plasmon Resonance (MetaSPR) represents another technological advancement that integrates SPR with localized SPR using periodic nanoarrays to achieve direct light-plasmon coupling without the need for prisms [4]. This innovation has enabled the development of high-throughput, real-time cell analysis platforms for monitoring cell proliferation, drug-target interactions, and immune cell-mediated cytotoxicity [4]. The compatibility of MetaSPR with standard 96-well plate formats facilitates automated, high-throughput screening while reducing costs compared to traditional SPR systems [4].
Integration with Complementary Technologies
The growing integration of artificial intelligence and machine learning with SPR data analysis represents a significant trend in the field [37] [38]. AI-assisted kinetic modeling can recommend assay settings, suggest structural modifications, and identify allosteric binding modes that might escape manual inspection [37]. Platforms like HydraScreen have demonstrated remarkable performance, achieving a 15.9% hit rate in the top 1% of ranked molecules and significantly outperforming legacy docking workflows [37].
The combination of SPR with other biophysical and structural techniques creates powerful workflows for comprehensive drug discovery. For instance, SPR screening of fragments or lead compounds can be seamlessly integrated with X-ray crystallography for structural validation, as demonstrated in the XChem facility at the Diamond synchrotron, which enables high-throughput crystallographic screening of fragment libraries [40]. Similarly, the combination of SPR with hydrogen-deuterium exchange mass spectrometry (HDX-MS) provides complementary information about binding sites and conformational changes induced by compound binding [36].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for SPR-Based Drug Discovery
| Reagent/Material | Function | Application Notes |
|---|---|---|
| CM5 Sensor Chip | Carboxymethylated dextran matrix for immobilization | Standard surface chemistry; suitable for most protein targets |
| NTA Sensor Chip | Nitrilotriacetic acid surface for His-tag capture | Reversible immobilization; maintains protein orientation |
| HPA Sensor Chip | Hydrophobic alkane thiol surface | Suitable for membrane protein studies |
| Amine Coupling Kit | Contains EDC, NHS, and ethanolamine for covalent immobilization | Standard chemistry for protein immobilization |
| Regeneration Solutions | Various pH and additive formulations | Screen for optimal conditions for each target |
| Running Buffers | HBS-EP, PBS-P, others with surfactant | Minimize non-specific binding; maintain protein stability |
| CRMs (Crude Reaction Mixtures) | Unpurified synthesis products | Accelerate SAR in lead optimization [40] |
Surface Plasmon Resonance continues to evolve as a powerful label-free technology that addresses critical needs throughout the drug discovery pipeline. From initial fragment screening through detailed antibody characterization and lead optimization, SPR provides invaluable kinetic and affinity data that guide decision-making in therapeutic development. The integration of emerging technologies such as LSPR, MetaSPR, and AI-assisted data analysis, combined with innovative approaches like CRM screening, promises to further enhance the efficiency and effectiveness of drug discovery. As the field advances, SPR technology will undoubtedly remain at the forefront of label-free detection methods, enabling researchers to overcome existing challenges and accelerate the development of novel therapeutics.
Surface Plasmon Resonance (SPR) biosensors have emerged as powerful analytical tools for label-free, real-time monitoring of biomolecular interactions in pharmaceutical and clinical research [25]. These optical devices enable the study of dynamic processes like protein-protein, antibody-antigen, protein-nucleic acid, and ligand-receptor interactions without the need for fluorescent or radioactive labeling [25]. The fundamental principle underlying SPR biosensing involves the excitation of surface plasmons—coherent oscillations of free electrons at the interface between a noble metal (typically gold) and a dielectric medium (such as a buffer solution) [42]. When biomolecular binding events occur on the functionalized sensor surface, they induce localized changes in the refractive index (RI) adjacent to the metal surface, which subsequently affect the light propagation constant [25].
The advantages of SPR biosensors include their label-free detection capability, real-time monitoring, high sensitivity, and minimal sample requirements [25]. Since their inception in biosensing during the 1990s, SPR technologies have evolved significantly and are now extensively utilized across numerous applications, including medical diagnostics, drug discovery, environmental monitoring, and food safety [25]. In medical diagnostics specifically, SPR biosensors have demonstrated remarkable potential for early cancer detection, biomarker analysis, and therapeutic monitoring, positioning them as transformative tools for improving patient outcomes through earlier and more accurate disease diagnosis [42] [43].
Fundamental Principles of SPR-Based Label-Free Detection
Optical Phenomenon and Biosensing Mechanism
The core principle of SPR biosensing relies on detecting alterations in the refractive index of the medium adjacent to a metallic surface where bioreceptors are immobilized [25]. In a typical Kretschmann configuration, which is the most common experimental setup, monochromatic, plane-polarized light is directed through a prism onto a thin gold film (approximately 50 nm) on the sensor chip [25] [2]. At a specific angle of incidence—the resonance angle—the energy of the incident light couples with the surface plasmons, creating an evanescent field that extends approximately 100-200 nm from the metal surface [2] [42]. This phenomenon manifests as a sharp dip in the intensity of reflected light at the resonance angle [25].
When analytes interact with bioreceptors immobilized on the chip surface, the local refractive index changes, altering the resonance condition [25] [2]. This shift can be measured in real-time as a change in the resonance angle (angular shift), resonance wavelength (wavelength shift), or intensity at a fixed angle [25]. The resulting sensorgram provides detailed information about binding kinetics, including association and dissociation rates, as well as affinity constants [25]. The exceptional sensitivity of SPR biosensors allows them to detect RI changes as small as 10⁻⁶ to 10⁻⁷ RIU, enabling the monitoring of biomolecular interactions without labels that could potentially alter binding affinities or native conformational dynamics [2].
SPR Experimental Setup and Sensorgram Interpretation
A standard SPR biosensor comprises several essential components: a light source, a biochip (typically a gold film coated with bioreceptor molecules), a glass prism, a detector, a flow cell, and a flow medium containing the target analytes [25]. The biosensing process involves four primary phases in the sensorgram: association phase (when analytes bind to receptors), equilibrium or steady-state phase, dissociation phase (when buffer flows over the surface and complexes dissociate), and regeneration phase (when the surface is prepared for a new cycle) [25]. Key parameters extracted from sensorgrams include Kon (binding affinity), Kd (rate of dissociation), and Koff (rate of association), which collectively provide a comprehensive understanding of molecular interactions [25].
SPR Biosensing Workflow and Output
Advanced SPR Sensing Platforms and Materials
Chip Designs and Functional Matrices
The biosensor chip is often regarded as the heart of the SPR instrument, requiring meticulous design to immobilize an adequate density of bio-recognition molecules while concurrently minimizing non-specific interactions [25]. Various chip designs have been developed to optimize performance, including self-assembled monolayers (SAMs), carboxymethylated dextran (CMD), and nitrilotriacetic acid (NTA)-functionalized platforms [25]. Recent advances have revealed that emerging techniques, including multifunctional biochips and zwitterionic coatings, outperform traditional CMD-based systems in complex biological matrices [25].
Critical to biosensor design is the effective immobilization of bioreceptors onto SPR sensor chips [25]. Two prevalent strategies for achieving stable attachment involve covalent coupling and affinity tag immobilization [25]. Covalent coupling typically utilizes carboxyl or thiol chemistry, allowing for robust chemical bonds between the bioreceptor and the sensor surface [25]. In contrast, affinity tag immobilization leverages specific interactions, such as biotin-streptavidin, for oriented immobilization that often preserves bioactivity [25]. The selection of immobilization chemistry significantly impacts the sensitivity, specificity, and reusability of SPR biosensors [25].
Nanomaterial-Enhanced SPR Platforms
The integration of various materials with plasmonic metals has become a significant area of investigation in SPR-based optical sensors [44]. The use of materials such as oxides like TiO₂, Transition Metal Dichalcogenides (TMDCs), and other nanostructures like carbon nanotubes (CNTs), graphene, and MXene has been explored to either enhance sensitivity or improve the chemical stability of the plasmonic layer [44]. For instance, a D-shaped photonic crystal fiber (PCF) SPR biosensor with optimized gold-TiO₂ layers demonstrated exceptional performance for multi-cancer detection [44] [45]. The TiO₂ layer was applied on top of the gold layer to enhance sensitivity, achieving a maximum wavelength sensitivity of 42,000 nm/RIU and a maximum figure of merit (FOM) of 1393.128 RIU⁻¹ [44].
Similarly, researchers have developed metasurface sensors that combine multiple two-dimensional materials for enhanced performance [46]. One design featured four identical figure-eight-shaped resonant elements coated with MXene, rectangular structures utilizing black phosphorus, and a central square-shaped element incorporating a single-layer graphene coating [46]. This multi-resonator metamaterial configuration was engineered to maximize electromagnetic field confinement and optimize wave-matter interactions, demonstrating competitive sensitivity (395 GHz/RIU) for protein biomarker detection [46].
Table 1: Advanced SPR Sensing Platforms and Their Performance Characteristics
| Platform Type | Sensing Materials | Detection Range | Sensitivity | Application | Reference |
|---|---|---|---|---|---|
| D-shaped PCF SPR | Au-TiO₂ | RI: 1.3-1.4 | 42,000 nm/RIU | Multi-cancer detection | [44] |
| LSPR | Au-Ag NPs in TiO₂ matrix | 0.05-2 ng mL⁻¹ | LOD: 7 pg mL⁻¹ | Ochratoxin-A in wine | [33] |
| Metasurface THz | MXene-BP-Graphene | 0.31-0.46 THz | 395 GHz/RIU | Protein biomarkers | [46] |
| Peptide-based MP-SPR | Short synthetic peptides | 5 pM to 9 nM | LOD: 0.34 pM | MMP-9 protease | [47] |
| Dual-polished PCF | Not specified | Not specified | 7,143 nm/RIU | Cancer detection | [44] |
SPR for Cancer Cell Detection
Direct Cancer Cell Detection via Refractive Index Monitoring
SPR biosensors have demonstrated remarkable capabilities in the direct detection of cancer cells through refractive index monitoring. In one advanced approach, researchers developed a D-shaped photonic crystal fiber (PCF) SPR biosensor with layers of gold and titanium oxide (TiO₂) for precise multi-cancer detection [44]. This biosensor was tested on multiple cancer cell samples, including Basal, MDA-MB-231, Jurkat, PC-12, and HeLa cells, achieving a maximum wavelength sensitivity of 42,000 nm/RIU, a maximum amplitude sensitivity of -1862.72 RIU, and a maximum figure of merit of 1393.128 RIU⁻¹ [44] [45]. The biosensor operated within a refractive index range of 1.3-1.4, corresponding to the typical cytoplasmic refractive index of cancer cells, and across the wavelength range from visible to near-infrared [44].
The detection mechanism relied on measuring changes in the refractive index of cell cytoplasm, which links detection to protein concentration changes and simplifies experimental procedures [44]. The synergistic combination of optimized Gold/TiO₂ layers proved critical for achieving the sensor's exceptional sensitivity [44]. The D-shaped design addressed practical fabrication challenges by polishing the surface of the PCF to create a flat area, enabling a homogeneous metal coating with minimal roughness [44]. This polished fiber surface positioned the metal layer closer to the fiber core, providing an effective solution to the coupling issue between the core mode and the surface plasmon polariton mode [44].
Extracellular Vesicle-Based Cancer Diagnosis
Extracellular vesicles (EVs) have emerged as valuable biomarkers for cancer diagnosis due to their specificity and accessibility in bodily fluids [42]. EVs are nanoscale particles (ranging from 30 to 150 nm) secreted by cells that carry biomolecules such as proteins and nucleic acids reflecting the physiological state of their cells of origin [42]. SPR-based biosensors offer a promising platform for EV detection, enabling real-time, label-free monitoring of these cancer-derived biomarkers [42].
Recent advances in SPR technology have improved the sensitivity of EV detection. Traditional gold-film-based SPR sensors lacked the sensitivity required to detect extremely low EV concentrations, critical for identifying early-stage tumors [42]. However, newer platforms, such as localized surface plasmon resonance and surface plasmon resonance imaging, have enhanced sensitivity and throughput, making it possible to detect single EVs and perform multiplexed measurements [42]. The development of nanostructured SPR substrates has further improved EV detection capabilities by creating electromagnetic hot spots that significantly enhance the local electric field and increase sensitivity [42].
Table 2: SPR-Based Cancer Detection Modalities and Their Performance
| Detection Modality | Cancer Type | Biomarker/Cell Line | Sensitivity | Specificity | Reference |
|---|---|---|---|---|---|
| Cytoplasmic RI detection | Multiple | Basal, HeLa, Jurkat, PC-12, MDA-MB-231 | 42,000 nm/RIU | Not specified | [44] |
| EV detection | Glioblastoma | CD44, CD133 | Not specified | Not specified | [42] |
| EV detection | Breast cancer | HER2 | Not specified | Not specified | [42] |
| EV detection | Ovarian cancer | CA-125 | Not specified | Not specified | [42] |
| EV detection | Prostate cancer | PSMA | Not specified | Not specified | [42] |
| Protease activity | Multiple | MMP-9 | 0.34 pM (LOD) | Not specified | [47] |
Biomarker Analysis Using SPR Biosensors
Protease Biomarker Tracking
SPR biosensors have been successfully employed for real-time tracking of protease biomarkers, which play central roles in cancer progression, neurodegeneration, inflammation, and wound pathology [47]. A research team developed the first short synthetic peptide-based biosensor for real-time tracking of the disease-related protease matrix metalloproteinase-9 (MMP-9) using multi-parametric surface plasmon resonance spectroscopy (MP-SPR) [47]. Unlike traditional methods like ELISA, which measure concentration rather than activity, this biosensor used short, synthetic peptides specifically engineered as MMP-9 substrates immobilized on the MP-SPR sensor surface [47].
When exposed to active MMP-9, these peptides undergo cleavage between Gly and Met residues, producing an immediate, measurable decrease in the SPR signal that allows researchers to observe real-time proteolysis [47]. The MP-SPR platform captured multiple optical parameters simultaneously, such as refractive index, surface coverage, and layer thickness, enabling modeling of SPR curves with much higher precision than conventional SPR methods [47]. The biosensor demonstrated remarkable analytical performance with a limit of detection of 0.34 pM in buffer and 0.56 pM in cell culture medium, with a dynamic range from 5 pM to 9 nM, spanning the clinically relevant concentration range of MMP-9 [47].
Food Safety and Environmental Monitoring Applications
Beyond medical diagnostics, SPR biosensors have found significant applications in food safety and environmental monitoring. A novel label-free LSPR-based biosensor was specifically designed for Ochratoxin-A detection, employing a portable LSPR spectroscopy sensing system for efficient on-site and cost-effective analysis [33]. This biosensor comprised monoclonal anti-OTA antibodies immobilized on the surface of sputtered Au-Ag nanoparticles embedded in a TiO₂ matrix [33].
Under optimized conditions, the LSPR-based biosensor demonstrated a linear dynamic response from 0.05 to 2 ng mL⁻¹, with an estimated limit of detection at 7 pg mL⁻¹, using only 55 μL of sample [33]. This performance outperformed commercial ELISA techniques in relevant bioanalytical parameters [33]. The robustness and feasibility of the presented LSPR-based biosensing was tested using spiked white wine, exhibiting a satisfactory recovery of 93%–113%, confirming its efficacy in a complex matrix [33]. This application highlights the versatility of SPR biosensors in detecting small molecules in challenging environments.
Experimental Protocols and Methodologies
Sensor Functionalization and Bioreceptor Immobilization
The functionalization of SPR sensor surfaces requires precise experimental protocols to ensure optimal performance. For antibody-based detection, a common approach involves creating self-assembled monolayers (SAMs) using alkanethiols on gold surfaces, followed by covalent immobilization of antibodies via carboxyl or amine groups [25]. In the case of the LSPR biosensor for Ochratoxin-A detection, monoclonal anti-OTA antibodies were immobilized on the surface of sputtered Au-Ag nanoparticles embedded in a TiO₂ matrix [33]. The protocol involved activating the surface with appropriate cross-linkers, incubating with antibody solutions at optimized concentrations, and blocking non-specific sites with inert proteins like bovine serum albumin [33].
For peptide-based biosensors, such as the MMP-9 activity sensor, short synthetic peptides were engineered as protease substrates and immobilized on the SPR sensor surface [47]. The immobilization protocol ensured proper orientation and accessibility of the cleavage site while minimizing non-specific binding [47]. The peptide functionalization allowed for real-time monitoring of enzymatic activity rather than mere concentration, providing dynamic information about protease function in physiological conditions [47].
Sample Preparation and Analysis Procedures
Proper sample preparation is critical for reliable SPR biosensing, particularly in complex matrices. For cancer cell detection using the D-shaped PCF SPR biosensor, the experimental setup involved generating electromagnetic waves with a tunable optical laser source transmitted through a single-mode fiber to a polarizer and then to the biosensor [44]. The analyte was introduced to and removed from the biosensor via a programmable pump, controlled through tubes and valves at appropriate pressure [44]. The output light from the biosensor was sent through a single-mode fiber to an optical spectrum analyzer, with collected data transmitted to a computer for analysis [44].
For EV detection from biological samples, protocols typically involve isolation of EVs from bodily fluids like blood or urine using ultracentrifugation or precipitation methods, followed by characterization of EV size and concentration before SPR analysis [42]. The EV samples are then introduced into the SPR system in appropriate running buffers, often with additives to minimize non-specific binding [42]. Real-time binding responses are recorded, and data processing algorithms are applied to extract quantitative information about EV concentration and biomarker expression levels [42].
SPR Experimental Protocol Workflow
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of SPR biosensing requires careful selection of research reagents and materials. The following table summarizes key components essential for developing and utilizing SPR biosensors in diagnostic applications.
Table 3: Essential Research Reagents and Materials for SPR Biosensing
| Category | Specific Examples | Function/Purpose | Application Notes |
|---|---|---|---|
| Plasmonic Materials | Gold, Silver, Au-Ag alloys | Generate surface plasmons | Gold offers chemical stability; silver provides higher sensitivity but requires protection [44] |
| Enhancing Materials | TiO₂, Graphene, MXene, Black Phosphorus | Enhance sensitivity and stability | TiO₂ layers on gold improve sensitivity; 2D materials enable field enhancement [44] [46] |
| Immobilization Chemistry | Carboxyl, Thiol, Biotin-Streptavidin | Covalent and affinity immobilization | Thiol chemistry for gold surfaces; biotin-streptavidin for oriented immobilization [25] |
| Bioreceptors | Antibodies, Peptides, Aptamers | Molecular recognition elements | Antibodies for specificity; peptides for enzyme activity; aptamers for stability [47] [25] |
| Surface Matrices | CMD, SAMs, NTA | Sensor chip functionalization | CMD provides hydrogel matrix; SAMs offer controlled density; NTA allows His-tag immobilization [25] |
| Blocking Agents | BSA, Casein, Zwitterionic polymers | Reduce non-specific binding | Essential for complex samples; zwitterionic coatings show superior antifouling [25] |
| Regeneration Solutions | Glycine, NaOH, SDS | Remove bound analyte without damaging receptors | Must be optimized for each molecular interaction to maintain sensor surface integrity [25] |
Future Perspectives and Integration with Artificial Intelligence
The future of SPR technology in biomedical analysis is poised for significant innovation, driven by the demand for higher sensitivity, real-time monitoring, and point-of-care diagnostics [25]. One major trend is the integration of SPR with artificial intelligence and machine learning for real-time data interpretation and predictive modeling of biomolecular interactions [25] [42]. Miniaturized SPR chips and lab-on-a-chip systems are expected to support in situ analysis of pharmacokinetics directly in biological fluids, enabling personalized medicine approaches [25].
The integration of machine learning and artificial intelligence technologies with SPR holds great promise for advancing data analysis, improving detection accuracy, and enhancing result reliability [42]. AI can play a pivotal role in the design and optimization of SPR-based biosensors, further expanding their clinical utility [42]. Specific approaches include using artificial neural networks (ANNs), deep neural networks (DNNs), convolutional neural networks (CNNs), and support vector machines (SVMs) to process complex SPR data, distinguish specific binding signals from noise in complex samples, and classify cancer subtypes based on EV signature profiles [42].
Future developments will likely focus on improving sensitivity to detect extremely low biomarker concentrations, enhancing specificity to minimize non-specific binding, and managing the complex data generated during detection [42]. Additionally, the combination of SPR with complementary techniques such as electrochemistry, mass spectrometry, and Raman spectroscopy is expected to provide multidimensional analytical information, further advancing the capabilities of SPR in biomedical diagnostics [25]. As these technologies mature, SPR biosensors will become increasingly accessible for clinical applications, potentially enabling routine early cancer detection and personalized treatment monitoring.
High-Throughput SPR Imaging (SPRI) for Multiplexed Biomolecular Interaction Analysis
Surface Plasmon Resonance Imaging (SPRi) represents a significant evolution in label-free detection technologies, enabling the real-time, parallel analysis of hundreds to thousands of biomolecular interactions. Within the broader context of label-free detection research, SPRI addresses a critical need for high-throughput analytical methods that preserve biomolecular native state and function by eliminating the requirement for fluorescent or radioactive labels that can sterically hinder interactions or alter binding kinetics [28] [2]. This technology has established itself as a powerful tool for researchers and drug development professionals who require quantitative data on binding specificity, affinity, and kinetics across multiple interactions simultaneously [48] [49].
The fundamental principle unifying label-free detection techniques is their reliance on inherent molecular properties—such as refractive index, mass, or charge—rather than engineered signal probes [28]. SPRI specifically exploits the sensitivity of surface plasmon resonance to local refractive index changes at a metal-dielectric interface, translating molecular binding events into quantifiable signals without the need for molecular tagging [50]. This capability is particularly valuable in pharmaceutical research and development, where the market for label-free detection technologies is projected to grow from $2.14 billion in 2024 to $4.48 billion by 2032, driven largely by applications in drug discovery and biomolecular interaction analysis [38].
Theoretical Foundations of SPRI
Basic SPR Phenomenon
Surface Plasmon Resonance (SPR) occurs when polarized light strikes a metal film (typically gold) under total internal reflection conditions, causing the collective oscillation of electrons at the metal-dielectric interface [50]. These resonant oscillations, known as surface plasmons, create an evanescent field that extends approximately 200-300 nanometers into the medium adjacent to the metal surface. The resonance condition is exquisitely sensitive to changes in the local refractive index within this evanescent field [2]. When biomolecules bind to the sensor surface, they displace buffer solution and increase the local refractive index, leading to a measurable shift in the SPR angle that is proportional to the mass concentration of bound analyte [50].
From SPR to SPRI: The Imaging Advantage
While conventional SPR monitors a single sensing spot, SPRI extends this capability to spatially resolve interactions across an array format through the use of charge-coupled device (CCD) or complementary metal-oxide-semiconductor (CMOS) detectors [48] [50]. This imaging approach maintains the kinetic and thermodynamic measurement capabilities of traditional SPR while enabling massively parallel data acquisition from hundreds to thousands of distinct interaction spots simultaneously [48]. The key distinction lies in the detection methodology: rather than tracking the angular position of the resonance minimum over time at a single point, SPRI systems typically employ fixed-angle illumination and measure intensity changes across the entire sensor surface, converting these spatial variations into binding data for each array element [51] [50].
Technical Implementation of SPRI
Instrumentation Components
A typical SPRI system integrates several key components: a light source (often a light-emitting diode or laser), a polarizer to ensure incident light has the correct polarization for SPR excitation, a high-index prism for coupling the light to the sensor chip, a microfluidic cartridge for sample delivery, and a high-resolution camera for capturing the SPR response across the sensor surface [51] [48]. Advanced systems may incorporate liquid crystal tunable filters (LCTF) for spectral scanning capabilities, enabling improved dynamic range and resolution [51]. Modern SPRI instruments can complete an SPR dip measurement within 4 seconds with a refractive index resolution of approximately 5.87×10⁻⁶ RIU, making them suitable for capturing rapid binding events [51].
Sensor Chip Functionalization
The foundation of successful SPRI experiments lies in the proper functionalization of the sensor surface. Gold-coated glass substrates are typically used due to gold's favorable SPR properties and well-established surface chemistry [48] [50]. These surfaces are often modified with self-assembled monolayers (SAMs) of alkanethiols containing terminal functional groups (e.g., carboxyl, amine, or hydroxyl) that facilitate the covalent immobilization of ligands [49]. Pre-activated functionalized hydrogel sensor chips (such as HCX series from XanTec) provide a three-dimensional matrix that increases loading capacity and can enhance sensitivity for lower molecular weight analytes [48].
Table: Key Technical Specifications of SPRI Systems
| Parameter | Typical Range | Significance |
|---|---|---|
| Refractive Index Resolution | 5.87×10⁻⁶ RIU [51] | Determines detection sensitivity for small molecules |
| Dynamic Detection Range | 4.63×10⁻² RIU [51] | Maximum measurable response range |
| Spot Measurement Time | ~4 seconds [51] | Impacts temporal resolution for kinetic studies |
| Array Density | Up to 500 spots [48] | Throughput capacity for multiplexed analysis |
| Molecular Weight Detection Limit | Single-protein detection (tens of kDa) [2] | Smallest detectable biomolecule |
Experimental Design and Protocol
Microarray Fabrication
The creation of ligand microarrays is a critical step in SPRI experimentation. Using non-contact piezoelectric printers (e.g., TopSpot system from BioFluidix), researchers can spot multiple ligands at different concentrations in a predefined array pattern [48]. A typical protocol involves:
Surface Preparation: Clean gold sensor chips are functionalized with appropriate capture surfaces, which may include carboxylated alkanethiol SAMs or specialized hydrogel layers [48] [49].
Ligand Printing: Ligands (antibodies, proteins, DNA, etc.) are prepared in spotting buffer at varying concentrations (e.g., 100-800 μg/mL for proteins) and deposited in a serial dilution pattern across the sensor surface [48]. The array typically includes control spots for background subtraction and normalization.
Immobilization: After printing, the sensor chip is incubated in a humidity chamber for 1 hour at room temperature to facilitate covalent attachment [48].
Quenching: Remaining active groups on the sensor surface are deactivated with ethanolamine (1 M, pH 8) for 10 minutes to prevent non-specific binding in subsequent steps [48].
Washing: The surface is intensively washed with running buffer to remove unbound ligand and prepare for analysis [48].
Table: Essential Research Reagents for SPRI
| Reagent/Chemical | Function | Application Notes |
|---|---|---|
| Gold-coated Sensor Chips | SPR-active substrate | Typically 50nm gold on glass with adhesion layer |
| Functionalized Hydrogel Chips (e.g., HCX 80m) | 3D matrix for ligand immobilization | Increases binding capacity; suitable for small molecule detection |
| Alkanethiol SAM Kits | Surface functionalization | Provide carboxyl, amine, or hydroxyl groups for conjugation |
| N-Hydroxysuccinimide (NHS)/1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) | Crosslinking chemistry | Activates carboxyl groups for amine coupling |
| Ethanolamine | Quenching reagent | Blocks unused activated esters after immobilization |
| HBS-EP Running Buffer | Sample delivery and baseline | Provides consistent pH and ionic strength; reduces non-specific binding |
| Regeneration Solutions (e.g., Glycine-HCl, NaOH) | Surface regeneration | Removes bound analyte while preserving ligand activity |
Binding Kinetics Measurement
The core SPRI experiment follows a systematic workflow to extract kinetic parameters:
SPRI Experimental Workflow
Baseline Stabilization: The functionalized sensor chip is mounted in the SPRI instrument, and running buffer is flowed across the surface until a stable baseline is established [48] [50].
Association Phase: Sample containing the analyte(s) of interest is injected and flowed over the sensor surface. Binding events are recorded in real-time as increases in SPR response [48] [49]. The duration of injection is optimized to approach binding equilibrium without consuming excessive sample.
Dissociation Phase: Running buffer is reintroduced, and the decrease in SPR response is monitored as analytes dissociate from their immobilized ligands [48].
Surface Regeneration: A regeneration solution (e.g., low pH buffer or detergent solution) is briefly injected to remove all bound analyte without damaging the immobilized ligand [48] [49]. The surface is then re-equilibrated with running buffer for the next analysis cycle.
For concentration analysis, the process is repeated with a series of analyte concentrations, typically spanning two orders of magnitude to adequately define the binding isotherm [48].
Data Analysis and Kinetic Parameter Extraction
SPRI data analysis transforms the raw sensorgram (SPR response versus time) into quantitative kinetic and affinity parameters. For a 1:1 binding model, the interaction is described by:
[ A + B \underset{kd}{\overset{ka}{\rightleftharpoons}} AB ]
Where (ka) is the association rate constant, (kd) is the dissociation rate constant, and the equilibrium dissociation constant (KD = kd/k_a) [48].
The binding response during the association phase follows:
[ \frac{dRt}{dt} = ka \cdot [A] \cdot (R{max} - Rt) - kd \cdot Rt ]
Where (Rt) is the response at time (t), (R{max}) is the maximum binding capacity, and ([A]) is the analyte concentration [48].
During the dissociation phase, the decay follows:
[ Rt = R0 \cdot e^{-k_d \cdot t} ]
Where (R_0) is the response at the start of dissociation [48].
Modern SPRI analysis software globally fits these equations to the entire dataset, simultaneously optimizing parameters across multiple analyte concentrations and ligand densities to yield robust kinetic constants [48] [49].
Applications in Biomolecular Research
High-Throughput Biomolecular Interaction Screening
SPRI's multiplexing capability makes it ideally suited for screening applications where numerous interactions must be characterized rapidly. In one demonstrated approach, researchers simultaneously analyzed three different antigen-antibody pairs by creating a 6×4 microarray with serial dilutions of human IgG, bovine IgG, and BSA, obtaining kinetic parameters for all interactions from a single analyte injection containing a mixture of the corresponding antibodies [48]. This methodology drastically reduces measurement time and reagent consumption compared to sequential analysis in conventional SPR [48].
Drug Discovery and Development
In pharmaceutical research, SPRI accelerates lead compound identification and optimization by enabling high-throughput kinetic screening of fragment libraries and small molecule candidates against therapeutic targets [37] [38]. The technology provides critical structure-activity relationship (SAR) data through precise measurement of binding kinetics and affinities, informing medicinal chemistry efforts [37]. With the U.S. Food and Drug Administration now accepting label-free kinetics in investigational new drug dossiers, SPRI data has gained regulatory relevance, further driving its adoption in pharmaceutical pipelines [37].
Diagnostic Biomarker Validation
SPRI facilitates the identification and validation of protein biomarkers from complex biological samples such as serum, cell lysates, and crude extracts [50] [49]. By immobilizing multiple capture molecules (antibodies, aptamers, etc.) in array format, researchers can screen for the presence and concentration of numerous potential biomarkers simultaneously [49]. For example, SPRI has been used to detect activated leukocyte cell adhesion molecule (ALCAM), a protein biomarker associated with various cancers, directly from human crude cell lysates, demonstrating the technique's capability to detect clinically relevant biomarkers in minimally processed samples [49].
Comparative Analysis with Other Label-Free Technologies
Table: Comparison of Label-Free Detection Technologies
| Technology | Detection Principle | Throughput | Kinetic Capability | Key Applications |
|---|---|---|---|---|
| SPRI | Refractive index change at metal surface [50] | High (hundreds to thousands of spots) [48] | Full real-time kinetics [48] [49] | Biomolecular interaction screening, diagnostic development [50] [49] |
| Bio-Layer Interferometry (BLI) | Interferometric pattern shift at biosensor tip [37] [52] | Medium (up to 96 samples) | Real-time kinetics [37] | Antibody characterization, protein-protein interactions [37] |
| Isothermal Titration Calorimetry (ITC) | Heat change upon binding [28] [52] | Low (single sample) | Thermodynamic parameters only | Binding affinity and stoichiometry [52] |
| Quartz Crystal Microbalance (QCM) | Mass-induced frequency change [28] | Medium (multiple flow cells) | Real-time kinetics | Protein adsorption, cell adhesion [28] |
Current Challenges and Future Perspectives
Despite its significant advantages, SPRI faces several technical challenges. The requirement for one interaction partner to be immobilized on the sensor surface can potentially affect its native conformation and binding activity [2]. Sensitivity limitations persist for small molecule detection, although advances in signal amplification and nanomaterials are gradually addressing this constraint [37] [2]. Additionally, the sophisticated instrumentation and operational expertise required present barriers to widespread adoption, particularly in resource-limited settings [37].
Future developments in SPRI technology are focusing on several key areas. Integration with artificial intelligence and machine learning algorithms is enhancing data analysis capabilities, reducing noise, and improving binding affinity predictions [37] [38]. Miniaturization and portability efforts are yielding compact SPRI systems suitable for point-of-care applications [37] [38]. The combination of SPRI with mass spectrometry for off-line identification of bound species from complex mixtures represents another promising direction, creating a powerful tool for biomarker discovery and systems biology research [49].
Future Development Directions of SPRI Technology
As label-free detection technologies continue to evolve, SPRI is positioned to play an increasingly central role in biomolecular interaction analysis, particularly as the market expands at a projected CAGR of 8.9-9.8% through 2032 [38] [52]. The technology's unique combination of multiplexing capability, real-time kinetic resolution, and label-free operation makes it an indispensable tool for researchers and drug development professionals seeking to understand complex biological systems at the molecular level.
Enhancing SPR Performance: Optimization, Troubleshooting, and Advanced Sensing Strategies
Surface Plasmon Resonance (SPR) is a powerful label-free optical biosensing technology that enables real-time monitoring of biomolecular interactions [6] [16]. The technique operates on the principle of total internal reflection, where polarized light strikes a sensor surface coated with a thin metal film (typically gold), generating electron charge density waves known as surface plasmons [16]. This energy transfer reduces the intensity of the reflected light at a specific resonance angle, which is exquisitely sensitive to changes in the refractive index at the sensor surface [6] [16]. When biomolecular binding events occur near this surface, the resulting refractive index shift alters the resonance angle, providing a direct measure of interaction kinetics and affinity without requiring fluorescent or radioactive labels [6].
The label-free nature of SPR technology has positioned it as a cornerstone in life sciences research and drug development, particularly for characterizing binding kinetics, affinity constants, and biomolecular concentration in diverse applications ranging from antibody characterization to fragment-based drug discovery [6] [16]. Within this context, achieving ultimate sensitivity has emerged as a critical frontier, enabling detection of low-concentration analytes down to the single-molecule level—a capability with profound implications for early disease diagnosis and fundamental biological research [53] [54].
The Need for Advanced Optimization in SPR Design
Traditional SPR optimization approaches have faced significant limitations in achieving the simultaneous enhancement of multiple performance parameters. Conventional single-variable scanning methods focus on improving one parameter at a time (typically sensitivity) while neglecting complex interrelationships between design variables and their collective impact on overall sensor performance [53]. This fragmented approach often results in suboptimal configurations where gains in one metric (e.g., sensitivity) come at the expense of others (e.g., signal clarity or detection limit) [53].
The detection of low-concentration analytes presents particular challenges for conventional SPR sensors, which traditionally struggle to identify molecules at concentrations lower than 1 × 10⁻¹⁵ g/mL, effectively preventing single-molecule detection capabilities [53]. Furthermore, structural optimizations involving specialized nano-fabrication processes or the incorporation of 2D sensitive materials like graphene and molybdenum disulfide often introduce complications related to stability, manufacturability, and cost [53]. These limitations have created an pressing need for comprehensive optimization strategies that can concurrently address multiple performance metrics while balancing practical implementation constraints.
Multi-Objective Optimization Frameworks
Core Optimization Algorithms
Particle Swarm Optimization (PSO) has emerged as a particularly effective algorithm for SPR sensor design due to its ability to rapidly identify optimal solutions within complex, multidimensional search spaces [53]. In a recent demonstration of its capabilities, PSO was employed to simultaneously optimize three critical SPR design parameters: incident angle, adhesive layer thickness (chromium), and metal layer thickness (gold) [53] [54]. This multi-objective approach specifically targeted three key performance metrics: sensitivity (S), figure of merit (FOM), and figure of merit with depth (DFOM), achieving remarkable improvements of 230.22%, 110.94%, and 90.85% respectively compared to conventional designs [53] [54].
The PSO process operates through an iterative refinement mechanism where a population of candidate solutions (particles) navigates the parameter space, continuously adjusting their positions based on both individual experience and collective intelligence [53]. After approximately 150 iterations, this approach typically converges on optimal parameter combinations that would be exceedingly difficult to identify through manual design processes [53]. To mitigate potential processing errors, researchers have successfully integrated k-means clustering methods to identify robust design parameters from the optimized solution set, enhancing manufacturability without compromising performance [53] [54].
Machine Learning and Explainable AI Integration
Beyond traditional optimization algorithms, Machine Learning (ML) regression techniques have demonstrated remarkable capabilities in predicting key optical properties of SPR sensors, significantly accelerating the design process while reducing computational costs [55] [56]. Researchers have successfully implemented multiple ML models—including Random Forest regression, Decision Trees, Gradient Boosting, and Extreme Gradient Boosting—to forecast critical sensor characteristics such as effective refractive index, confinement loss, and amplitude sensitivity with high predictive accuracy [55].
A groundbreaking advancement in this domain involves the integration of Explainable AI (XAI) methods, particularly SHapley Additive exPlanations (SHAP), to interpret model outputs and identify the most influential design parameters [55] [56]. SHAP analysis has revealed that wavelength, analyte refractive index, gold thickness, and pitch represent the most critical factors influencing PCF-SPR sensor performance [55] [56]. This interpretable approach not only accelerates optimization but also provides valuable physical insights into the fundamental relationships between design parameters and sensor performance, bridging the gap between data-driven predictions and theoretical understanding.
Performance Metrics and Evaluation
Comprehensive SPR optimization requires careful balancing of multiple performance metrics, each capturing different aspects of sensor functionality. The most critically evaluated parameters include:
- Sensitivity (S): Quantifies the shift in resonance parameters (wavelength or angle) per unit change in refractive index, directly determining the sensor's ability to detect small molecular interactions [53] [55].
- Figure of Merit (FOM): A composite metric that incorporates both sensitivity and resonance peak width, providing a more comprehensive assessment of sensor capability [53].
- Depth of Resonant Dip (DRD): Influences signal detection clarity and measurement precision, particularly important for low-concentration analytes [53].
- Detection Limit (LOD): The lowest analyte concentration that can be reliably detected, representing the ultimate measure of sensor performance for trace analysis [53] [54].
- Confinement Loss (CL): Particularly relevant for photonic crystal fiber SPR sensors, representing optical power attenuation and ultimately determining signal-to-noise ratios [55] [56].
Table 1: Algorithm-Assisted SPR Sensor Performance Comparison
| Sensor Type | Optimization Method | Max Sensitivity (nm/RIU) | Figure of Merit (FOM) | Detection Limit | Key Applications |
|---|---|---|---|---|---|
| Kretschmann SPR [53] | Multi-objective PSO | 24,482.86 | Not specified | 54 ag/mL (0.36 aM) | Mouse IgG detection |
| PCF-SPR [55] | ML + SHAP Analysis | 125,000 | 2,112.15 | 8×10⁻⁷ RIU resolution | Cancer cell detection |
| Conventional SPR [53] | Single-variable scanning | ~7,400 (baseline) | ~15 (baseline) | >1×10⁻¹⁵ g/mL | General biomolecular interaction |
Experimental Protocols and Methodologies
Sensor Fabrication and Design Parameters
The implementation of algorithm-optimized SPR sensors begins with careful fabrication based on the identified optimal parameters. For conventional Kretschmann-configuration SPR sensors, the multi-objective PSO approach typically generates specific recommendations for three primary design variables: incident angle (precisely controlled through optical alignment), adhesive layer thickness (chromium, typically optimized at nanoscale dimensions), and metal film thickness (gold, optimized to enhance plasmon excitation and propagation) [53] [54]. The resulting sensor structure employs a prism coupling mechanism with sequential metal layers deposited using precise physical vapor deposition techniques to ensure uniformity and consistency [53].
For Photonic Crystal Fiber SPR (PCF-SPR) sensors, the optimization parameters expand to include air hole geometry, pitch distance (spacing between air holes), gold layer thickness applied to selective channels, and perfectly matched layer specifications to minimize boundary reflections [55] [56]. Fabrication typically involves stack-and-draw methods for the PCF structure followed by precise metal deposition using techniques such as sputtering or chemical plating to achieve the algorithm-recommended metal thickness with nanometer precision [55] [56].
Measurement Protocols and Data Acquisition
Robust experimental protocols are essential for validating algorithm-optimized SPR sensors. For bulk refractive index characterization, a standardized approach involves flowing solutions with known refractive indices (typically sucrose or glycerol solutions at varying concentrations) across the sensor surface while monitoring the resonance shift [53] [55]. For biomolecular interaction studies, the protocol follows a sequential process:
- Surface Functionalization: Immobilization of capture ligands (e.g., antibodies, receptors) on the sensor surface using covalent coupling chemistry or capture systems [53] [16].
- Baseline Stabilization: Flowing buffer solution to establish a stable reference signal and condition the sensor surface [16].
- Association Phase: Introducing the analyte solution at varying concentrations while monitoring binding in real-time to determine association rates (kₑₙ) [16].
- Dissociation Phase: Replacing analyte solution with buffer to monitor complex dissociation and determine dissociation rates (kₒₑ₇) [16].
- Regeneration: Applying a regeneration solution (e.g., low pH or high salt buffer) to remove bound analyte and prepare the surface for subsequent analysis cycles [16].
Throughout these measurements, data acquisition occurs continuously with high temporal resolution, typically generating sensorgrams that plot response units against time [16]. For PCF-SPR sensors, additional measurements include confinement loss spectra obtained using optical spectrum analyzers and amplitude sensitivity calculations based on transmission intensity variations [55].
Data Processing and Kinetic Analysis
The raw data from SPR experiments undergoes sophisticated processing to extract meaningful kinetic and affinity parameters. For binding interactions, the sensorgram data is fitted to appropriate interaction models (e.g., 1:1 Langmuir binding, bivalent analyte, or heterogeneous ligand models) using specialized software algorithms [6] [16]. The quality of fitting is assessed through chi-squared (χ²) values and residual analysis to ensure model appropriateness [16].
For algorithm-optimized sensors targeting ultimate sensitivity, additional signal processing techniques are often employed, including digital filtering to reduce high-frequency noise, baseline drift correction, and reference surface subtraction to eliminate bulk refractive index effects and non-specific binding contributions [53] [55]. In ML-enhanced approaches, the processed data further serves as training and validation sets for refining predictive models, creating a continuous improvement cycle where experimental results inform algorithmic refinements [55] [56].
Research Reagent Solutions
Successful implementation of algorithm-optimized SPR sensors requires carefully selected reagents and materials tailored to specific experimental needs. The table below outlines essential components and their functions in typical SPR experiments.
Table 2: Essential Research Reagents and Materials for SPR Biosensing
| Reagent/Material | Function | Specification Considerations | Application Context |
|---|---|---|---|
| Sensor Chips [53] [16] | Platform for ligand immobilization and plasmon generation | Gold film thickness (45-55 nm), chromium adhesion layer (1-2 nm), dextran matrix for covalent coupling | Kretschmann configuration SPR |
| Coupling Reagents [16] | Covalent immobilization of ligands | EDC/NHS chemistry for amine coupling, surface activation protocols | Protein, antibody immobilization |
| Running Buffers [16] | Maintain consistent biochemical conditions | HEPES-buffered saline (HBS), pH 7.4, optional surfactants (e.g., Tween 20) | All SPR binding studies |
| Regeneration Solutions [16] | Remove bound analyte without damaging immobilized ligand | Glycine-HCl (pH 2.0-3.0), high salt solutions (1-2 M NaCl) | Surface reuse for multiple cycles |
| Reference Proteins [53] | System validation and performance verification | Mouse IgG, BSA, well-characterized binding pairs | Sensor calibration and QC |
| 2D Nanomaterials [53] | Sensitivity enhancement layers | Graphene, molybdenum disulfide, tungsten disulfide | Signal-amplified SPR detection |
Technological Workflows and Signaling Pathways
The algorithm-assisted optimization process follows a systematic workflow that integrates computational design with experimental validation. The diagram below illustrates the key stages in this integrated approach.
SPR Algorithm Optimization Workflow
The signaling pathway in SPR detection relies on the precise coupling of light energy to surface plasmon waves, with subsequent modulation by molecular binding events. The following diagram illustrates this fundamental process and how algorithmic optimization enhances each stage.
SPR Signaling Pathway and Algorithm Enhancement
The integration of algorithmic optimization with SPR sensor design represents a paradigm shift in biosensing technology, moving beyond traditional trial-and-error approaches toward predictive, data-driven design methodologies. The demonstrated achievements—including attomolar detection limits, 230% sensitivity enhancements, and comprehensive performance improvements across multiple metrics—underscore the transformative potential of these approaches [53] [54]. As algorithm-assisted design continues to evolve, several emerging trends promise to further advance the capabilities of SPR biosensors.
The convergence of multi-objective optimization with explainable artificial intelligence creates a powerful framework for both optimizing sensor performance and understanding the fundamental physical principles governing these enhancements [55] [56]. Meanwhile, the growing emphasis on miniaturization and point-of-care applications drives algorithmic innovations tailored to portable SPR systems with reduced dimensionality and complexity [37] [57]. The integration of high-throughput screening capabilities with algorithm-optimized sensors further expands the technology's utility in pharmaceutical development and clinical diagnostics [37].
Looking forward, algorithm-assisted SPR design will increasingly focus on automated optimization pipelines that seamlessly transition from computational design to experimental implementation, potentially incorporating digital twin concepts where virtual sensor replicas guide real-world optimization [55] [58]. These advancements, coupled with the growing accessibility of machine learning tools and computational resources, promise to democratize ultra-sensitive SPR technology, making single-molecule detection capabilities available to broader research communities and accelerating discoveries across life sciences, medical diagnostics, and pharmaceutical development [53] [55] [54].
Surface Plasmon Resonance (SPR) is a cornerstone optical technique for label-free detection, enabling researchers to monitor biomolecular interactions—such as protein-ligand binding or antibody-antigen recognition—in real-time without the need for fluorescent or radioactive labels [59] [60]. The fundamental principle involves the excitation of surface plasmons, which are coherent electron oscillations at the interface between a metal (typically gold or silver) and a dielectric medium (e.g., a buffer solution) [61]. When the energy and momentum of incident light, usually passed through a prism, match that of the surface plasmons, a resonance condition is met. This results in a sharp dip in the intensity of the reflected light, measured as the SPR angle [60]. Any change in the refractive index at the metal surface, such as that caused by molecules binding to it, shifts this resonance angle, providing a direct and quantifiable measure of the binding event [61].
The significance of SPR in modern biosensing is profound. It provides critical insights into binding kinetics, affinity, specificity, and concentration of biomolecules, which are indispensable for drug discovery, diagnostic development, and biological research [38] [60]. The global label-free detection market, where SPR is a dominant technology, is projected to grow significantly, reflecting its increasing adoption [38] [59]. However, the performance and sensitivity of conventional SPR sensors are limited by the properties of the materials used. This has driven extensive research into novel materials and structures—including two-dimensional (2D) nanomaterials, metal oxides like titanium dioxide (TiO₂), and specialized D-shaped optical fibers—to push the boundaries of sensitivity, detection limit, and overall figure of merit (FOM) [62] [44] [61].
The Role of 2D Materials in SPR Enhancement
Two-dimensional nanomaterials have emerged as powerful components for enhancing SPR biosensors due to their exceptional surface-to-volume ratio, strong adsorption capabilities for various analyte molecules, and unique optoelectronic properties [61]. These materials, when used as an ultra-thin layer on top of the plasmonic metal, significantly intensify the local electromagnetic field and enhance the sensor's interaction with target molecules.
Graphene and its Derivatives: Graphene was one of the first 2D materials explored for SPR enhancement. Its single-atom-thick structure provides a large surface area for molecular adsorption and exhibits high charge carrier mobility, which can modify the surface plasmon waves [61]. Recent studies have investigated fluorinated graphene (FG), where the controlled addition of fluorine atoms allows for tuning its optical and chemical properties. One proposed sensor design using an Al/TiO₂/FG structure demonstrated a remarkably high Figure of Merit (FOM) of 462.8 RIU⁻¹ in the near-infrared region, showcasing FG's potential for high-performance sensing [63].
Transition Metal Dichalcogenides (TMDCs): Materials like Molybdenum Disulfide (MoS₂) and Tungsten Diselenide (WSe₂) offer advantages over graphene, including a tunable bandgap and higher absorption coefficients. The strong "soft-soft" interaction between sulfur atoms in MoS₂ and mercury ions (Hg²⁺) has been exploited to create an ultrasensitive SPR sensor for environmental monitoring, achieving a detection limit as low as 1.0 pM for Hg²⁺ in water—200 times more sensitive than conventional methods [64]. First-principles studies suggest that TMDCs can provide a superior platform for adsorbing certain biomolecules compared to graphene [62].
MXenes: This class of 2D transition metal carbides/nitrides (e.g., Ti₃C₂Tx) combines metallic conductivity with hydrophilic surfaces, making them excellent for biomolecular sensing. A theoretical study modeled an SPR sensor using a Copper/Silicon Nitride/MXene stack for cancer detection, reporting an angular sensitivity of 312° RIU⁻¹ for a breast cancer model. This represents a more than doubling of sensitivity compared to a sensor with only a copper layer [65].
Black Phosphorus (BP): Known for its high charge carrier density and puckered structure, BP provides a large surface area and strong interaction with analyte molecules. In a multi-layer sensor design incorporating Ag, TiO₂, and BP for detecting glucose and hemoglobin, a high sensitivity of 682.34 °/RIU was achieved, underscoring BP's significant enhancement potential [66].
Table 1: Performance Comparison of SPR Sensors Enhanced by Different 2D Materials
| 2D Material | Sensor Structure | Target Analyte | Key Performance Metric | Reference |
|---|---|---|---|---|
| Fluorinated Graphene (FG) | Al / TiO₂ / FG | Hemoglobin | FOM: 462.8 RIU⁻¹ | [63] |
| MoS₂ (Superstructure) | Au / MoS₂ | Mercury Ions (Hg²⁺) | Detection Limit: 1.0 pM | [64] |
| MXene (Ti₃C₂Tx) | Cu / Si₃N₄ / MXene | Cancer Biomarkers | Sensitivity: 312 °/RIU | [65] |
| Black Phosphorus (BP) | Ag / TiO₂ / BP | Glucose, Hemoglobin | Sensitivity: 682.34 °/RIU | [66] |
| Graphene | Ag / Graphene | General Bio-sensing | FOM improved ~3.5x with Cu | [62] |
Titanium Dioxide (TiO₂) as a Performance-Multiplying Layer
The integration of dielectric layers like titanium dioxide (TiO₂) has proven to be a highly effective strategy for boosting SPR sensor performance. TiO₂ acts as a high-refractive-index spacer, strategically positioned to engineer the distribution of the electromagnetic field, thereby enhancing the evanescent field at the sensor-analyte interface. This leads to a stronger interaction with the target molecules and a more pronounced shift in the resonance signal [63].
The performance multiplier effect of TiO₂ is clearly demonstrated in a numerical study of a D-shaped photonic crystal fiber (PCF) SPR biosensor. The design featuring a gold layer topped with TiO₂ achieved an exceptional wavelength sensitivity of 42,000 nm/RIU and a high FOM of 1393.128 RIU⁻¹ for the detection of various cancer cells (e.g., Basal, HeLa, MDA-MB-231) [44]. The TiO₂ layer not only enhanced sensitivity but also contributed to the sensor's stability. Beyond performance enhancement, a secondary and crucial role of TiO₂ and similar dielectric layers is to protect the plasmonic metal from oxidation and corrosion, particularly when using more reactive but highly sensitive metals like silver or copper [63]. This dual functionality makes TiO₂ an invaluable component in the design of robust, high-performance SPR sensors.
D-Shaped Fiber Structures: Practical Sensitivity Gains
While the traditional Kretschmann prism configuration remains the bedrock of SPR technology, D-shaped optical fibers present a innovative and practical platform that offers several distinct advantages [44] [61]. These sensors are fabricated by polishing a standard optical fiber to create a flat, D-shaped surface, onto which the metal and sensing layers are deposited. This geometry overcomes a significant fabrication challenge: achieving a uniform, thin metal coating with minimal roughness on a curved fiber surface. The flat platform ensures a homogeneous layer and brings the fiber core closer to the metal, facilitating more efficient coupling between the core mode and the surface plasmon polariton (SPP) mode [44].
The practical benefits of this structure translate directly into performance gains. The D-shaped PCF-SPR sensor with Au/TiO₂ layers mentioned previously is a prime example, achieving its high sensitivity and FOM partly due to this optimized geometry [44]. Furthermore, the fiber-optic approach enables miniaturization, remote sensing capabilities, and requires smaller sample volumes compared to bulky prism-based systems. This makes D-shaped fiber SPR sensors particularly attractive for point-of-care diagnostics and field-deployable environmental monitoring devices [44] [61].
Synergistic Material Combinations and Performance Metrics
The most significant advances in SPR sensitivity are achieved by combining different classes of novel materials to create synergistic heterostructures. These multi-layer stacks are engineered to leverage the unique advantages of each material, resulting in a collective performance that surpasses the capabilities of any single layer.
A key demonstration of this principle is the substitution of the conventional silver layer with copper in a 2D-material-based sensor. While silver typically offers higher sensitivity, it suffers from oxidation and tarnishing. Research has shown that a copper-based sensor, when combined with appropriate 2D materials, can achieve a 3.3 to 3.6-fold increase in Figure of Merit (FOM) compared to standard designs, successfully balancing performance with stability and cost [62]. Another powerful combination involves using a dielectric spacer layer (e.g., Silicon Nitride, Si₃N₄) together with a 2D material. The dielectric layer serves to confine and sharpen the plasmonic resonance, while the 2D material on top provides a high-affinity substrate for capturing analyte molecules. This architecture was validated in a Cu/Si₃N₄/MXene stack, which showed a more than 100% enhancement in sensitivity compared to a copper-only sensor [65].
Table 2: High-Performance SPR Sensor Designs Utilizing Combined Material Strategies
| Sensor Structure | Interrogation Method | Target Application | Sensitivity | Figure of Merit (FOM) | Reference |
|---|---|---|---|---|---|
| Au / TiO₂ / D-Shaped PCF | Wavelength (NIR) | Multi-Cancer Detection | 42,000 nm/RIU | 1393.128 RIU⁻¹ | [44] |
| Al / TiO₂ / Fluorinated Graphene | Angle (NIR, 1550 nm) | Bio-sensing (e.g., Hemoglobin) | Not Specified | 462.8 RIU⁻¹ | [63] |
| Ag / TiO₂ / Black Phosphorus | Angle | Glucose & Hemoglobin | 682.34 °/RIU | 171.698 RIU⁻¹ | [66] |
| Cu / Si₃N₄ / MXene | Angle (633 nm) | Cancer Biomarkers | 312 °/RIU | ~48-58 RIU⁻¹ (Quality Factor) | [65] |
To objectively compare the performance of these advanced sensors, several key metrics are used:
- Sensitivity: The magnitude of sensor output change (e.g., resonance angle or wavelength shift) per unit change in refractive index (RIU) [61] [65].
- Figure of Merit (FOM): A comprehensive metric often defined as Sensitivity divided by the spectral width of the resonance dip, providing a balanced measure of sensor quality [63] [65].
- Detection Accuracy (DA): Related to the sharpness of the resonance dip, calculated as the ratio of the resonance shift to the full width at half maximum [65].
- Limit of Detection (LoD): The smallest change in refractive index that the sensor can reliably distinguish from noise [65].
Experimental Protocols and the Scientist's Toolkit
Implementing novel material-enhanced SPR sensors requires meticulous experimental procedures, from sensor fabrication to data acquisition. The following protocol outlines the key steps for a prism-based configuration, which can be adapted for D-shaped fibers.
Sensor Fabrication and Functionalization
- Substrate Preparation: Begin with a clean glass prism (e.g., SF11, BK7). The surface must be free of contaminants for uniform metal deposition [62] [63].
- Plasmonic Metal Deposition: Deposit a thin film (typically 40-55 nm) of plasmonic metal (e.g., Gold, Silver, or Copper) onto the prism face using techniques such as sputtering or thermal evaporation. The precise thickness is critical and must be optimized for the specific design [65] [66].
- Dielectric/2D Material Transfer: Subsequent layers are added sequentially.
- For a dielectric layer like TiO₂, a precise thickness (e.g., 5-15 nm) is deposited via sputtering [63].
- For 2D materials (graphene, MoS₂, MXene), transfer the nanomaterial layer onto the metal/dielectric surface. This can be achieved using wet transfer techniques or Langmuir-Blodgett assembly [64] [65]. The number of layers significantly impacts performance and must be carefully controlled [64].
- Surface Functionalization: To impart specificity, immobilize a biorecognition element (e.g., an antibody, DNA probe, or enzyme) onto the sensor surface. This often involves creating a self-assembled monolayer (SAM) on the metal or using the 2D material's functional groups for covalent attachment [64] [60].
Data Acquisition and Analysis
- Instrument Setup: Mount the sensor chip in the SPR instrument, forming the base of a flow cell. Use a polarizer to ensure the incident light is p-polarized [44] [60].
- Baseline Establishment: Flow a running buffer through the cell until a stable baseline reflectance is achieved.
- Ligand Immobilization: If not done during fabrication, introduce the ligand solution to be captured on the functionalized surface [60].
- Analyte Injection: Introduce the analyte (sample) in the buffer over the sensor surface. Biomolecular binding causes a local change in refractive index, shifting the SPR angle or wavelength [60].
- Real-Time Monitoring: Continuously monitor the reflectance (at a fixed angle or wavelength) to obtain a sensorgram—a plot of response versus time. The association and dissociation phases provide raw kinetic data [60].
- Regeneration: Remove the bound analyte using a regeneration buffer (low pH or other mild denaturing conditions) to prepare the sensor for a new cycle [60].
- Data Processing: Analyze the sensorgram using appropriate software to extract kinetic parameters (association rate kᵒⁿ, dissociation rate kᵒff) and the equilibrium dissociation constant (K_D) [60].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Materials and Reagents for Advanced SPR Sensor Development
| Category / Item | Specific Examples | Function in SPR Sensor Development |
|---|---|---|
| Plasmonic Metals | Gold (Au), Silver (Ag), Copper (Cu), Aluminum (Al) | Forms the core layer for generating surface plasmon waves. Choice affects sensitivity and chemical stability. |
| 2D Nanomaterials | Graphene, Fluorinated Graphene (FG), MoS₂, WS₂, MXene (Ti₃C₂Tx), Black Phosphorus (BP) | Enhances sensitivity and provides a high-surface-area platform for immobilizing probe molecules. |
| Dielectric Spacers | Titanium Dioxide (TiO₂), Silicon Nitride (Si₃N₄) | Enhances field confinement, protects the metal from oxidation, and improves overall sensor performance. |
| Prism Couplers | BK7, SF10, SF11 Glass Prisms | Enables phase-matching between incident light and surface plasmons in the Kretschmann configuration. |
| Optical Components | Polarizer, Tunable NIR Laser (1550 nm), Optical Spectrum Analyzer | Provides controlled, polarized light and detects the output signal for analysis. |
| Bio-Reagents | Antibodies, DNA Probes, Urease, Specific Proteins | Serves as the biorecognition element to impart specificity for the target analyte (e.g., cancer marker, virus, ion). |
The integration of novel materials such as 2D nanomaterials, titanium dioxide, and the adoption of D-shaped fiber geometries is unequivocally advancing the capabilities of Surface Plasmon Resonance sensors. These innovations directly address the core needs of label-free detection research by delivering substantial improvements in sensitivity, detection limit, and overall sensor performance. The synergistic combination of different material classes into heterostructures represents the most promising path forward, enabling researchers to tailor sensor properties for specific applications, from ultra-sensitive cancer biomarker detection to environmental monitoring of trace pollutants.
Future research will likely focus on several key areas. The integration of Artificial Intelligence (AI) and Machine Learning (ML) is poised to play a larger role in optimizing sensor designs and analyzing complex data from multiplexed detection [38]. Furthermore, the push toward point-of-care diagnostics will drive the development of more robust, portable, and cost-effective SPR systems, where D-shaped fibers and stable, non-precious metal combinations (like copper with protective layers) will be crucial [44] [65]. As the synthesis and transfer techniques for 2D materials continue to mature, their widespread implementation in commercial SPR platforms will accelerate, unlocking new possibilities in drug discovery, clinical diagnostics, and fundamental biological research.
Label-free detection strategies represent a powerful class of technical methods for analyzing target molecules without requiring covalent labeling with signal probes such as fluorophores or electrochemical indicators. These methods instead rely on the inherent properties of analytes, including refractive index, electrical impedance, surface charge, size, and atomic mass for determination [28]. Among the various label-free technologies, surface plasmon resonance (SPR) has emerged as a particularly versatile platform for chemical and biological sensing and imaging [67].
SPR biosensors have become indispensable tools in life sciences, therapeutic drug monitoring, and clinical diagnostics due to their significant advantages: they enable real-time monitoring of molecular interactions, require no fluorescent or radioactive labels, need only small sample volumes, and can reuse sensor chips for multiple analyses [68]. The technology has evolved remarkably from conventional SPR spectroscopy to surface plasmon resonance microscopy (SPRM), which provides unprecedented capability for imaging single entities ranging from several nanometers to micrometers with high spatiotemporal resolution [67]. This advancement has opened new frontiers in pushing detection limits to single-molecule levels, a critical capability for understanding fundamental biological processes and developing ultra-sensitive diagnostic assays.
Fundamental Principles of SPR Biosensing
The Physical Mechanism of SPR
Surface plasmon resonance occurs when incident light, under specific conditions, excites collective oscillations of free electrons at a metal-dielectric interface. These electron oscillations, known as surface plasmon polaritons (SPPs), propagate along the interface and create an evanescent wave that decays exponentially in the vicinity of the interface [67]. The resonance condition is extremely sensitive to subtle refractive index variations adjacent to the interface (within approximately 300 nm), making it ideal for detecting molecular binding events [28] [69].
The most common implementation of SPR uses the Kretschmann configuration in a total internal reflection geometry. In this setup, a beam of p-polarized monochromatic light is focused onto a gold-coated glass substrate. When the incident angle matches the resonance condition, a sharp decrease in reflectivity occurs due to the transformation of radiant energy into surface plasmons [67]. The resonance angle (θR) is given by:
[ \sin(\thetaR) = \sqrt{\frac{\varepsilon1 \varepsilonm}{(\varepsilon1 + \varepsilonm) \varepsilon2}} ]
where ε₁ is the dielectric constant of the buffer solution, ε₂ is the dielectric constant of the objective, and εm is the real part of the dielectric constant of the metal film [67].
From Bulk Sensing to Single-Molecule Detection
Traditional SPR sensors detect biomolecular interactions by measuring shifts in the resonance angle caused by changes in refractive index at the sensor surface. While powerful for studying binding kinetics and affinity, conventional SPR lacks the sensitivity for single-molecule detection. The evolution to SPRM has addressed this limitation by incorporating a high numerical aperture (NA) objective, providing unprecedented spatial resolution and enabling the visualization and analysis of single molecules [67].
When a single molecule or nanoparticle interacts with the sensor surface, it creates a distinct parabolic-shaped diffraction pattern due to the intrinsic interferential detection scheme of SPRM. This characteristic pattern enables the detection and analysis of individual molecular binding events, even for particles below the diffraction limit in size [67].
Diagram 1: Fundamental SPR working principle and signal transduction pathway.
Advanced Strategies for Enhanced Sensitivity
Nanomaterial-Enhanced SPR Sensing
The integration of novel nanomaterials has dramatically improved SPR sensor performance for low-abundance analyte detection. Two-dimensional materials with high specific surface areas and robust analyte binding capabilities have proven particularly effective [53]. These materials enhance sensitivity through several mechanisms: increased surface area for biorecognition element immobilization, improved mass transfer characteristics, and unique optical properties that strengthen the plasmonic effect.
Recent research demonstrates that carbon nanomembranes (CNMs) as thin as 1 nm can significantly enhance sensitivity when used to functionalize SPR sensors. In one study, CNMs terminated with azide linkers enabled covalent bonding of SARS-CoV-2 antibodies, allowing specific immobilization of viral proteins to the sensor surface. This approach achieved remarkably low detection limits of approximately 190 pM for nucleocapsid protein and 10 pM for spike protein [69].
Other promising nanomaterials include graphene, molybdenum disulfide (MoS₂), tungsten disulfide (WS₂), and black phosphorus, though challenges with stability and reproducible fabrication remain [53]. These materials can be implemented as thin films on the metal sensor surface or as functional nanoparticles conjugated with recognition elements.
Algorithm-Assisted Optimization
Comprehensive optimization of SPR sensor design parameters through computational approaches represents a powerful strategy for enhancing sensitivity. Multi-objective optimization using particle swarm optimization (PSO) algorithms can concurrently optimize multiple sensing parameters, including sensitivity (S), figure of merit (FOM), and depth of resonant dip (DRD) [53].
This approach optimizes critical design parameters such as incident angle, adhesive layer thickness (e.g., chromium), and metal layer thickness (e.g., gold). Recent implementations have demonstrated dramatic improvements: 230.22% increase in bulk refractive index sensitivity, 110.94% improvement in FOM, and 90.85% enhancement in DFOM compared to conventional designs. The optimized sensor achieved a detection limit of 54 ag/mL (0.36 aM) for mouse IgG, enabling effective identification of low-abundance single molecules at femtogram-per-milliliter concentrations [53].
Signal Amplification Strategies
Various signal amplification strategies have been developed to push SPR detection limits to single-molecule levels:
Plasmonic-Electrochemical Impedance Microscopy (P-EIM): This hybrid technique applies an alternating current potential to the sensing surface, tuning the electron density and dielectric constant of the metal surface. P-EIM converts electrical signals into optical readouts, providing local impedance information with superior sensitivity to surface charge instead of mass [67].
Magnetic Nanoparticle Sandwich Assays: Utilizing target analytes captured by receptor-modified magnetic nanoparticles, with biotinylated recognition elements attached to form sandwich hybrid structures. These structures are delivered to neutravidin-modified SPR fluidic channels, resulting in enhanced signals down to 1 fM for DNA and 10 fM for Aβ40 [68].
Nucleic Acid Amplification Integration: Combining SPR with amplification techniques like hybridization chain reaction (HCR), rolling circle amplification (RCA), and catalytic hairpin assembly (CHA) to achieve highly sensitive detection [68].
Table 1: Performance Comparison of Advanced SPR Strategies for Low-Abundance Detection
| Strategy | Detection Limit | Analyte | Key Innovation | Reference |
|---|---|---|---|---|
| CNM-functionalization | 10 pM | SARS-CoV-2 S-protein | 1 nm thick carbon nanomembranes | [69] |
| Multi-objective PSO Optimization | 0.36 aM (54 ag/mL) | Mouse IgG | Algorithm-assisted design optimization | [53] |
| SPR Microscopy | Single molecules | DNA helicase | 3D tracking of 100 single molecules | [70] |
| Magnetic Sandwich Hybrids | 1 fM | DNA | Magnetic preconcentration & signal amplification | [68] |
| Aptamer-based SPR | ~pM range | Various viruses | High-specificity aptamer receptors | [71] |
Experimental Protocols for Single-Molecule SPR
Sensor Surface Functionalization with CNMs
The following protocol details the functionalization of SPR sensors with carbon nanomembranes for ultra-sensitive detection, adapted from successful SARS-CoV-2 protein detection studies [69]:
Surface Preparation: Clean gold-coated SPR sensor chips using oxygen plasma treatment or piranha solution (3:1 concentrated H₂SO₄:30% H₂O₂) for 10 minutes, followed by thorough rinsing with ethanol and deionized water.
Self-Assembled Monolayer (SAM) Formation: Immerse the clean gold substrate in a 1 mM solution of 4'-nitro-[1,1']-biphenyl-4-thiol (NBPT) in ethanol for 24 hours at room temperature under nitrogen atmosphere to form a uniform SAM.
CNM Formation via Electron Irradiation: Expose the NBPT SAM to low-energy electrons (approximately 100 eV) in a vacuum chamber to convert the nitro groups to amino groups, forming an amino-terminated CNM (NH₂-CNM) approximately 1 nm thick.
Azide Functionalization: React the NH₂-CNM with azidoacetyl chloride linker (1 mM in dichloromethane with 0.1% triethylamine) for 4 hours at room temperature to form azide-terminated CNM (N₃-CNM).
Antibody Functionalization: Modify specific antibodies (e.g., anti-SARS-CoV-2) with dibenzocyclooctyne (DBCO) linkers using N-hydroxysuccinimide (NHS) ester reaction. Incubate DBCO-modified antibodies (10 μg/mL in PBS, pH 7.4) with the N₃-CNM surface for 2 hours at 25°C for copper-free click chemistry conjugation.
Surface Passivation: Block non-specific binding sites by incubating with casein solution (1% in PBS) for 1 hour at room temperature.
Each functionalization step should be verified using surface characterization techniques such as X-ray photoelectron spectroscopy (XPS) and polarization-modulation infrared reflection absorption spectroscopy (PM-IRRAS) [69].
Multiparametric SPR Measurements
Multiparametric SPR operating at multiple wavelengths (e.g., 670 nm, 785 nm, and 980 nm) significantly enhances measurement accuracy and sensitivity by simultaneously quantifying both thickness and refractive index of the analyte layer [69]:
Instrument Calibration: Calibrate the SPR instrument using standard solutions with known refractive indices. Establish a stable baseline with running buffer (typically HBS-EP: 10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.005% surfactant P20, pH 7.4) at a constant flow rate (typically 30 μL/min).
Ligand Immobilization: Immobilize the capture molecule (antibody, aptamer, etc.) using standard amine coupling, streptavidin-biotin interaction, or the previously described CNM method. The immobilization level should be optimized for the specific application (typically 5000-15000 response units).
Analyte Binding Kinetics: Inject analyte samples in series of concentrations (covering a range of at least 100-fold) with contact times sufficient to observe association, followed by dissociation phases. Use higher flow rates (≥30 μL/min) to minimize mass transport limitations.
Data Collection: Monitor real-time shifts of the resonance angle (θSPR) simultaneously at multiple wavelengths. Include reference channel measurements to correct for bulk refractive index changes and non-specific binding.
Data Analysis: Determine kinetic parameters (association rate kₐ, dissociation rate kd) and equilibrium dissociation constant (KD) by globally fitting the binding data to appropriate interaction models (1:1 Langmuir, two-state reaction, etc.) using software such as BIAevaluation or Scrubber.
Diagram 2: Logic flow for algorithm-assisted optimization of SPR biosensors.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Single-Molecule SPR Studies
| Material/Reagent | Function | Application Notes | Reference |
|---|---|---|---|
| Carbon Nanomembranes (CNMs) | Ultra-thin 2D functionalization layer | 1 nm thick; enables covalent antibody immobilization; enhances sensitivity | [69] |
| DBCO-NHS Ester | Crosslinker for copper-free click chemistry | Reacts with antibody amines; binds to azide-functionalized surfaces | [69] |
| Gold Sensor Chips | SPR-active substrate | Standard 50 nm gold films on glass with 2 nm chromium adhesion layer | [53] |
| Magnetic Nanoparticles (MNPs) | Signal amplification | Form sandwich structures; enable magnetic preconcentration | [68] |
| Specific Aptamers | Biorecognition elements | DNA/RNA sequences with high specificity; replace antibodies in some applications | [71] [68] |
| Casein | Blocking agent | Reduces non-specific binding; more effective than BSA for certain applications | [69] |
| Multi-wavelength SPR System | Instrumentation | Enables simultaneous thickness and refractive index measurement | [69] |
| Particle Swarm Optimization Algorithm | Computational design | Multi-objective optimization of sensor parameters | [53] |
Future Perspectives and Challenges
The field of SPR-based single-molecule detection continues to evolve with several promising research directions. Integration with other single-molecule techniques such as fluorescence correlation spectroscopy and atomic force microscopy could provide complementary information about molecular interactions. Development of novel plasmonic materials including metamaterials and hyperbolic metamaterials may offer unprecedented control over light-matter interactions at the nanoscale [28].
The miniaturization of SPR systems into lab-on-a-chip platforms and point-of-care devices represents another important direction, potentially enabling widespread use in clinical diagnostics and environmental monitoring [68]. Additionally, machine learning-assisted data analysis could extract more information from complex single-molecule binding events, potentially distinguishing between different interaction modalities and conformational states [53].
Despite significant progress, challenges remain in standardizing single-molecule SPR protocols, improving throughput for statistical significance, and reducing non-specific binding at ultra-low concentrations. The dynamic nature of biological systems also presents challenges in maintaining native conformations of biomolecules during surface immobilization and analysis.
As these technical hurdles are addressed, SPR biosensors will continue to push the boundaries of detection sensitivity, enabling new discoveries in fundamental biology and transformative applications in diagnostics and therapeutic monitoring. The convergence of nanomaterials engineering, computational optimization, and innovative signal amplification strategies promises to make single-molecule analysis by SPR increasingly accessible and informative for the scientific community.
Surface Plasmon Resonance (SPR) is a powerful, label-free technology that enables the real-time analysis of biomolecular interactions by detecting changes in the refractive index at a metal sensor surface [72]. Its application spans critical fields from drug discovery to clinical diagnostics [73] [74]. However, the data generated by SPR biosensors can be compromised by several experimental artifacts, primarily bulk effects, non-specific binding, and regeneration issues. Effectively navigating these challenges is essential for obtaining kinetically and quantitatively accurate results, thereby ensuring the reliability of the research. This guide provides an in-depth technical examination of these core artifacts, offering researchers detailed methodologies for their identification and mitigation.
Bulk Effects: Identification and Compensation
Understanding Bulk Effect Artifacts
Bulk effects, or bulk refractive index changes, occur when the overall composition of the running buffer flowing over the sensor chip changes, shifting the SPR signal independently of any specific binding interaction. These shifts are indistinguishable from true binding events in a standard sensorgram and are a common source of false positives and inaccurate kinetics [72]. A key characteristic of a bulk effect is that it causes an immediate, sharp signal change that stabilizes rapidly and reverses immediately when the buffer composition returns to its original state.
Common scenarios that induce bulk effects include:
- Sample Solvent Effects: Running a sample dissolved in a buffer containing DMSO, glycerol, or other solvents different from the running buffer.
- Buffer Inconsistencies: Differences in salt concentration, pH, or other additives between the sample buffer and the running buffer.
- Complex Media Analysis: Introducing crude samples like serum, cell lysates, or nasopharyngeal swab samples directly into the system [69] [75].
Experimental Protocols for Mitigation
A. Reference Channel Subtraction The most robust and widely used method to correct for bulk effects is the use of a reference channel on the SPR instrument [69].
- Procedure:
- Surface Preparation: A reference flow cell is prepared identically to the active sensor surface but without the immobilization of the ligand (capture molecule). For instance, if the active surface is functionalized with an antibody, the reference surface should undergo the same functionalization and blocking steps, omitting only the antibody.
- Data Collection: The sample is injected simultaneously over both the active and reference surfaces.
- Data Processing: The response from the reference channel is electronically or digitally subtracted from the response of the active channel. The resulting signal represents the specific binding interaction, with the bulk effect artifact largely eliminated.
B. Careful Sample Preparation Standardizing buffer conditions is a fundamental preventive measure.
- Dialysis or Buffer Exchange: Prior to analysis, the analyte should be dialyzed into the exact running buffer used in the SPR experiment.
- Sample Dilution: If buffer exchange is not feasible, the sample should be diluted into the running buffer immediately before injection. This ensures the DMSO concentration and ionic strength match the running buffer as closely as possible. Modern systems like Alto Digital SPR are particularly compatible with analyzing compounds in complex media, broadening the application scope [75].
Non-Specific Binding (NSB): Strategies for Reduction
Defining Non-Specific Binding
Non-specific binding (NSB) occurs when analyte molecules adsorb to the sensor surface through interactions other than the specific, high-affinity interaction being studied. This can include electrostatic, hydrophobic, or van der Waals interactions with the chip matrix, the ligand, or the surrounding chemical layers [69]. NSB leads to an overestimation of binding response, complicates data interpretation, and can obscure weak but specific interactions.
Experimental Protocols for Mitigation
A. Surface Passivation with Blocking Agents A critical step after ligand immobilization is to "block" the remaining reactive or adhesive sites on the sensor surface.
- Procedure:
- Following ligand immobilization, inject a solution of an inert, non-reactive protein or polymer.
- Allow the solution to incubate for a sufficient time (e.g., 10-30 minutes) to saturate non-specific sites.
- Wash thoroughly with running buffer to remove unbound blocking agent.
- The choice of blocking agent is empirical. As demonstrated in SARS-CoV-2 sensor development, casein was found to be the most effective in reducing non-specific adsorption of antigens compared to other agents [69]. Other common blocking agents include Bovine Serum Albumin (BSA) and synthetic polymers like poly(ethylene glycol) (PEG).
B. Optimized Surface Functionalization The chemistry used to attach the ligand to the sensor surface can profoundly influence NSB. Advanced functionalization strategies can create a more bio-inert background.
- Protocol: Functionalization with Carbon Nanomembranes (CNMs) [69]:
- Form a self-assembled monolayer (SAM) of 4’-nitro-[1,1’]-biphenyl-4-thiol (NBPT) on a gold sensor chip.
- Convert the NBPT SAM into an ~1 nm thick amino-terminated CNM (NH2-CNM) via low-energy electron irradiation.
- Graft an azide linker (azidoacetyl chloride) to the amino groups to form an azide-terminated CNM (N3-CNM).
- Functionalize the SARS-CoV-2 antibodies with dibenzocyclooctyne (DBCO) linkers.
- Covalently attach the DBCO-modified antibodies to the N3-CNM surface via copper-free click chemistry.
- Passivate the surface with a blocking agent like casein. This hierarchical functionalization creates a stable, well-defined, and oriented antibody presentation that minimizes non-specific interactions, as confirmed by XPS and PM-IRRAS analysis [69].
C. Kinetic Data Analysis to Identify NSB True specific binding typically follows a classical 1:1 interaction model. Deviations from this model can indicate NSB.
- Signs of NSB in Sensorgrams:
- A rapidly associating signal that does not reach a steady state during injection.
- An incomplete or very slow dissociation phase, even with a strong regeneration solution, indicating the analyte is tightly stuck to the surface rather than specifically bound to the ligand.
- A poor fit of the experimental data to standard binding models.
Table 1: Common Blocking Agents and Their Applications
| Blocking Agent | Mechanism of Action | Typical Concentration | Best For | Considerations |
|---|---|---|---|---|
| Casein | Forms a passive protein layer, effective for reducing protein adsorption [69]. | 0.1 - 1% | General protein interactions, immunoassays | Excellent performance in reducing non-specific adsorption for viral protein detection [69]. |
| Bovine Serum Albumin (BSA) | Saturates hydrophobic and charged sites on the surface. | 0.1 - 1% | A wide range of biomolecular interactions | Inexpensive and widely available; can sometimes interact with certain analytes. |
| PEG-based Polymers | Creates a hydrated, bio-inert brush layer that sterically hinders approach. | Varies by polymer | Crude samples, serum analysis, low-affinity interactions | Highly effective; may require specific chemical coupling for surface attachment. |
| Ethanolamine | Blocks unreacted ester groups on NHS/EDC activated carboxyl surfaces [76]. | 1 M, pH 8.5 | Surfaces activated by EDC/NHS chemistry | A small molecule blocker, not sufficient for passivating the entire surface against proteins. |
Regeneration Issues: Achieving Surface Stability
The Challenge of Regeneration
Regeneration is the process of removing tightly bound analyte from the immobilized ligand without permanently damaging (denaturing) the ligand's binding activity. An ideal regeneration strategy completely resets the binding response to baseline while allowing the ligand to be used for multiple analysis cycles. Common issues include:
- Incomplete Regeneration: Residual signal leads to drifting baselines and inaccurate quantification in subsequent cycles.
- Over-Regeneration: A gradual, cycle-by-cycle loss of binding capacity due to ligand degradation.
Experimental Protocol for Scouting Regeneration Conditions
Finding the optimal regeneration solution is an empirical process that requires systematic scouting.
- Procedure:
- Immobilize the ligand on the sensor surface.
- Inject a single concentration of analyte to achieve a high binding response.
- Allow the dissociation phase to proceed in running buffer for a short time (1-2 minutes) to establish a baseline dissociation rate.
- Inject a candidate regeneration solution for 15-60 seconds.
- Monitor the sensorgram: a sharp drop in signal back to the original baseline is ideal.
- Re-inject the same analyte concentration. Compare the new binding response level to the initial one.
- A response loss of >10% indicates the regeneration condition is too harsh. A failure to return to baseline indicates it is too weak.
- Test a panel of solutions (e.g., low pH glycine, high salt, mild surfactants, chelating agents like EDTA) to identify the mildest effective condition.
Table 2: Common Regeneration Solutions and Their Targets
| Regeneration Solution | Typical Composition | Mechanism of Action | Application Example |
|---|---|---|---|
| Low pH | 10-100 mM Glycine-HCl, pH 2.0 - 3.0 | Disrupts electrostatic and hydrogen bonding interactions; can protonate key residues. | Antibody-antigen complexes [76]. |
| High pH | 10-50 mM NaOH, 1 mM NaOH | Disrupts hydrophobic interactions and can deprotonate key residues. | Some high-affinity protein-protein interactions. |
| High Salt | 1-4 M MgCl₂, NaCl, or KCl | Shields and disrupts electrostatic interactions. | DNA-protein complexes. |
| Chaotropic Agents | 1-6 M Guanidine-HCl | Disrupts the native structure of water, weakening hydrophobic interactions. | Very stable complexes. |
| Chelators | 10-100 mM EDTA | Removes essential metal ions from metal-dependent interactions. | Integrins, metalloproteases. |
| Mild Detergents | 0.05-0.5% SDS | Disrupts hydrophobic and electrostatic interactions. | A last resort for very sticky interactions; can denature the ligand. |
Case Study: Integrated Artifact Mitigation in SARS-CoV-2 Detection
A 2025 study on detecting SARS-CoV-2 proteins provides a compelling case study in integrated artifact management [69]. The researchers developed an SPR sensor functionalized with 1 nm thick carbon nanomembranes (CNMs) to immobilize antibodies against the virus's N- and S-proteins.
- Mitigating NSB and Ensuring Specificity: The hierarchical functionalization (NBPT SAM → NH₂-CNM → N₃-CNM → DBCO-Antibody) provided a stable, oriented antibody presentation. This was followed by passivation with casein, which was empirically determined to be the most effective blocking agent. The high specificity of the developed sensors was confirmed via their negligible cross-reactivity with SARS-CoV-1 and MERS-CoV proteins.
- Overcoming Regeneration Challenges: The covalent nature of the antibody immobilization via click chemistry created a robust sensor surface. This stability allowed the sensor to be stored at 4°C for over a year while retaining functionality, indicating excellent resistance to ligand degradation over many regeneration cycles.
- Achieving High Sensitivity: This comprehensive approach resulted in sensors with exceptionally low limits of detection (LOD of ~10 pM for the S-protein) and precise kinetic measurements (K_D of 22 ± 2 pM for the S-protein), demonstrating the payoff of meticulous artifact control [69].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for SPR Artifact Management
| Reagent/Material | Function | Example Use Case |
|---|---|---|
| Carbon Nanomembranes (CNMs) | Ultra-thin 2D functionalization layer for stable, oriented ligand immobilization [69]. | Creating highly sensitive and specific biosensors for viral detection [69]. |
| EDC/NHS Chemistry | Standard carbodiimide crosslinking chemistry for activating carboxyl groups on the sensor surface for covalent coupling to amine-containing ligands [76]. | Immobilizing antibodies on a carboxyl-terminated SAM; one of three strategies compared for AFP cancer biomarker detection [76]. |
| EDA/Glutaraldehyde Chemistry | An alternative coupling strategy that uses a longer linker (Ethylene Diamine and Glutaraldehyde) to attach amine-containing ligands to a carboxylated surface [76]. | Immobilizing AFP antibody; afforded the highest sensitivity in a comparative study [76]. |
| Casein | A highly effective protein-based blocking agent for passivating surfaces against non-specific binding [69]. | Reducing non-specific adsorption of SARS-CoV-2 antigens in nasopharyngeal swab samples [69]. |
| Glycine-HCl (Low pH Buffer) | A common regeneration solution for disrupting antibody-antigen complexes. | Regenerating an antibody-based sensor surface between analyte injections [76]. |
Experimental Workflow and System Relationships
The following diagram illustrates the core workflow of an SPR experiment and the specific points where the three major artifacts manifest and are mitigated.
Diagram: SPR Workflow and Artifact Management. This flowchart outlines the key stages of an SPR experiment (yellow, green, blue, and red nodes). The red, dashed nodes represent the core artifacts, which manifest at specific points in the workflow. The green nodes indicate the primary mitigation strategies that correspond to each experimental stage to prevent or correct these artifacts.
SPR Versus Other Techniques: Validation, Comparative Analysis, and Orthogonal Approaches
Within the broader thesis of how Surface Plasmon Resonance (SPR) functions in label-free detection research, it is crucial to understand its operational principles and how it compares to other key technologies in the field. Label-free detection methodologies have become indispensable in biologics research and drug discovery as they enable the real-time analysis of biomolecular interactions without the need for fluorescent or radioactive labels, thereby preserving the natural state and function of the molecules under investigation [77] [78]. Two well-established techniques that dominate this landscape are Surface Plasmon Resonance (SPR) and Biolayer Interferometry (BLI). These technologies provide critical insights into the kinetics, affinity, and specificity of interactions, which are fundamental parameters in therapeutic development [79] [80].
SPR and BLI, while sharing the core advantage of being label-free, are built on distinct physical principles and operational workflows. SPR is a flow-based system that relies on a continuous fluidic system to deliver analytes over a sensor surface [78] [81]. In contrast, BLI operates on a "dip-and-read" format, where biosensor tips are immersed into solutions containing the analyte [79] [77]. This fundamental difference in operation creates a cascade of implications for throughput, sensitivity, data quality, and appropriate application scenarios. This guide provides a detailed technical comparison of these two systems, equipping researchers and drug development professionals with the knowledge to select the optimal tool for their specific research objectives within the expanding label-free detection domain, a market poised to grow from $570 million in 2023 to $863 million by 2028 [82].
Core Principles and Instrumentation
The Mechanism of Surface Plasmon Resonance (SPR)
Surface Plasmon Resonance is an optical technique that measures molecular interactions in real-time by detecting changes in the refractive index at a metal surface. The core components of an SPR instrument include a sensor chip with a thin gold film, a microfluidic system, and an optical prism [78] [81]. The fundamental principle involves the immobilization of one binding partner (the ligand) onto the gold sensor surface. The other partner (the analyte) is then flowed over this surface in solution. When polarized light strikes the gold film at a specific angle, it excites surface plasmons (electron charge density waves), leading to a phenomenon known as surface plasmon resonance, which is observed as a sharp dip in the intensity of reflected light [81].
When an analyte binds to the immobilized ligand on the sensor surface, it causes a change in the mass at the surface, which in turn alters the local refractive index. This change shifts the resonance angle required to produce the plasmon resonance effect [78]. This shift, measured in resonance units (RU), is monitored in real-time and is directly proportional to the mass bound to the sensor surface, allowing for the detailed quantification of binding events without any labeling requirements [81]. The continuous flow in SPR provides a constant supply of fresh analyte and maintains a stable baseline, which is critical for obtaining high-resolution kinetic data [79].
The Mechanism of Biolayer Interferometry (BLI)
Biolayer Interferometry is also an optical, label-free technique but operates on a different principle and with a simpler apparatus. BLI uses disposable fiber-optic biosensors (or "dips"), on the tip of which one binding partner is immobilized [77] [78]. This biosensor tip is then dipped into a well containing the analyte solution. The core mechanism involves directing a beam of white light down the length of the sensor. This light is reflected at two surfaces: a reference layer at the tip of the sensor and the surface where the biomolecular layer is immobilized. The reflected lights interfere with each other, creating a characteristic interference pattern [77].
When the analyte binds to the immobilized ligand on the sensor tip, it increases the optical thickness (the physical thickness multiplied by the refractive index) of the biolayer. This change in thickness causes a shift in the interference pattern of the reflected white light [78] [81]. The shift in wavelength is measured in real-time and is reported as a binding curve, or sensorgram. The "dip-and-read" format of BLI eliminates the need for complex microfluidics, simplifying the operational workflow and making the system inherently suitable for higher-throughput screening applications [79] [77].
Comparative Workflow Visualization
The fundamental difference between SPR and BLI is their operational workflow. The following diagram illustrates the step-by-step processes for both techniques, highlighting the flow-based versus dip-and-read methodologies.
Technical Comparison: Performance and Practical Application
Quantitative Performance and Workflow Comparison
The distinct operational principles of SPR and BLI translate into direct differences in performance metrics and practical workflow requirements. The following table summarizes these critical parameters to facilitate an objective comparison.
Table 1: Technical and Workflow Comparison of SPR and BLI [83] [79] [77]
| Feature | Surface Plasmon Resonance (SPR) | Biolayer Interferometry (BLI) |
|---|---|---|
| Core Principle | Measures refractive index changes via resonance angle shift on a gold film. [81] | Measures thickness changes of biomolecular layers via interference pattern shifts. [81] |
| Fluidics System | Requires complex microfluidics for continuous flow. [81] | Fluidics-free; uses dip-and-read method. [77] |
| Throughput | Moderate; depends on the number of flow channels. [84] [81] | High; typically supports 96 or 384-well plates for parallel processing. [79] [77] |
| Sensitivity | High; excellent for detecting low-concentration samples and small molecules. [79] [81] | Moderate; better suited for medium/high concentrations. Less ideal for small molecules. [79] [81] |
| Sample Consumption | Low (e.g., ~2 µL for full kinetics on some modern systems). [83] | Relatively high (e.g., 40-220 µL per concentration). [83] |
| Hands-on Time | Lower in automated systems (e.g., 28 minutes reported). [83] | Can be significantly higher (e.g., 66 minutes reported). [83] |
| Data Quality | High-resolution kinetic curves; considered the gold standard for kinetics and affinity. [79] [84] | Good for screening; kinetic accuracy and resolution can be lower than SPR. [79] [81] |
| Key Artifacts | Potential for mass transport limitation; microfluidics can clog. [83] [79] | Evaporation over long runs; potential for rebinding due to static incubation. [78] |
Experimental Protocol Considerations
The choice between SPR and BLI significantly influences experimental design. Below is a detailed methodology for a typical kinetic characterization assay, highlighting the procedural differences.
Kinetic Characterization of an Antibody-Antigen Interaction
1. Ligand Immobilization (Common to Both):
- SPR: The antibody (ligand) is covalently immobilized onto a dextran-coated gold sensor chip (e.g., CM5 chip) using standard amine-coupling chemistry. This involves activating the dextran carboxyl groups with a mixture of EDC and NHS, followed by injection of the antibody and then deactivation of excess groups. [78]
- BLI: The antibody is immobilized onto a compatible biosensor tip (e.g., Anti-Human IgG Capture biosensor). The tip is typically hydrated, then dipped into a solution containing the antibody to allow capture, resulting in a loaded sensor. [77]
2. Baseline Establishment:
- SPR: The continuous flow of running buffer (e.g., HBS-EP) over the sensor surface establishes a stable refractive index baseline. [78]
- BLI: The loaded biosensor is dipped into a well containing only buffer to establish an optical baseline. [77]
3. Association Phase:
- SPR: Multiple concentrations of the antigen (analyte) are injected sequentially over the sensor surface using the microfluidic system. The binding is monitored in real-time as the analyte flows over the immobilized ligand. [78]
- BLI: The biosensor tip is transferred to wells containing different concentrations of the antigen. The tip remains immersed in the static solution for a defined period while binding is monitored. [77]
4. Dissociation Phase:
- SPR: The flow is switched back to running buffer alone. The dissociation of the analyte from the ligand is monitored as it is constantly washed away from the surface by the flow. [78] This controlled flow minimizes rebinding events.
- BLI: The biosensor is moved to a well containing only buffer. Dissociation occurs as the analyte diffuses away from the sensor into the static buffer. This environment can sometimes lead to rebinding of analyte. [78]
5. Regeneration:
- SPR: A regeneration buffer (e.g., glycine-HCl, pH 2.0) is injected for a short period to break the antibody-antigen bonds without damaging the immobilized ligand. The surface is then re-equilibrated with running buffer, ready for the next cycle. [78] This step requires careful optimization.
- BLI: The biosensor can be regenerated similarly by dipping it into a regeneration solution, or it may simply be discarded, which avoids regeneration optimization but increases consumable costs. [77]
Essential Research Reagent Solutions
Successful execution of SPR and BLI experiments relies on a set of core reagents and materials. The following table details these essential components and their functions.
Table 2: Key Research Reagent Solutions for SPR and BLI [77] [78] [85]
| Item | Function | Application Notes |
|---|---|---|
| Sensor Chips (SPR) | Provides the gold surface for ligand immobilization. Often have a covalently attached hydrogel (e.g., dextran) derivatized with functional groups. | Available with different surface chemistries (e.g., carboxymethylated for amine coupling, streptavidin for biotin capture). Choice depends on ligand properties and immobilization strategy. [78] |
| Biosensors (BLI) | Disposable fiber-optic tips functionalized for binding. Act as both the immobilization surface and the optical reader. | Available pre-functionalized for common applications (e.g., Protein A, Anti-IgG, Streptavidin, Ni-NTA for His-tagged proteins). Selection is critical for assay success. [77] [85] |
| Immobilization Reagents | Chemicals to covalently attach the ligand to the sensor surface. | For SPR amine coupling: EDC (1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide) and NHS (N-Hydroxysuccinimide) are standard. [78] |
| Running Buffer | The solution used to establish baseline, dilute analytes, and wash the surface. | Must be optimized to maintain protein stability and activity. Often contains a surfactant like Tween-20 to minimize non-specific binding. [78] |
| Regeneration Buffer | A solution that dissociates bound analyte from the ligand without denaturing it. | Requires extensive optimization. Common choices are low pH buffers (e.g., Glycine-HCl), high pH buffers, or high salt solutions. [78] |
| Microplates (BLI) | To hold analyte and buffer solutions for the dip-and-read process. | Typically 96-well or 384-well plates. Plate quality and sealing are important to prevent evaporation during long experiments. [77] |
Application Scenarios and Selection Guide
Ideal Use Cases for SPR and BLI
The decision to use SPR or BLI is not a matter of one technology being universally superior, but rather which is better suited to the specific research question and context.
SPR is typically the preferred choice for:
- High-Resolution Kinetic Studies: When precise determination of association ((k\text{on})) and dissociation ((k\text{off})) rate constants is paramount, such as in the lead optimization phase of drug discovery. The continuous flow provides superior data quality. [79] [81]
- Small Molecule Characterization: Due to its high sensitivity, SPR is more effective at detecting the subtle refractive index changes caused by the binding of low molecular weight compounds. [79]
- Complex Interaction Analysis: For studying interactions where meticulous control of the liquid environment is needed to minimize artifacts like rebinding or mass transport limitation. [83]
BLI excels in applications such as:
- High-Throughput Screening: Rapidly screening hundreds of samples, such as hybridoma supernatants for antibody discovery or fractions from chromatography purifications. [79] [77]
- Analysis of Crude Samples: The fluidics-free design makes BLI more robust for analyzing samples like cell culture supernatants, bacterial lysates, or sera without prior purification, as there is no risk of clogging microfluidics. [79] [77]
- Rapid Concentration Measurement: Quickly determining the titer of antibodies, viruses (e.g., AAV), or other proteins without the need for a standard curve. [77]
Complementary and Orthogonal Use
In many industrial and academic settings, SPR and BLI are not competitors but complementary tools used in an orthogonal strategy. A common workflow involves using BLI for the initial, high-throughput screening of a large number of candidates directly from crude mixtures due to its speed and simplicity. [79] [77] The most promising hits from this screen are then purified and subjected to detailed, high-resolution kinetic analysis using SPR to confirm affinity and kinetic parameters with high confidence. [79] This combined approach leverages the strengths of both platforms to maximize efficiency and data reliability throughout the drug discovery pipeline.
Within the framework of label-free detection research, both SPR and BLI serve as powerful, yet distinct, tools for elucidating biomolecular interactions. SPR, with its flow-based design, remains the gold standard for obtaining high-quality, reproducible kinetic data, offering high sensitivity and detailed mechanistic insights. Conversely, BLI's dip-and-read methodology provides a robust, flexible, and high-throughput platform ideal for rapid screening and applications involving crude samples. The choice between them is not binary but should be guided by the specific stage of the research pipeline, the required data quality, and the nature of the samples. By understanding their core principles, performance characteristics, and ideal applications, researchers and drug development professionals can strategically deploy these technologies to accelerate biologic discovery and development.
Surface Plasmon Resonance (SPR) technology has established itself as a cornerstone of label-free detection in modern bioanalytical research. Its capacity to provide real-time, quantitative data on biomolecular interactions without the need for labels has revolutionized workflows in drug discovery, diagnostics, and basic science. This whitepaper provides a technical guide to the core comparative advantages of SPR, with a focused analysis of its sensitivity, throughput, and unique suitability for complex samples. By synthesizing recent advancements and current market trends, this document aims to equip researchers and drug development professionals with a comprehensive understanding of how SPR continues to address critical challenges in biomolecular interaction analysis.
Unmatched Sensitivity in Label-Free Detection
The exceptional sensitivity of SPR stems from its fundamental physical principle: the measurement of minute changes in the refractive index (RI) at a metal-dielectric interface. When a binding event occurs on the sensor surface, it alters the local RI, resulting in a measurable shift in the resonance angle or wavelength [1]. The sensitivity is so profound that SPR can routinely detect interactions with limits of detection (LOD) in the picomolar (pM) to nanomolar (nM) range for a wide array of analytes [1].
The origin of this high sensitivity is the evanescent field, an electromagnetic field generated at the sensor surface that decays exponentially with distance, typically extending 200-300 nm into the solution [1] [6]. This field interacts specifically with molecules within this narrow zone, making the technology exquisitely sensitive to surface binding events while being largely immune to bulk solution effects. This allows for the detection of subtle interactions, including those involving low-molecular-weight compounds, which are often challenging for other techniques.
Recent innovations have further pushed the boundaries of sensitivity. For instance, the development of fiber end-facet SPR biosensors featuring quasi-3D plasmonic crystal structures has demonstrated a refractive index detection limit on the order of 10⁻⁷ RIU (Refractive Index Units), enabling the potential detection of biomolecules down to the ng/mL range in bulk solution [86]. Similarly, the integration of metasurfaces (MetaSPR)—periodic nanoarrays that facilitate direct light-plasmon coupling—has created enhanced sensing platforms with high sensitivity to local RI changes [4].
Table 1: Key Sensitivity Metrics of SPR and Related Technologies
| Technology | Typical Detection Limit | Key Strengths | Key Limitations |
|---|---|---|---|
| SPR (Traditional) | pM - nM range [1] | Real-time, label-free kinetics; High sensitivity | Bulk instrument size; Requires skilled operation |
| SPR (Fiber-Optic) | ~10⁻⁷ RIU; ng/mL [86] | Compact, "dip-and-read" capability; Simplified fluidics | Lower Q-factor than some prism-coupled systems |
| Bio-Layer Interferometry (BLI) | Varies by application | Fiber-based, simple operation; Lower cost | Generally lower Q-factor and sensitivity compared to SPR [86] |
| MetaSPR | High sensitivity to local RI [4] | High-throughput; Label-free; Real-time cell monitoring | Emerging technology, less established protocols |
High-Throughput Analysis for Accelerated Discovery
The paradigm of SPR has shifted from a low-throughput, characterization-focused tool to a powerful platform for high-throughput screening (HTS). This transformation is critical for applications like therapeutic antibody discovery, where characterizing the kinetic parameters and epitope diversity of thousands of candidates is necessary [87].
Modern high-throughput SPR systems achieve this through several key technological advancements:
- SPR Imaging (SPRi): Instead of monitoring a single spot, SPRI uses a CCD camera to visualize binding events across a full biochip prepared in an array format. This allows for the simultaneous analysis of hundreds or thousands of interactions in real-time [6].
- Multiplexed Microfluidics: Proprietary fluidic systems, such as Hydrodynamic Isolation (HI) technology, enable the presentation of discrete fluid volumes to individually addressable locations on a 2D sensor surface without the need for physical valves. This allows for the creation of high-density arrays for multiplexed analysis, drastically increasing throughput and reducing reagent consumption [88].
- Automated Array Processing: Integrated systems combine automated liquid handling with SPRI to run parallel investigations into kinetics, affinities, and epitope specificities from the very start of the drug discovery process [87].
The impact on drug discovery is profound. High-throughput SPR enables researchers to empirically screen large antibody libraries early in the discovery process, fully appreciating the epitope landscape and identifying superior leads with reduced risk of failure and minimized sample consumption [87]. This "front-loading" of information changes the traditional screening paradigm from a sequential, triage-based process to a comprehensive, parallel characterization workflow.
Suitability for Complex and Challenging Samples
A defining advantage of SPR is its ability to analyze interactions in complex biological matrices and with challenging targets, most notably G protein-coupled receptors (GPCRs) and live cells, with minimal sample preparation.
Analysis of Membrane Proteins (GPCRs)
GPCRs are a prime class of drug targets but are notoriously difficult to study due to their instability outside their native membrane environment. SPR has been successfully adapted to handle these challenging proteins through innovative immobilization strategies that maintain receptor stability and function [89]. The main approaches include:
- Native Environment Immobilization: Immobilizing whole cells or membrane fragments directly onto the sensor chip, preserving the receptor's lipid environment [89].
- Membrane Mimetics: Using liposomes, lipoparticles, nanodiscs, or lentiviral particles to embed the GPCR in a structure that mimics the natural cell membrane before immobilization [89].
- Stabilized Receptor Immobilization: Isolating and stabilizing the receptor using detergents or protein engineering (e.g., introducing stabilizing mutations) before covalent coupling to the chip surface [89].
These strategies allow researchers to perform real-time, label-free analysis of drug binding to GPCRs, providing invaluable kinetic and affinity data (kon and koff) that are crucial for lead optimization in pharmaceutical development [89].
Real-Time, Label-Free Cellular Analysis
SPR's application has been extended to monitor entire cellular processes in real-time. The MetaSPR platform exemplifies this, functioning as a high-throughput, label-free, and long-term cell analysis platform [4]. It can be used to monitor:
- Cell proliferation and confluence directly on the sensor surface.
- Drug-target cell interactions and subsequent responses, such as apoptosis.
- Cell adhesion and spreading kinetics.
- Immune cell-mediated cytolysis (e.g., Natural Killer cell activity against cancer cells) [4].
In these assays, cells are cultured directly on the MetaSPR chip, which is integrated into a 96-well plate format. Cellular responses—such as attachment, detachment, or changes in morphology—alter the local refractive index, producing a quantifiable signal (Response Units, RU) that is tracked over time. This provides a dynamic view of cellular behavior that is not possible with endpoint assays like ELISA or flow cytometry [4].
Table 2: SPR Configurations for Complex Sample Analysis
| Sample Type | SPR Configuration / Strategy | Key Application | Experimental Consideration |
|---|---|---|---|
| GPCRs & Membrane Proteins | Immobilization via membrane mimetics (nanodiscs, liposomes) [89] | Drug discovery for GPCR targets | Maintaining receptor stability and native conformation on sensor chip |
| Live Cells | MetaSPR platform in 96-well format [4] | Real-time monitoring of cell signaling, drug efficacy, cytotoxicity | Cells must be adherent; signal reflects overall mass change |
| Secretome / Biofluids | Traditional SPR with specific capture ligands (e.g., antibodies, aptamers) [90] | Quantification of biomarkers (e.g., HGF, VEGF in MSC secretome) [90] | Surface functionalization to minimize non-specific binding |
| Small Molecules | High-sensitivity SPR (SPR+, Sierrra SPR Pro) [88] | Fragment-based drug discovery | Low molecular weight requires ultra-sensitive detection systems |
Experimental Protocols for Key Applications
Protocol: Kinetic Analysis of an Antibody-Antigen Interaction
This protocol outlines the steps for characterizing the binding kinetics of a monoclonal antibody to its soluble antigen using a traditional SPR instrument.
- Sensor Chip Functionalization: A CM5 (carboxymethylated dextran) sensor chip is activated using a standard amine-coupling kit (e.g., EDC/NHS chemistry). The antigen is diluted into a suitable sodium acetate buffer (pH 4.0-5.0) and injected over the activated surface to achieve a desired immobilization level (typically 50-100 Response Units). Remaining activated groups are deactivated with ethanolamine [88].
- System Equilibration: The running buffer (e.g., HBS-EP) is flowed continuously over the sensor surface at a constant temperature (e.g., 25°C) until a stable baseline is achieved.
- Binding Kinetics Measurement:
- Association Phase: A series of antibody solutions at different concentrations (e.g., 0, 3.125, 6.25, 12.5, 25 nM) are injected sequentially over the antigen surface and a reference surface for 2-5 minutes. The binding is monitored in real-time as an increase in RU.
- Dissociation Phase: The flow is switched back to running buffer, and the dissociation of the bound antibody is monitored for 10-30 minutes.
- Data Analysis: The resulting sensorgrams for each concentration are processed (reference and buffer subtracted). The data set is fitted to a 1:1 Langmuir binding model using the instrument's software to calculate the association rate (kₐ or kon), dissociation rate (kd or koff), and the equilibrium dissociation constant (KD = k_d/kₐ).
Protocol: Real-Time Monitoring of NK Cell-Mediated Cytotoxicity Using MetaSPR
This protocol describes how to use a MetaSPR platform to assess the cytolytic activity of immune cells [4].
- Sensor Plate Preparation: A 96-well MetaSPR biosensor plate is coated with a suitable extracellular matrix protein (e.g., poly-L-lysine) to promote cell adhesion.
- Target Cell Seeding: Adherent target cells (e.g., a cancer cell line) are seeded into the wells and allowed to attach and proliferate until confluence is reached, as indicated by a stabilization of the MetaSPR signal (RU values).
- Effector Cell Introduction: Natural Killer (NK) cells are added to the wells containing the target cells at varying Effector:Target (E:T) ratios. Wells containing only target cells serve as a negative control.
- Real-Time Monitoring: The MetaSPR platform, housed inside a cell culture incubator, continuously monitors the RU values for up to several days. The lysis of target cells by the NK cells causes them to detach from the sensor surface, resulting in a quantifiable decrease in the RU signal.
- Data Quantification: The rate and extent of RU decrease are calculated and correlated with the cytolytic activity. The results are validated against traditional methods like flow cytometry or CCK-8 assays [4].
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for SPR Biosensing
| Reagent / Material | Function | Example Use Case |
|---|---|---|
| Gold Sensor Chips | Provides the thin metal film required to generate the plasmon resonance. The foundation for all surface chemistry. | Standard kinetic and affinity measurements [1]. |
| Carboxymethylated Dextran (CM5) Chip | A hydrogel matrix that increases surface area for ligand immobilization and reduces non-specific binding. | Amine coupling of proteins, standard for antibody-antigen studies [88]. |
| Amine Coupling Kit (EDC/NHS) | Enables covalent immobilization of proteins, peptides, or other biomolecules containing primary amines to the sensor chip surface. | Immobilizing a recombinant protein antigen for antibody screening [88]. |
| Lipid Nanodiscs / Liposomes | Membrane mimetics used to solubilize and stabilize integral membrane proteins like GPCRs while maintaining a native-like lipid environment. | Studying the binding of small molecule drugs to a GPCR target [89]. |
| High-Throughput SPR (SPRi) Instrument | Instrumentation capable of SPR imaging, automated fluidics, and analysis of array-based sensor chips. | Simultaneous epitope binning of hundreds of antibody clones [87] [6]. |
| MetaSPR 96-Well Biosensor Plate | A high-throughput plate integrating metasurface chips for label-free cellular analysis. | Real-time, long-term monitoring of cell proliferation or drug-induced cytotoxicity [4]. |
Surface Plasmon Resonance technology has evolved far beyond its origins as a specialized tool for biophysicists. Its label-free nature, exceptional and ever-improving sensitivity, and transformative high-throughput capabilities make it an indispensable technology for researchers and drug developers. Furthermore, its adaptability to study complex biological systems—from unstable membrane proteins like GPCRs to dynamic live-cell interactions—solidifies its role as a versatile and powerful platform. As SPR technology continues to advance through miniaturization, integration with other analytical techniques, and the development of novel sensor surfaces, its impact on accelerating scientific discovery and therapeutic development is poised to grow even further.
In the field of biomolecular research, label-free detection technologies have become indispensable tools for studying interactions in their natural state, without the potential artifacts introduced by fluorescent or radioactive labels. Among these, Surface Plasmon Resonance (SPR) and Bio-Layer Interferometry (BLI) have emerged as two leading platforms that provide real-time, quantitative data on binding events [78] [77]. The convergence of these technologies offers researchers a powerful framework for orthogonal verification—the practice of validating experimental results using two or more independent methods. This approach is particularly crucial in drug development, where decisions involving billions of dollars in investment rely on accurate characterization of therapeutic candidates [77] [91].
The global label-free detection market, valued at approximately $2.5 billion in 2024 and projected to reach $5.1 billion by 2033, reflects the growing importance of these technologies in pharmaceutical and biotechnology industries [92]. This growth is driven by increasing demands for real-time analysis, rising pharmaceutical R&D expenditure, and the shift toward personalized medicine [92] [52]. Within this expanding landscape, understanding how to strategically integrate SPR and BLI provides researchers with a competitive advantage in generating reliable, reproducible data for critical decision-making.
Fundamental Principles of SPR and BLI
Surface Plasmon Resonance (SPR) Technology
SPR technology is based on an optical phenomenon that occurs when polarized light strikes a thin metal film, typically gold, at the interface between two media with different refractive indices [78] [81]. Under specific conditions, photons from the incident light couple with electron oscillations in the metal film, creating surface plasmons that absorb the light energy. This results in a measurable drop in the reflected light intensity at a specific angle known as the resonance angle [81].
The core components of an SPR system include:
- A sensor chip with a thin gold film
- A microfluidic flow channel system
- An optical detection unit with a polarized light source
- A prism to direct light to the sensor surface [78] [81]
When biomolecular binding occurs on the sensor surface, the increased mass changes the local refractive index, shifting the resonance angle. This shift is measured in real-time and plotted as a sensorgram, providing detailed information about binding kinetics, including association rates (kₐ), dissociation rates (kḍ), and equilibrium binding constants (Kᴅ) [78]. Commercial SPR platforms like Biacore (Cytiva) have become industry standards for precise kinetic characterization in drug discovery and basic research [78].
Bio-Layer Interferometry (BLI) Technology
BLI operates on a different principle based on white light interferometry. The system employs fiber-optic biosensors with a proprietary coating that reflects white light [77]. When light is directed down the sensor, it reflects from two surfaces: an internal reference layer and the sensor tip where biomolecular binding occurs. The interference pattern between these two reflected light beams creates a wavelength shift that is measured in real-time [78] [77].
Key components of BLI systems include:
- Disposable fiber-optic biosensors
- A 96- or 384-well microplate platform
- A white light source and spectrometer
- No requirement for complex microfluidics [77]
As molecules bind to the sensor tip, the increase in optical thickness causes a shift in the interference pattern, which is measured as a response in nanometers. This signal is proportional to the number of molecules bound to the sensor surface [78] [77]. The "dip-and-read" operation of BLI systems, such as the Octet series (Sartorius), offers simplicity and higher throughput compared to flow-based systems [77].
Table 1: Core Technological Principles of SPR and BLI
| Feature | Surface Plasmon Resonance (SPR) | Bio-Layer Interferometry (BLI) |
|---|---|---|
| Fundamental Principle | Measures refractive index changes via resonance angle shift on a gold film [81] | Measures thickness changes of biomolecular layers via interference pattern shifts [81] |
| Core Components | Gold-coated sensor, microfluidic system, optical prism [78] [81] | Fiber-optic biosensor, no fluidics required [78] [77] |
| Signal Detection | Shift in resonance angle (resonance units) [78] | Shift in interference pattern (nanometers) [78] |
| Measurement Output | Binding/dissociation rates, affinity constants [78] | Binding levels, affinity constants [78] |
| Immobilization Requirement | One binding partner immobilized on sensor chip [78] | One binding partner immobilized on sensor tip [78] |
Diagram 1: Fundamental principles and workflow comparison of SPR and BLI technologies
Comparative Analysis: Strategic Advantages and Limitations
Performance Characteristics and Applications
The strategic integration of SPR and BLI begins with understanding their complementary strengths and limitations. While both technologies provide label-free, real-time interaction data, they differ significantly in their operational parameters, sensitivity, and optimal application scenarios [77] [81].
SPR systems excel in applications requiring high sensitivity and precise kinetic characterization. The continuous flow system minimizes mass transport limitations and provides superior data quality for determining kinetic parameters [78] [81]. This makes SPR particularly valuable for characterizing small molecule interactions, fragment-based drug discovery, and detailed mechanistic studies where accurate kinetic rate constants are essential [77] [38]. Recent advancements in SPR technology, such as the Sartorius Octet SF3 with OneStep injection capabilities, have further enhanced its precision in measuring binding kinetics and affinity from single analyte concentrations [77].
BLI technology offers distinct advantages in throughput, operational simplicity, and flexibility. The dip-and-read approach eliminates complex microfluidics, reducing maintenance requirements and operational complexity [77]. This makes BLI ideal for high-throughput applications such as hybridoma screening, clone selection, and rapid protein quantification [77] [81]. BLI systems can analyze crude samples, including cell cultures and lysates, without purification, enabling researchers to study interactions under near-native conditions [77]. The recent introduction of the Octet R8e system with "near-SPR sensitivity" has further narrowed the performance gap between these platforms, particularly for small molecule and weak interaction studies [93].
Practical Implementation Considerations
From an implementation perspective, SPR and BLI present different requirements in terms of cost, expertise, and infrastructure. SPR systems typically represent a higher initial investment, with advanced instruments often exceeding $200,000, coupled with the need for specialized technical expertise for operation and data interpretation [92] [81]. The microfluidic components require careful maintenance and are susceptible to clogging or bubble formation, potentially impacting experimental workflow [81].
BLI systems generally have a lower entry cost, simpler operation, and minimal maintenance requirements due to the absence of complex fluidics [81]. This accessibility makes BLI particularly attractive for core facilities, academic laboratories, and industrial settings where multiple users require training on the platform. However, BLI has historically demonstrated lower sensitivity for low-concentration samples and less precise kinetic data compared to SPR, though recent technological improvements have addressed some of these limitations [93] [81].
Table 2: Comparative Performance Analysis of SPR and BLI
| Parameter | SPR | BLI | Orthogonal Advantage |
|---|---|---|---|
| Sensitivity | High (detects low-concentration samples) [81] | Moderate (improving with new systems) [93] [81] | SPR for low-abundance targets; BLI for concentrated samples |
| Throughput | Moderate (depends on flow channels) [81] | High (supports 96/384-well plates) [77] [81] | BLI for primary screening; SPR for detailed follow-up |
| Sample Consumption | Higher volume requirements [81] | Lower volume requirements (as low as 50-200 pmol) [94] | BLI for precious samples; SPR when volume is not limiting |
| Kinetic Data Quality | Excellent (precise kₐ, kḍ determination) [78] [81] | Good (improved with latest platforms) [93] | SPR for definitive kinetic characterization |
| Operational Complexity | High (requires fluidics expertise) [81] | Low (dip-and-read operation) [77] [81] | BLI for routine analysis; SPR for specialized applications |
| Sample Compatibility | Purified samples recommended [77] | Crude samples (lysates, supernatants) [77] | BLI for native conditions; SPR for controlled environments |
Orthogonal Verification: Methodologies and Experimental Design
Framework for Complementary Implementation
Orthogonal verification using SPR and BLI follows a strategic framework that leverages the unique strengths of each technology at different stages of the research workflow. A well-designed orthogonal approach begins with understanding the specific questions to be addressed and selecting the appropriate sequence of technologies to answer them comprehensively [77] [91].
A typical integrated workflow might include:
- Primary Screening: BLI is ideal for initial screening phases due to its high-throughput capabilities and ability to handle multiple samples simultaneously in 96- or 384-well formats [77]. This allows rapid identification of potential hits from large libraries with minimal sample preparation.
Hit Validation: Confirmed hits from BLI screening can be further validated using SPR for more precise kinetic characterization [77]. The flow-based system of SPR provides superior data quality for determining binding mechanisms and eliminating false positives.
Detailed Kinetic Analysis: SPR delivers comprehensive kinetic profiling with high sensitivity, making it suitable for characterizing weak binders, small molecules, and complex interaction mechanisms [81] [38].
Secondary Assays and Counter-Screening: BLI can be employed for subsequent assays, including epitope binning, concentration measurements, and specificity profiling against related targets [77].
This sequential approach was validated in a comprehensive study comparing multiple label-free systems, which analyzed 1,378 data points and concluded that "binding constants cannot be determined as easily as other constants" and strongly recommended "using at least two different systems in parallel to achieve meaningful results" [91].
Experimental Protocol: Fragment Screening Against RNA Targets
A recent study demonstrates the power of combining BLI with orthogonal methods for fragment-based drug discovery against RNA targets [94]. The researchers employed BLI to identify fragment binders of the flavin mononucleotide (FMN) riboswitch, followed by NMR spectroscopy for validation. The detailed protocol included:
BLI Experimental Conditions:
- Biosensor: Super Streptavidin (SSA) sensors
- RNA Preparation: Riboswitches biotinylated at 3' end, refolded in 10 mM MgCl₂
- Immobilization: Optimal loading achieved with 10 mM MgCl₂ during folding
- Binding Assay: Performed in presence of 2 mM MgCl₂
- Fragment Screening: Library screened against FMN and TPP riboswitches
- Dose-Response: Primary hits confirmed through concentration series
- Data Analysis: Steady-state and kinetic fit models applied [94]
Key Findings:
- Mg²⁺ concentration critically affected both RNA immobilization and ligand binding
- Highest immobilization achieved with 10 mM MgCl₂ during folding
- Optimal binding responses observed with 2 mM MgCl₂ in assay buffer
- Seven novel competitive FMN riboswitch binders identified with affinities ranging from 14-500 μM [94]
This study highlights how BLI serves as an effective primary screening method, with subsequent orthogonal validation confirming binding mechanisms and specificity.
Diagram 2: Integrated workflow for orthogonal verification combining BLI and SPR technologies
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of orthogonal verification strategies requires careful selection of reagents and materials optimized for each technology platform. The following table outlines key solutions and their applications in SPR and BLI experiments.
Table 3: Research Reagent Solutions for SPR and BLI Experiments
| Reagent/Material | Function | SPR Application | BLI Application |
|---|---|---|---|
| Sensor Chips | Provides immobilization surface | CM3, CM4, CM5 chips with carboxymethyl dextran [78] | Super Streptavidin (SSA), Anti-His, Ni-NTA biosensors [77] [94] |
| Immobilization Chemistry | Covalent attachment of ligands | Amine coupling, thiol coupling, surface capture [78] | Streptavidin-biotin, His-tag capture, antibody-antigen [77] [94] |
| Running Buffer | Maintains physiological conditions | HBS-EP, PBS with surfactants [78] | PBS with Mg²⁺ for RNA interactions [94] |
| Regeneration Solutions | Removes bound analyte | Glycine-HCl (pH 1.5-3.0), NaOH, SDS [78] | Similar solutions with optimization for sensor integrity [78] |
| Quality Control Analytes | System performance verification | Concentration series of known interactors [91] | Certified reference materials for calibration [91] |
Emerging Trends and Future Directions
The field of label-free detection continues to evolve with technological advancements that further enhance the complementary relationship between SPR and BLI. Several emerging trends are particularly noteworthy:
Integration of Artificial Intelligence and Machine Learning: AI and ML algorithms are being incorporated into both SPR and BLI platforms to enhance analysis precision, reduce noise, and improve binding affinity predictions [92]. These technologies assist in interpreting complex datasets and identifying subtle interaction patterns that might be missed by conventional analysis methods [92] [38].
Automation and Miniaturization: Increasing automation through robotic liquid handling and integration with microfluidics is accelerating throughput while reducing sample requirements [92]. The development of portable SPR and BLI systems enables field applications and point-of-care testing, expanding the potential applications of these technologies [92].
Enhanced Sensitivity and Performance: Recent introductions of BLI systems with "near-SPR sensitivity," such as the Octet R8e, are blurring the historical performance boundaries between these platforms [93]. This convergence enables researchers to obtain higher quality data from both technologies, strengthening orthogonal verification outcomes.
Sustainability Focus: The elimination of dyes and reagents in label-free detection aligns with growing emphasis on sustainable laboratory practices [92]. This environmental consideration is becoming an additional factor in technology selection for many research institutions and pharmaceutical companies.
These advancements collectively support more robust orthogonal verification strategies, enabling researchers to address increasingly complex biological questions with greater confidence in their results.
Orthogonal verification through the strategic integration of SPR and BLI represents a powerful paradigm for robust biomolecular characterization in modern drug discovery and basic research. By leveraging the complementary strengths of these technologies—SPR's superior sensitivity and kinetic resolution with BLI's throughput and operational simplicity—researchers can generate more reliable, reproducible data for critical decision-making [77] [91].
As the label-free detection market continues to expand at a considerable pace [92] [52] [38], the value of orthogonal approaches becomes increasingly apparent. The convergence of these technologies, coupled with emerging advancements in AI integration, automation, and sensitivity, promises to further enhance their complementary relationship. For researchers seeking to maximize the impact of their biomolecular interaction studies, embracing this integrated framework provides a pathway to more definitive conclusions and accelerated scientific progress.
Positioning SPR Among Other Label-Free and Label-Based Techniques (e.g., ELISA, ITC, QCM)
Surface Plasmon Resonance (SPR) has established itself as a cornerstone technology in the landscape of label-free detection methods. As a phenomenon that occurs when electrons in a thin metal sheet become excited by light directed at a specific angle of incidence, SPR enables real-time, quantitative analysis of biomolecular interactions without the need for fluorescent tags or other labeling strategies [95]. The fundamental principle underpinning SPR biosensing is the sensitivity of surface plasmon polaritons to changes in the refractive index at the interface between a metal sensor surface and the sample medium, allowing researchers to monitor binding events as they happen [6] [95]. This capability has positioned SPR as an indispensable tool across numerous fields, including drug discovery, clinical diagnostics, and fundamental biomedical research, where understanding the intricacies of molecular interactions is paramount.
The evolution of SPR from a specialized laboratory technique to a mainstream analytical platform reflects a broader shift in the life sciences toward label-free methodologies that preserve the native state of biomolecules. Traditional label-based techniques, while powerful, introduce potential artifacts through the labeling process itself—modifications that may alter binding affinities, obscure active sites, or interfere with natural conformational dynamics [2]. In contrast, label-free strategies like SPR eliminate these concerns by relying on inherent molecular properties, providing a more physiologically relevant window into biomolecular behavior [28] [96]. The technological advancements in SPR instrumentation, including the development of SPR imaging (SPRI) for high-throughput applications and multi-parametric SPR for detailed structural characterization, have further expanded its utility in modern bioscience [6] [95].
Fundamental Principles of SPR Technology
Physical Mechanism and Instrumentation
The operational principle of Surface Plasmon Resonance hinges on an optical phenomenon that occurs under specific conditions at a metal-dielectric interface. When polarized light strikes a thin metal film (typically gold or silver) under total internal reflection conditions at an appropriate angle, it can excite collective oscillations of free electrons at the metal surface, known as surface plasmons [6] [95]. This excitation results in a measurable drop in the intensity of reflected light at a specific angle—the resonance angle—that is exquisitely sensitive to changes in the refractive index within approximately 300 nanometers of the metal surface [6]. The Kretschmann configuration is the most commonly implemented optical arrangement in commercial SPR instruments, where light is directed through a prism onto a thin metal film deposited on the prism surface, and the reflected light is detected by a position-sensitive detector or CCD camera [6] [95].
In practical biosensing applications, one binding partner (the ligand) is immobilized on the sensor surface, while the other (the analyte) is introduced in solution flowing over the surface. When molecular binding occurs, the accumulated mass on the sensor surface alters the local refractive index, leading to a shift in the resonance angle that is directly proportional to the mass concentration of bound analyte [6]. This response is measured in resonance units (RU), where 1 RU typically corresponds to a shift of 10⁻⁴ degrees, approximately equivalent to 1 pg/mm² of protein binding [6]. This direct relationship between binding mass and signal output enables SPR to provide quantitative data on binding affinity, kinetics, and concentration without requiring external labels or probes.
Experimental Workflow for SPR Analysis
The standard SPR experiment follows a systematic workflow designed to yield robust, interpretable data on molecular interactions. The process begins with surface preparation, where the sensor chip is functionalized to enable covalent immobilization of the ligand molecule through various chemistries (e.g., amine coupling, thiol coupling, or capture methodologies) [95]. This immobilization step is critical, as it must preserve the biological activity of the ligand while providing a stable surface for binding interactions. Once prepared, the surface is brought into contact with a continuous flow of buffer to establish a stable baseline signal—the reference point against which all binding events are measured [6].
The analytical phase consists of sequentially introducing the analyte solutions while continuously monitoring the SPR signal in real-time. Each injection cycle typically includes an association phase (where analyte flows over the surface and binding occurs), followed by a dissociation phase (where pure buffer flows over the surface and bound complexes dissociate) [6] [95]. The resulting sensorgram—a plot of response units versus time—provides a rich dataset from which binding parameters can be extracted. Through iterative analysis of binding responses at different analyte concentrations and global fitting of the combined data, researchers can determine the association rate (kₐ), dissociation rate (kḍ), and equilibrium dissociation constant (K_D = kḍ/kₐ) that define the interaction [97] [95]. The sensor surface can typically be regenerated by applying conditions that remove bound analyte without damaging the immobilized ligand, allowing multiple analysis cycles on the same surface.
Comparative Analysis: SPR Versus Label-Based Techniques
SPR vs. ELISA: A Paradigm Shift in Binding Analysis
The enzyme-linked immunosorbent assay (ELISA) represents one of the most established and widely used label-based techniques in biomedical research and clinical diagnostics. As a plate-based assay, ELISA relies on the specific binding between an antigen and an antibody, with detection enabled by an enzyme-linked secondary antibody that produces a measurable colorimetric, chemiluminescent, or fluorescent signal [98]. While ELISA excels at providing sensitive, quantitative endpoint measurements of biomolecule concentrations, it falls short in its ability to elucidate the dynamic aspects of molecular interactions. The requirement for multiple labeling, incubation, and washing steps not only makes ELISA labor-intensive and time-consuming (often requiring more than a day to complete) but also introduces potential artifacts through chemical modification of the molecules of interest [98].
SPR fundamentally outperforms ELISA in several critical aspects, most notably in its capacity to provide real-time kinetic information. Unlike ELISA, which provides a single endpoint measurement, SPR monitors binding events as they occur, enabling researchers to determine both association and dissociation rate constants in addition to equilibrium binding affinity [98] [97]. This capability is particularly valuable for characterizing weak or transient interactions that might be missed during ELISA's washing steps, where rapidly dissociating complexes are lost before detection [98]. Comparative studies have demonstrated that SPR can detect anti-drug antibodies with significantly higher sensitivity than ELISA, identifying up to 7-490 times higher antibody levels and detecting positives in samples classified as negative by ELISA [97]. Furthermore, SPR eliminates the need for labeled secondary reagents, streamlining assay development and reducing the potential for cross-reactivity or steric hindrance that can compromise ELISA results [98].
Limitations of ELISA in Affinity Measurement
A critical limitation of ELISA in binding characterization is its inability to confirm when binding equilibrium has been reached—a fundamental requirement for accurate affinity determination. Research has revealed that approximately 70% of published studies using ELISA fail to confirm equilibrium, often using incubation times of one hour or less despite evidence that full equilibration for many protein complexes can require several hours [97]. This methodological shortcoming leads to systematic underestimation of binding affinity, as demonstrated in a comparative study of alpaca antibodies where ELISA-reported KD values were 14.1 to 43.7-fold higher (indicating lower affinity) than those determined by SPR [97]. The time to equilibrium (tequil), which can only be reliably determined through kinetic analysis using SPR, must guide ELISA incubation times to generate accurate affinity measurements—a consideration frequently overlooked in practice.
Comparative Analysis: SPR Versus Other Label-Free Techniques
The landscape of label-free detection technologies extends beyond SPR to include several complementary platforms, each with distinct physical principles and application strengths. Bio-Layer Interferometry (BLI) shares SPR's ability to measure binding kinetics and affinity but employs a different optical principle based on white light interference patterns generated at the tip of fiber-optic biosensors [99]. Isothermal Titration Calorimetry (ITC) uniquely provides comprehensive thermodynamic profiling by directly measuring the heat absorbed or released during binding interactions, though it cannot determine binding kinetics [99]. Quartz Crystal Microbalance (QCM) detects mass changes on a sensor surface through shifts in resonance frequency of a quartz crystal, offering sensitivity to structural changes in addition to bound mass [28] [96]. Other notable label-free techniques include Microscale Thermophoresis (MST), which monitors molecule movement in temperature gradients, and various interferometric methods that detect changes in optical path length [99].
The selection of an appropriate label-free technique depends heavily on the specific research question and the nature of the molecular system under investigation. SPR stands out for its well-established position in regulatory submissions, with acceptance by the FDA and EMA for characterizing therapeutic biologics—a endorsement not uniformly shared by other label-free methods [99]. The technology's robust fluidics systems, precise temperature control, and ability to analyze crude samples (including undiluted serum) further distinguish it in pharmaceutical applications where these attributes are essential [99]. While newer techniques like BLI offer advantages in ease of use and minimal fluidics maintenance, they may lack the temperature control and rigorous fluid handling necessary for certain demanding applications [98] [99].
Technical Comparison of Label-Free Methods
Table 1: Comparison of Key Label-Free Detection Technologies
| Technique | Measured Parameters | Sample Consumption | Throughput | Key Applications | Main Limitations |
|---|---|---|---|---|---|
| SPR | Affinity (K_D), kinetics (kₐ, kḍ), concentration, thermodynamics | Low (μL volumes) | Moderate to High | Drug discovery, antibody characterization, biomarker validation | Requires immobilization; high instrument cost |
| BLI | Affinity (K_D), kinetics (kₐ, kḍ), concentration | Low (μL volumes) | High | Rapid screening, antibody profiling, protein quantification | Limited temperature control; lower data quality |
| ITC | Thermodynamics (ΔG, ΔH, ΔS), binding stoichiometry, affinity (K_D) | High (mL volumes) | Low | Mechanism studies, fragment screening, enzyme kinetics | No kinetic data; low throughput; large sample requirement |
| QCM | Bound mass, viscoelastic properties, structural changes | Low to Moderate | Moderate | Cell adhesion, polymer films, complex biomolecular layers | Lower sensitivity for small molecules; mass not specific |
| MST | Affinity (K_D), binding stoichiometry, enzyme kinetics | Very Low | Moderate | Fragment screening, protein-small molecule interactions | Requires fluorescent labeling; no kinetic data |
SPR's versatility across a broad range of applications and analyte types positions it as the most comprehensive solution for biomolecular interaction analysis. The technology supports the characterization of diverse molecular interactions—including protein-protein, protein-small molecule, antibody-antigen, and nucleic acid interactions—with a dynamic affinity range spanning picomolar to millimolar dissociation constants [99]. This breadth of applicability, combined with its status as the regulatory gold standard for binding assays, makes SPR particularly valuable in biopharmaceutical development where data quality and regulatory compliance are paramount [99]. While techniques like ITC provide more detailed thermodynamic profiles and BLI offers advantages in throughput and ease of use, SPR remains unique in delivering comprehensive kinetic and affinity data with the precision and reproducibility required for critical decision-making in drug development.
Essential Reagents and Materials for SPR Analysis
Core Components of SPR Experimental Systems
Successful SPR analysis requires careful selection of reagents and materials optimized for the specific biological system under investigation. The foundation of any SPR experiment is the sensor chip, which provides the gold surface functionalized with specific chemical groups to facilitate ligand immobilization. The most common sensor surfaces include carboxymethylated dextran (CM5) for amine coupling, nitrilotriacetic acid (NTA) for His-tagged protein capture, streptavidin-coated surfaces for biotinylated molecules, and hydrophobic association (HPA) chips for membrane protein studies [6] [95]. Each surface chemistry offers distinct advantages for different biomolecule types and must be selected based on the ligand properties and the desired orientation after immobilization.
The running buffer used throughout the SPR experiment serves as both the solvent for analyte dilution and the continuous flow medium, making its composition critical for maintaining biological activity and minimizing non-specific binding. Optimal running buffers typically include physiological salt concentrations (e.g., HEPES-buffered saline, PBS) with added non-ionic detergent (such as Tween-20) to reduce surface adsorption, and sometimes carrier proteins like BSA to stabilize low-concentration analytes [6]. Regeneration solutions—carefully selected to disrupt the specific interaction without damaging the immobilized ligand—complete the core reagent set. Common regeneration agents include mild acids (glycine-HCl, pH 1.5-3.0), bases (NaOH, up to 100 mM), high salt solutions (1-4 M MgCl₂ or NaCl), or chaotropic agents (guanidine-HCl) [6] [95]. The development of a robust regeneration protocol is essential for reusable sensor surfaces and reproducible results across multiple binding cycles.
Research Reagent Solutions for SPR
Table 2: Essential Materials for SPR Experiments
| Reagent/Material | Function | Key Considerations | Common Examples |
|---|---|---|---|
| Sensor Chips | Provides functionalized surface for ligand immobilization | Surface chemistry must match ligand properties and coupling strategy | CM5 (dextran), NTA (His-tag capture), SA (streptavidin), HPA (lipid membranes) |
| Running Buffer | Maintains physiological conditions; minimizes non-specific binding | pH, ionic strength, and additives must preserve binding activity | HBS-EP (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% P20 surfactant) |
| Regeneration Solutions | Removes bound analyte without damaging immobilized ligand | Stringency must be optimized for each molecular interaction | Glycine-HCl (pH 1.5-3.0), NaOH (10-100 mM), MgCl₂ (1-4 M) |
| Immobilization Reagents | Activates surface for covalent coupling | Compatibility with ligand functional groups and stability | EDC/NHS for amine coupling, 2-(2-pyridinyldithio)ethaneamine for thiol coupling |
| Ligand Molecules | The immobilized binding partner | Purity and activity critical for meaningful results | Antibodies, receptors, antigens, nucleic acids, small molecules |
Applications and Future Directions in SPR Research
Current Applications Across Biotechnology and Medicine
SPR technology has found diverse applications spanning basic research, drug discovery, clinical diagnostics, and quality control in biomanufacturing. In pharmaceutical development, SPR is extensively used for antibody characterization, including affinity measurements, epitope binning, and kinetic profiling of candidate therapeutic molecules [37] [99]. The technology's ability to precisely quantify binding interactions makes it invaluable for biosimilar development, where demonstrating comparability to reference products requires sensitive detection of subtle differences in binding behavior [99]. Beyond antibody characterization, SPR supports fragment-based drug discovery by identifying weak binders that might be missed by endpoint assays, and facilitates mechanistic studies of enzyme inhibition by providing real-time data on inhibitor binding and residence time [6] [95].
In clinical applications, SPR-based immunoassays have been developed for biomarker detection, pathogen identification, and therapeutic drug monitoring [6] [96]. The label-free nature of SPR eliminates the need for multiple reagent incubations and washing steps, potentially reducing assay time and complexity compared to conventional ELISA [98] [97]. Recent advances in SPR imaging (SPRI) have further expanded these applications by enabling multiplexed detection of hundreds of interactions simultaneously, creating opportunities for high-throughput biomarker validation and comprehensive serological profiling [6] [96]. The technology's sensitivity to conformational changes has also been leveraged to study protein folding and structural transitions, providing insights into fundamental biological processes and disease mechanisms [6].
Emerging Trends and Technological Advancements
The future trajectory of SPR technology is being shaped by several convergent trends, including miniaturization, integration with complementary analytical techniques, and the incorporation of artificial intelligence for enhanced data analysis. Miniaturized fiber-optic SPR sensors are emerging for decentralized point-of-care testing, potentially expanding access to sophisticated binding analysis beyond central laboratories [37]. The integration of SPR with mass spectrometry creates powerful hybrid platforms that combine detailed interaction data with structural identification capabilities, while combinations with chromatography systems enable analysis of complex mixtures without pre-purification [99]. These integrated approaches are particularly valuable for characterizing heterogeneous samples and identifying novel binding partners in biological fluids.
Artificial intelligence and machine learning algorithms are increasingly being applied to SPR data analysis, enhancing the precision of kinetic parameter estimation and enabling the identification of subtle binding patterns that might escape manual interpretation [37] [92]. AI-assisted kinetic modeling has demonstrated potential to accelerate hit-to-lead timelines in drug discovery from the traditional 18-24 months to as little as 6-12 months, representing a significant efficiency gain for pharmaceutical R&D [37]. Concurrently, improvements in sensor design and surface chemistries continue to push the sensitivity boundaries of SPR, with recent developments achieving detection limits in the low picogram per milliliter range for protein analytes [6] [96]. These advances collectively ensure that SPR will maintain its position as a cornerstone analytical technique while expanding into new application areas in basic research, clinical diagnostics, and therapeutic development.
Conclusion
Surface Plasmon Resonance stands as a cornerstone technology in the label-free analysis landscape, offering unparalleled capabilities for real-time, kinetic profiling of biomolecular interactions. Its foundational principle of detecting refractive index changes provides a versatile platform applicable from basic research to clinical diagnostics. The continuous innovation in sensor design, material science, and data optimization, including algorithm-driven enhancements and novel hybrid materials, is consistently pushing the boundaries of sensitivity and specificity. These advancements, exemplified by developments in SPRi and PCF-SPR sensors, are paving the way for more precise and accessible applications in personalized medicine, ultra-early disease detection, and high-throughput drug screening. As the technology evolves and integrates with complementary techniques like BLI, SPR is poised to remain an indispensable tool, delivering critical insights that accelerate therapeutic development and deepen our understanding of biological mechanisms.
References
- 1. Surface Plasmon Resonance-Based Biodetection Systems [https://pmc.ncbi.nlm.nih.gov/articles/PMC11763797/]
- 2. Label-free single-molecule optical detection | npj Biosensing [https://www.nature.com/articles/s44328-025-00048-9]
- 3. Comparison of methods for quantitative biomolecular ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8748344/]
- 4. Metasurface plasmon resonance platform for high ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566325009285]
- 5. Plasmon-induced resonance energy transfer for solar ... [https://www.nature.com/articles/nphoton.2015.142]
- 6. Surface Plasmon Resonance: A Versatile Technique for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4481982/]
- 7. Surface Plasmon Resonance (SPR)-Based Biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4402104/]
- 8. Surface Plasmon Resonance (SPR) 101: Kretschmann ... [https://eureka.patsnap.com/article/surface-plasmon-resonance-spr-101-kretschmann-configuration-explained]
- 9. Experimental study of sensing performance using ... [https://www.sciencedirect.com/science/article/abs/pii/S0030399224009216]
- 10. Plasmonic Biosensing with Aluminum Thin Films under the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8371922/]
- 11. A Surface Plasmon Resonance-Based Integrated Assay for ... [https://pubmed.ncbi.nlm.nih.gov/40616443/]
- 12. Surface Plasmon Resonance: An Introduction to a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3045209/]
- 13. Surface Plasmon Resonance Biosensors: A Review of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10886473/]
- 14. SPR Theory [https://www.sprpages.nl/spr-overview/spr-theory]
- 15. Trends in SPR Cytometry: Advances in Label-Free ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6315638/]
- 16. An Introduction to Surface Plasmon Resonance [https://www.jacksonimmuno.com/secondary-antibody-resource/immuno-techniques/an-introduction-to-surface-plasmon-resonance/]
- 17. SPR Sensorgram Explained [https://www.affiniteinstruments.com/post/spr-sensorgram-explained]
- 18. Surface Plasmon Resonance Sensorgram: A Step-by-Step ... [https://www.iaanalysis.com/surface-plasmon-resonance-sensorgram-guide.html]
- 19. Use of Surface Plasmon Resonance (SPR) to Determine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8489108/]
- 20. Guide to SPR Data Analysis on the ProteOn™ XPR36 ... [https://www.bioradiations.com/guide-to-spr-data-analysis-on-the-proteon-xpr36-system/]
- 21. Exponential [https://www.sprpages.nl/sensorgrams/exponential]
- 22. A Curve [https://www.sprpages.nl/sensorgrams/a-curve]
- 23. Dissociation [https://www.sprpages.nl/kinetics/dissociation]
- 24. Biosensor-Surface Plasmon Resonance: Label Free ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6774244/]
- 25. Review Surface plasmon resonance biosensor chips [https://www.sciencedirect.com/science/article/abs/pii/S0731708525003590]
- 26. Enhancing Shiga toxin detection using surface plasmon ... [https://www.nature.com/articles/s41598-025-01686-9]
- 27. Advancements in surface plasmon resonance sensors for real ... [https://pubs.rsc.org/en/content/articlehtml/2025/tc/d4tc04890c]
- 28. Recent advances in label-free detection in vitro and ... [https://www.sciencedirect.com/science/article/abs/pii/S0010854525008975]
- 29. Nanoplasmonic Biosensors: A Comprehensive Overview ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12067471/]
- 30. Use of Surface Plasmon Resonance (SPR) to Determine Binding Affinities and Kinetic Parameters Between Components Important in Fusion Machinery - PubMed [https://pubmed.ncbi.nlm.nih.gov/30317506/]
- 31. Two common methods for kinetic analysis with SPR [https://nicoyalife.com/blog/analyzing-binding-kinetics-mck-sck/]
- 32. Kinetic models [https://www.sprpages.nl/data-fitting/kinetic-models]
- 33. Label-free localized surface plasmon resonance (LSPR) ... [https://www.sciencedirect.com/science/article/pii/S0039914024016175]
- 34. Overview of Surface Plasmon Resonance Technology [https://biosensingusa.com/technologies/]
- 35. Label-free detection with surface plasmon resonance ... [https://pubmed.ncbi.nlm.nih.gov/21370074/]
- 36. SPR Analysis - Surface Plasmon Resonance Services [https://covalx.com/services/spr-analysis/]
- 37. Label-free Detection (LFD) Market Size & Share Analysis [https://www.mordorintelligence.com/industry-reports/label-free-detection-lfd-market]
- 38. Label-Free Detection Market: Size, Share & Forecast 2025 [https://www.meticulousresearch.com/product/label-free-detection-market-3408]
- 39. SPR-based fragment screening: advantages and ... [https://pubmed.ncbi.nlm.nih.gov/17979772/]
- 40. Rapid optimisation of fragments and hits to lead ... [https://www.nature.com/articles/s42004-020-00367-0]
- 41. Experimental Validation of a Fragment Library for Lead ... [https://www.sciencedirect.com/science/article/pii/S247255522207736X]
- 42. Advancing cancer diagnosis through surface plasmon ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993625001669]
- 43. Surface plasmon resonance biosensors and their medical ... [https://www.sciencedirect.com/science/article/pii/S0956566325001824]
- 44. Advanced SPR biosensor with optimized gold-TiO2 D ... [https://www.nature.com/articles/s41598-025-24697-y]
- 45. Advanced SPR biosensor with optimized gold-TiO2 D ... [https://pubmed.ncbi.nlm.nih.gov/41266570/]
- 46. Plasmonic SPR biosensor with bayesian regression for ... [https://www.nature.com/articles/s41598-025-22949-5]
- 47. Real-Time Protease Biomarker Tracking With a Peptide ... [https://www.spectroscopyonline.com/view/real-time-protease-biomarker-tracking-with-a-peptide-spr-biosensor]
- 48. - High surface plasmon resonance throughput -based... imaging [https://pubs.rsc.org/en/content/articlehtml/2010/ay/c0ay00112k]
- 49. Surface Plasmon Resonance Multiplexed for... | SpringerLink Imaging [https://link.springer.com/protocol/10.1007/978-1-62703-134-9_30]
- 50. SPRi Peptide Analysis - Creative Peptides [https://www.creative-peptides.com/services/surface-plasmon-resonance-imaging-spri-service.html]
- 51. Fast spectral surface plasmon resonance imaging sensor for real-time... [https://pubmed.ncbi.nlm.nih.gov/27936268/]
- 52. Label-Free Detection Market Report | Global Forecast From ... [https://dataintelo.com/report/global-label-free-detection-market]
- 53. Algorithm assisted comprehensive optimization of SPR ... [https://www.sciencedirect.com/science/article/abs/pii/S0956566325007225]
- 54. Algorithm assisted comprehensive optimization of SPR ... [https://pubmed.ncbi.nlm.nih.gov/40812130]
- 55. Design optimization of high-sensitivity PCF-SPR biosensor ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0330944]
- 56. Design optimization of high-sensitivity PCF-SPR biosensor ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12435684/]
- 57. Label Free Detection Systems 2025 Trends and Forecasts ... [https://www.archivemarketresearch.com/reports/label-free-detection-systems-484151]
- 58. SPR-based PCF sensors for next generation research [https://www.sciencedirect.com/science/article/pii/S2211379725003031]
- 59. - Label Technology: Revolutionizing Biological... Free Detection [https://www.linkedin.com/pulse/label-free-detection-technology-revolutionizing-biological-vaidya-m-sikdf]
- 60. Video: Surface Plasmon Resonance SPR : Label - Free Detection [https://www.jove.com/v/5697/surface-plasmon-resonance-spr-label-free-detection]
- 61. 2D Nanomaterial-Based Surface Plasmon Resonance ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7463818/]
- 62. High performance SPR sensor using 2-D materials [https://www.sciencedirect.com/science/article/abs/pii/S0924424724001523]
- 63. TiO2-FG-based plasmonic sensor with enhanced figure of ... [https://www.sciencedirect.com/science/article/abs/pii/S0030402625002098]
- 64. Novel SPR sensing platform based on superstructure MoS ... [https://www.sciencedirect.com/science/article/abs/pii/S0925400519300048]
- 65. A novel Mxene-SPR-based sensor for sensing different types ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12364876/]
- 66. Sensitivity enhancement of SPR sensor assisted by Ag, TiO ... [https://link.springer.com/article/10.1007/s11082-023-05217-6]
- 67. Surface Plasmon Resonance Microscopy: From Single- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7020607/]
- 68. The Current Status and Future Promise of SPR Biosensors [https://pmc.ncbi.nlm.nih.gov/articles/PMC9687760/]
- 69. Highly sensitive and label-free detection of SARS-CoV-2 ... [https://www.nature.com/articles/s41598-025-16342-5]
- 70. New microscopy method offers 3D tracking of 100 single ... [https://news.asu.edu/20211119-new-microscopy-method-offers-3d-tracking-100-single-molecules-once]
- 71. Trends and Challenges of SPR Aptasensors in Viral ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12026110/]
- 72. Surface plasmon resonance technology: Recent advances ... [https://www.sciencedirect.com/science/article/pii/S0165993623001668]
- 73. Optimizing drug discovery: Surface plasmon resonance ... [https://www.sciencedirect.com/science/article/pii/S2667022423002530]
- 74. Surface plasmon resonance (SPR) biosensors in ... [https://pubmed.ncbi.nlm.nih.gov/25558771/]
- 75. How does Digital SPR fit into early drug discovery? [https://nicoyalife.com/blog/digital-spr-in-early-drug-discovery/]
- 76. Electrochemical surface plasmon resonance based ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12081753/]
- 77. BLI vs. SPR: Choosing the Ideal Method for Analyzing ... [https://www.sartorius.hr/en/news/blog/bli-vs-spr-choosing-the-ideal-method-for-analyzing-biomolecular-interactions/]
- 78. Applications of Surface Plasmon Resonance and Biolayer ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9027846/]
- 79. BLI vs SPR: Picking the Right Tool for Discovery ... [https://immunebiosolutions.com/en/bli-vs-spr-picking-the-right-tool-for-biotech-discovery/]
- 80. Biolayer interferometry and its applications in drug ... [https://www.sciencedirect.com/science/article/pii/S0165993624002243]
- 81. Surface Plasmon Resonance (SPR) vs Biolayer Interferometry ... [https://www.iaanalysis.com/surface-plasmon-resonance-biolayer-interferometry-choice.html]
- 82. Label Free Detection Industry: Emerging Trends and ... [https://www.marketsandmarkets.com/ResearchInsight/label-free-detection-industry.asp]
- 83. SPR vs BLI | Fc Receptor-IgG Characterization [https://nicoyalife.com/blog/alto-fc-receptors-spr-vs-bli/]
- 84. Biolayer Interferometry vs Surface Plasmon Resonance ... [https://nicoyalife.com/blog/biolayer-interferometry-vs-surface-plasmon-resonance/]
- 85. Label-Free Detection [https://www.sartorius.com/en/applications/life-science-research/label-free-detection]
- 86. Sensitive surface plasmon resonance label-free biosensor ... [https://www.nature.com/articles/s41377-022-01025-x]
- 87. High Throughput Surface Plasmon Resonance [https://carterra-bio.com/understanding-surface-plasmon-resonance/]
- 88. Surface Plasmon Resonance for high-throughput analysis [https://www.bruker.com/en/products-and-solutions/surface-plasmon-resonance.html]
- 89. Latest surface plasmon resonance advances for G protein- ... [https://www.sciencedirect.com/science/article/pii/S2095177925001984]
- 90. Emerging Applications and Challenges in SPR-Based ... [https://pubmed.ncbi.nlm.nih.gov/41054903/]
- 91. Comparison of different label-free imaging high-throughput ... [https://www.sciencedirect.com/science/article/abs/pii/S0003269719302106]
- 92. Label-Free Detection Market Report 2025: A Strategic ... [https://www.linkedin.com/pulse/label-free-detection-market-report-2025-strategic-8hjde/]
- 93. Near-SPR Performance Built for Real-World Biologics [https://www.news-medical.net/news/20250819/Redefining-BLI-Near-SPR-performance-built-for-real-world-biologics.aspx]
- 94. Discovery of RNA-binding fragments using biolayer ... [https://pubs.rsc.org/en/content/articlehtml/2025/md/d5md00673b]
- 95. Surface plasmon resonance [https://en.wikipedia.org/wiki/Surface_plasmon_resonance]
- 96. Label‐free detection techniques for protein microarrays [https://pmc.ncbi.nlm.nih.gov/articles/PMC7167936/]
- 97. SPR vs. ELISA: SPR Guides Accurate ELISA Results [https://www.rapidnovor.com/spr-vs-elisa/]
- 98. SPR vs ELISA | Comparing techniques for biomolecular ... [https://nicoyalife.com/blog/spr-vs-elisa-comparing-techniques-for-biomolecular-detection/]
- 99. Comparison of Biomolecular Interaction Techniques — SPR ... [https://www.reichertspr.com/insights/white-papers/comparison-of-biomolecular-interaction-techniques---white-paper]