Ligand Engineering in Perovskite Quantum Dots: A Comparative Analysis of OA/OAm vs. Benzoic Acid for Advanced Optoelectronic Devices
This article provides a comprehensive comparison of traditional oleic acid/oleylamine (OA/OAm) ligands and benzoic acid (BA) based ligands in perovskite quantum dot (PQD) devices.
Ligand Engineering in Perovskite Quantum Dots: A Comparative Analysis of OA/OAm vs. Benzoic Acid for Advanced Optoelectronic Devices
Abstract
This article provides a comprehensive comparison of traditional oleic acid/oleylamine (OA/OAm) ligands and benzoic acid (BA) based ligands in perovskite quantum dot (PQD) devices. It explores the foundational roles of these ligands in PQD synthesis and stability, details methodological advances in ligand exchange and application, addresses key challenges in device optimization, and presents a rigorous validation of performance metrics. By synthesizing recent research, this review serves as a critical resource for researchers and scientists developing next-generation PQD-based optoelectronic devices, with particular relevance for applications requiring high charge transport and environmental stability.
The Fundamental Roles of OA/OAm and Benzoic Acid Ligands in PQD Synthesis and Stability
Perovskite quantum dots (PQDs), particularly all-inorganic CsPbX₃ (X = Cl, Br, I) nanocrystals, represent a significant class of functional materials characterized by their three-dimensional quantum confinement and exceptional optical properties, including high color purity, tunable bandgaps, and high photoluminescence quantum yield (PLQY) [1]. The crystal structure features Cs⁺ at lattice corners, Pb²⁺ at the cube center, and X⁻ anions forming [PbX₆] octahedra, creating a framework where structural stability is governed by the Goldschmidt tolerance factor and octahedral factor [1].
Surface ligands are indispensable molecules that coordinate with PQD surfaces, serving critical functions in nucleation, growth, defect passivation, and aggregation prevention [1]. Traditional long-chain ligands like oleic acid (OA) and oleylamine (OAm) dynamically bind to surfaces but readily detach, causing structural and optical instability under environmental stressors like humidity, oxygen, and light [1]. Ligand engineering through in situ and post-synthesis strategies using multidentate or strongly-coordinating ligands is essential to enhance stability and luminescence performance for optoelectronic and biomedical applications [2] [1].
Performance Comparison of OA/OAm versus Alternative Ligands
Quantitative Comparison of Ligand Performance
Table 1: Experimental Performance Data of OA/OAm vs. Alternative Ligands
| Ligand Type | PLQY Improvement | Stability Retention | Key Findings | Experimental Conditions |
|---|---|---|---|---|
| OA/OAm (Traditional) | Baseline | Significant degradation under environmental stress [1] | Dynamic binding leads to detachment; limits practical application [1] | Standard hot-injection method; ambient conditions [1] |
| L-Phenylalanine (L-PHE) | 3% PLQY enhancement [2] | >70% initial PL intensity after 20 days UV exposure [2] | Superior photostability; effective defect passivation [2] | CsPbI₃ PQDs; UV exposure testing [2] |
| Trioctylphosphine (TOP) | 16% PLQY enhancement [2] | Data not specified in study [2] | Effective suppression of non-radiative recombination [2] | CsPbI₃ PQDs; optimized synthesis [2] |
| Trioctylphosphine Oxide (TOPO) | 18% PLQY enhancement [2] | Data not specified in study [2] | Highest PL enhancement among tested ligands [2] | CsPbI₃ PQDs; coordinated with undercoordinated Pb²⁺ ions [2] |
| Multidentate Ligands | Not quantitatively specified | Significantly improved compared to OA/OAM [1] | Strengthened binding with PQD surfaces; reduced environmental impact [1] | Various ligand engineering approaches [1] |
Analysis of Comparative Performance
The experimental data reveals substantial improvements in both optical performance and environmental stability when alternative ligands replace traditional OA/OAm systems. While OA and OAm facilitate initial synthesis, their dynamic binding nature creates inherent instability, with ligand detachment leading to rapid PQD degradation [1]. In contrast, specialized ligands like TOPO and L-PHE demonstrate significantly enhanced coordination with undercoordinated Pb²⁺ ions and surface defects, resulting in PLQY enhancements of 18% and 3% respectively, with L-PHE particularly excelling in photostability retention [2].
The superior performance of engineered ligands stems from their stronger coordination binding and improved passivation of surface defects, which effectively suppress non-radiative recombination pathways that diminish luminescence efficiency [2] [1]. Multidentate ligands offer particularly promising stability enhancements due to their multiple binding sites, creating more robust connections to PQD surfaces that resist environmental stressors like humidity, temperature fluctuations, and prolonged illumination [1].
Experimental Protocols and Methodologies
PQD Synthesis and Ligand Modification Protocol
Table 2: Detailed Experimental Methodology for PQD Synthesis and Ligand Testing
| Experimental Phase | Protocol Details | Parameters & Controls |
|---|---|---|
| PQD Synthesis | Hot-injection method with precise precursor control [2] | Cs₂CO₃, PbI₂ precursors; 1-octadecene solvent; 140-180°C temperature range [2] |
| Ligand Modification | Surface passivation using TOP, TOPO, and L-PHE [2] | Coordination with undercoordinated Pb²⁺ ions and surface defects [2] |
| Optical Characterization | Photoluminescence intensity and quantum yield measurements [2] | Emission wavelength (698-713 nm), FWHM (24-28 nm) [2] |
| Stability Assessment | Continuous UV exposure testing [2] | PL intensity measurement over 20 days [2] |
| Optimization Parameters | Temperature, injection volume, reaction duration [2] | Optimal temperature: 170°C; Hot-injection volume: 1.5 mL [2] |
Experimental Workflow Visualization
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for PQD Ligand Studies
| Reagent/Material | Function/Purpose | Application Notes |
|---|---|---|
| Cesium Carbonate (Cs₂CO₃) | Cesium precursor for PQD synthesis [2] | 99% purity; requires precise molar ratios [2] |
| Lead Iodide (PbI₂) | Lead source for perovskite formation [2] | 99% purity; stoichiometric control critical [2] |
| 1-Octadecene (ODE) | Reaction solvent medium [2] | Non-polar solvent for high-temperature synthesis [2] |
| Oleic Acid (OA) | Traditional carboxylic acid ligand [2] [1] | Chelates with lead atoms; prevents aggregation [1] |
| Oleylamine (OAm) | Traditional amine ligand [1] | Binds to halide ions through hydrogen bonding [1] |
| Trioctylphosphine (TOP) | Phosphine-based ligand modifier [2] | 99% purity; provides 16% PLQY enhancement [2] |
| Trioctylphosphine Oxide (TOPO) | Phosphine oxide ligand modifier [2] | 99% purity; provides 18% PLQY enhancement [2] |
| L-Phenylalanine (L-PHE) | Amino acid ligand modifier [2] | 98% purity; superior photostability retention [2] |
Ligand-PQD Interaction Mechanisms
Ligand Binding and Surface Passivation
The interaction between ligands and PQD surfaces follows distinct coordination mechanisms that determine their effectiveness. Traditional OA ligands coordinate with surface lead atoms through carboxylate groups, while OAm interacts with halide ions via hydrogen bonding [1]. These monodentate binding modes create relatively weak, dynamic attachments that readily dissociate under environmental stress.
In contrast, engineered ligands like TOP, TOPO, and L-PHE form more stable coordination complexes with undercoordinated Pb²⁺ ions at the PQD surface [2]. The phosphine groups in TOP and TOPO, and the amino-carboxylate functionality in L-PHE, provide stronger binding that effectively passivates surface defects which would otherwise serve as non-radiative recombination centers, thereby enhancing both luminescence efficiency and material stability [2].
Stability Enhancement Pathways
The comprehensive comparison between traditional OA/OAm ligands and emerging alternatives reveals significant advancements in PQD performance through strategic ligand engineering. While OA/OAm systems provide fundamental synthesis capabilities, their instability limitations restrict practical device applications. The experimental data demonstrates that specialized ligands like TOPO and L-PHE offer substantial improvements in PLQY (up to 18% enhancement) and photostability (>70% retention after 20 days UV exposure), addressing critical barriers to PQD commercialization.
For researchers and drug development professionals, these findings highlight the importance of surface chemistry optimization in developing robust PQD-based diagnostic and therapeutic platforms. The ligand engineering approaches discussed provide a framework for designing PQD systems with enhanced stability and optical performance, essential requirements for biomedical applications including biosensing, bioimaging, and targeted drug delivery where consistent performance under physiological conditions is paramount.
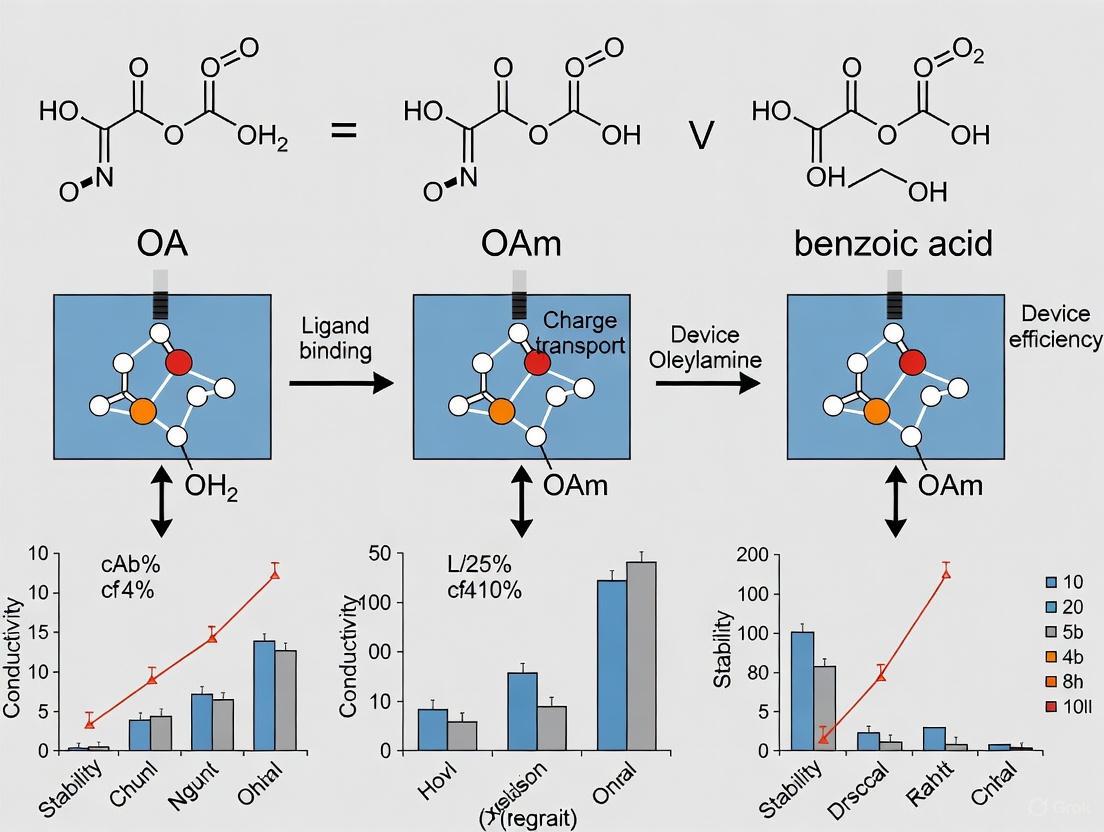
In the field of colloidal synthesis, particularly for perovskite quantum dots (PQDs) and other functional nanomaterials, the ligand pair of oleic acid (OA) and oleylamine (OAm) has long been the conventional choice for controlling nanocrystal growth, stability, and dispersion. These long-chain alkyl ligands are indispensable in standard synthesis protocols like hot-injection and ligand-assisted reprecipitation (LARP), where they facilitate the dissolution of inorganic precursors, control nucleation and growth kinetics, and passivate surface defects to enhance optical properties [1] [3]. OA typically chelates with metal atoms (e.g., Pb²⁺) on the nanocrystal surface, while OAm interacts with halide ions through hydrogen bonding, creating a dynamic organic shell that prevents aggregation during synthesis [1]. Despite their widespread adoption, this conventional ligand system suffers from inherent limitations that severely restrict the performance and commercial viability of resulting materials in optoelectronic devices. This review objectively compares the OA/OAm system with emerging alternatives, particularly benzoic acid (BA)-based ligands, within PQD device research, providing experimental data and methodologies to guide material selection and innovation.
Fundamental Limitations of the OA/OAm Ligand System
Dynamic Binding and Ligand Desorption
The primary limitation of OA and OAm ligands lies in their dynamic binding behavior to the PQD surface. These ligands attach through relatively weak coordinate bonds, leading to easy detachment during processing, storage, or operation.
- Instability Consequences: Ligand desorption creates unprotected surface sites, causing rapid degradation of optical properties and structural integrity. Studies show that OA/OAm-capped PQDs can lose over 30% of their photoluminescence quantum yield (PLQY) within hours when exposed to ambient conditions [1] [4].
- Surface Defect Formation: Detached ligands leave behind uncoordinated lead atoms and halide vacancies that act as trap states, promoting non-radiative recombination and reducing device efficiency [5].
Steric Hindrance and Poor Charge Transport
The long hydrocarbon chains (C18) of OA and OAm create a thick insulating layer (∼2-3 nm) around individual PQDs, severely impeding inter-dot electronic coupling.
- Interparticle Separation: This steric barrier maintains a large interparticle distance (>5 nm) in films, disrupting wavefunction overlap and carrier transport [6].
- Conductivity Limitations: Films capped with OA/OAm exhibit exceptionally low conductivity (∼8× lower than short-chain ligand alternatives), creating significant bottlenecks in device performance [5].
Table 1: Quantitative Performance Comparison of OA/OAm versus Alternative Ligand Systems
| Performance Metric | OA/OAM Ligands | Benzoic Acid (BA) Ligands | N-alkylmonoamine Ligands | FASCN Ligands |
|---|---|---|---|---|
| Photoluminescence Quantum Yield (PLQY) Retention | <70% over 30 days (ambient) [4] | >90% over 30 days [7] | ~70% after 30 days [4] | >90% after treatment [5] |
| Film Conductivity | Baseline (Reference) | 8× improvement [5] | Data not available | 8× improvement [5] |
| Solar Cell PCE | ~10% (reference) [6] | 12.13%-14.27% [7] | Data not available | Data not available |
| Binding Energy | -0.22 eV (OA); -0.18 eV (OAm) [5] | Data not available | Data not available | -0.91 eV [5] |
| Water Resistance | Rapid degradation (minutes) [4] | Significant improvement [7] | >1 hour stability [4] | >30 minutes at >99% humidity [5] |
Limited Packing Density and Morphological Control
The bulky nature of OA and OAM leads to suboptimal packing of PQDs in solid films, directly impacting device architecture.
- Void Formation: Films deposited with OA/OAm-capped PQDs contain significant voids due to steric repulsion, reducing active material density and charge transport efficiency [6].
- Binary Mixing Challenge: Research shows that even strategic mixing of different PQD sizes only marginally improves packing density to ∼37%, as the long-chain ligands still prevent optimal face-to-face contact [6].
Benzoic Acid-Based Ligands: A Promising Alternative
Sequential Ligand Exchange with Benzoic Acid
Recent advances demonstrate benzoic acid (BA) as a superior alternative when deployed in a sequential ligand exchange strategy. This approach typically involves initial removal of long-chain ligands followed by BA passivation.
Diagram 1: Sequential ligand exchange workflow for OA/OAm replacement
Experimental Protocol for Sequential Ligand Exchange
Materials: FAPbI3 PQDs with OA/OAm ligands, dipropylamine (DPA), benzoic acid (BA), n-hexane, n-octane, methyl acetate (MeOAc).
Procedure:
- DPA Treatment: Centrifuge the pristine PQD solution (3 mg/mL in hexane) at 9000 rpm for 5 minutes. Redisperse the precipitate in DPA (0.5 vol% in octane) and stir for 30 seconds. Add methyl acetate as anti-solvent and centrifuge again to obtain the treated PQDs [7].
- BA Treatment: Redisperse the DPA-treated PQDs in BA solution (1.5 mg/mL in octane) and stir for 30 seconds. Precipitate with methyl acetate and centrifuge to obtain the final DPA+BA-treated PQDs [7].
- Film Fabrication: Dissolve the treated PQDs in octane (30 mg/mL) and spin-coat onto substrates at 2000-3000 rpm for 30 seconds to form uniform films without thermal annealing [7].
Performance Advantages of Benzoic Acid-Modified PQDs
- Enhanced Electronic Properties: BA-treated FAPbI3 PQD films exhibit significantly improved electronic coupling and suppressed carrier non-radiative recombination, leading to superior device performance [7].
- Mechanical Stability: Flexible PQD solar cells using BA-treated PQDs maintain ~90% of initial PCE after 100 bending cycles (7 mm radius), demonstrating exceptional mechanical robustness [7].
- Scalability: The one-step fabrication process enabled by BA treatment allows for larger-area devices with minimal efficiency loss - a critical advantage over conventional OA/OAm systems [7].
Table 2: Device Performance Metrics for Different Ligand Systems in PQD Solar Cells
| Device Characteristics | OA/OAM-Based Devices | BA-Based Flexible Devices | Improvement |
|---|---|---|---|
| Power Conversion Efficiency (PCE) | ~10% (reference CsPbI3) [6] | 12.13% (0.06 cm²) [7] | +21.3% |
| Larger-Area Performance | Significant efficiency drop [6] | 9.96% (0.98 cm²) [7] | Minimal scaling loss |
| Bending Cycle Stability | Rapid degradation [7] | ~90% PCE retention after 100 cycles [7] | Dramatic improvement |
| Charge Transport | Low conductivity, high recombination [6] | Enhanced electronic coupling [7] | Significant improvement |
Beyond Benzoic Acid: Other Emerging Ligand Strategies
N-alkylmonoamine Ligands
Straight-chain alkylmonoamines (e.g., dodecylamine, tetradecylamine) form more ordered protective layers on PQD surfaces compared to the haphazard arrangement of OAm. These ligands demonstrate:
- Exceptional Stability: >70% PLQY retention after 30 days in ambient air [4]
- Water Resistance: Stability for over 1 hour in water [4]
- Thermal Endurance: 60% PLQY retention after 60 minutes at 80°C [4]
Bidentate Ligands
Formamidine thiocyanate (FASCN) represents a breakthrough in ligand design with its bidentate binding capability and short carbon chain (<3 atoms).
- Superior Binding Affinity: FASCN exhibits fourfold higher binding energy (-0.91 eV) compared to OA/OAm, preventing ligand desorption [5].
- Enhanced Conductivity: FASCN-treated films show eightfold higher conductivity than OA/OAm-capped films [5].
- Record Device Performance: FASCN-enabled near-infrared LEDs achieve 23% external quantum efficiency - double that of control devices [5].
Diagram 2: OA/OAm limitations and corresponding ligand engineering solutions
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents for Ligand Engineering Studies
| Reagent/Material | Function in Research | Application Context |
|---|---|---|
| Oleic Acid (OA) | Conventional X-type ligand; coordinates with metal cations | Baseline comparison; control experiments [1] [3] |
| Oleylamine (OAm) | Conventional L-type ligand; interacts with halide anions | Standard synthesis protocol component [1] [3] |
| Benzoic Acid (BA) | Short-chain acidic ligand for surface passivation | OA replacement in sequential ligand exchange [7] |
| Dipropylamine (DPA) | Medium-chain amine for initial ligand stripping | First step in sequential ligand exchange [7] |
| Formamidine Thiocyanate (FASCN) | Bidentate ligand with strong coordination | High-efficiency device fabrication [5] |
| N-alkylmonoamines | Straight-chain amines for dense protective layers | Stability enhancement studies [4] |
| Methyl Acet/Anti-solvent | Precipitation agent for ligand exchange procedures | PQD purification and processing [7] |
| 1-Octadecene (ODE) | Non-polar solvent for high-temperature synthesis | Reaction medium in hot-injection methods [1] [6] |
The conventional OA/OAm ligand system, while foundational to colloidal nanocrystal synthesis, presents significant limitations for advanced optoelectronic applications. The dynamic binding nature, long insulating chains, and resultant poor stability of these ligands fundamentally constrain device performance and durability. Emerging alternatives—particularly benzoic acid-based ligands in sequential exchange protocols—demonstrate markedly superior properties through enhanced electronic coupling, improved charge transport, and exceptional mechanical stability in flexible device architectures.
Future research directions should focus on developing multidentate ligand systems with optimized chain lengths and binding groups, establishing standardized ligand exchange protocols for reproducible device fabrication, and exploring environmentally benign ligand alternatives to address sustainability concerns. The continued advancement of ligand engineering will undoubtedly play a crucial role in unlocking the full potential of perovskite quantum dots and other nanomaterials for next-generation optoelectronic devices.
The performance of perovskite quantum dot (PQD) optoelectronic devices, such as solar cells and light-emitting diodes (LEDs), is profoundly influenced by the surface chemistry of the quantum dots. Ligand engineering has emerged as a critical strategy to enhance the electronic properties and stability of these materials. For years, long-chain insulating ligands like oleic acid (OA) and oleylamine (OAm) have been standard in colloidal synthesis for their ability to stabilize nanocrystals. However, their poor charge transport characteristics severely limit device performance [1]. This guide objectively compares the performance of conventional OA/OAm ligands with short-chain conductive ligands, specifically benzoic acid, within the context of PQD devices.
The broader thesis is that replacing dynamic, long-chain ligands with robust, short-chain alternatives like benzoic acid directly addresses the key trade-off between nanocrystal stability and charge transport. Benzoic acid (C6H5COOH) presents a unique molecular structure—a benzene ring attached to a carboxyl group—that facilitates strong binding to the PQD surface while enabling superior inter-dot coupling [8] [9]. Its conjugated phenethylammonium (PEA+) derivatives further enhance this effect. This article provides a comparative analysis of these ligand systems, supported by experimental data and detailed methodologies, to inform researchers and scientists in the field.
Molecular Structures and Key Properties
The fundamental differences in performance between OA/OAm and benzoic acid-based ligands originate from their distinct molecular structures and physical properties.
Oleic Acid (OA) and Oleylamine (OAm): These are long-chain aliphatic ligands (C18). OA is a carboxylic acid, while OAm is a primary amine. During the hot-injection synthesis of PQDs, they coordinate to the surface ions—OA chelates with lead atoms, and OAm binds to halide ions via hydrogen bonding [1]. Their long hydrocarbon chains provide excellent steric hindrance, preventing aggregation and ensuring good colloidal stability in non-polar solvents. However, these same insulating chains create a physical barrier that impedes charge transfer between adjacent PQDs, leading to high film resistivity [9] [1].
Benzoic Acid and Its Derivatives: Benzoic acid (
C7H6O2) is an aromatic carboxylic acid. The presence of the benzene ring makes it a conjugated molecule. In its phenethylammonium iodide (PEAI) form, which is commonly used in solid-state ligand exchange, the PEA+ cation (C6H5C2H4NH3+) features a phenyl group connected to an ammonium group via a two-carbon alkyl chain. This structure offers multiple advantages [8]:- The short-chain nature of the molecule reduces the distance between PQDs.
- The conjugated phenyl group can enhance electronic coupling between dots.
- The ammonium group provides strong ionic binding to the halide-rich PQD surface, passivating surface defects more effectively than dynamically bound OA/OAm.
Table 1: Comparative Properties of Common PQD Ligands.
| Property | Oleic Acid (OA) / Oleylamine (OAm) | Benzoic Acid / PEAI |
|---|---|---|
| Chain Type | Long, aliphatic (insulating) | Short, aromatic (conjugated) |
| Primary Function | Colloidal stabilization during synthesis | Conductive capping and defect passivation |
| Binding Nature | Dynamic, labile | Robust, ionic |
| Impact on Inter-Dot Distance | Large (~nanometers) | Reduced (sub-nanometer) |
| Charge Transport | Poor, high resistivity | Enhanced, high conductivity |
| Defect Passivation | Moderate, often incomplete | Strong, especially with LBL treatment [8] |
Binding Mechanisms and Surface Chemistry
The binding mechanisms of these ligands to the PQD surface are pivotal for understanding their effectiveness.
Conventional Ligand Dynamics
In a typical CsPbI3 PQD synthesis, OA and OAm cap the surface, creating a core-shell structure. However, this binding is dynamic and labile. The ligands are easily desorbed during processing or under operational stress, creating surface vacancy defects that act as traps for charge carriers, non-radiatively recombining them and degrading device performance [9] [1]. Furthermore, subsequent post-treatment steps with short ligands like formamidinium iodide (FAI) often only passivate the top layer of the film, leaving underlying trap states unaddressed [8].
Benzoic Acid and PEAI Binding
Benzoic acid and its derivative PEAI interact with the PQD surface through a more controlled and stable process. The short-chain phenethylammonium (PEA+) ion in PEAI binds ionically to the [PbX6]4- octahedra on the PQD surface. The aromatic ring facilitates π-π interactions, promoting closer packing and better electronic coupling between adjacent dots [8]. A key advancement is the layer-by-layer (LBL) solid-state exchange strategy. In this method, the PEAI solution is applied after the deposition of each PQD layer during film fabrication, rather than as a single post-treatment at the end. This ensures thorough and uniform removal of the pristine OAm+ ligands and their replacement with PEA+ throughout the entire film thickness, leading to superior defect passivation and a more homogeneous structure [8].
Experimental Performance Comparison
The transition from OA/OAm to benzoic acid-based ligands yields measurable improvements in the performance and stability of PQD devices, particularly solar cells.
Photovoltaic Performance
Research demonstrates that employing a PEAI layer-by-layer (LBL) strategy on CsPbI3 PQDs results in solar cells with a champion power conversion efficiency (PCE) of 14.18%, a significant improvement over devices relying on conventional ligand management [8]. This is attributed to a higher open-circuit voltage (V_OC) of 1.23 V and improved fill factor, stemming from reduced trap-assisted recombination. Furthermore, using methyl benzoate (MeBz) as an antisolvent, which hydrolyzes to release benzoate ions, within an alkaline-augmented hydrolysis strategy has pushed the certified PCE of hybrid FA0.47Cs0.53PbI3 PQD solar cells to a record 18.3% [9]. This method facilitates a near-complete substitution of insulating OA ligands with conductive benzoate capping.
Electroluminescent Performance
The same surface management that enhances photovoltaic performance also benefits electroluminescence. CsPbI3 PQD films treated with the PEAI-LBL method exhibit a narrow electroluminescence peak at 691 nm and an enhanced luminance of 130 Cd/m² when the device is operated as an LED [8]. This dual functionality highlights the balanced carrier transport and injection achieved by benzoic acid-based ligands, enabling efficient devices that can function both as solar cells and light emitters.
Environmental Stability
Devices incorporating benzoic acid ligands show markedly improved stability. Unencapsulated CsPbI3 PQD solar cells based on PEAI-LBL films "exhibited excellent stability under a high-humidity environment" (30-50% relative humidity, ~25 °C) [8]. This is due to the higher hydrophobicity of the phenyl group in the PEA+ ligand compared to aliphatic chains, which better protects the moisture-sensitive perovskite core.
Table 2: Quantitative Performance Comparison of PQD Solar Cells with Different Ligand Systems.
| Performance Metric | Conventional OA/OAm with FAI Post-Treatment | Benzoic Acid Derivative (PEAI-LBL) | Alkaline-Augmented Methyl Benzoate |
|---|---|---|---|
| Champion PCE (%) | ~10.77 (Early report) [1] | 14.18 [8] | 18.30 (Certified) [9] |
Open-Circuit Voltage (V_OC) |
Lower (deficit) | 1.23 V [8] | High |
| Steady-State PCE (%) | N/A | N/A | 17.85 [9] |
| Electroluminescence Luminance | Weak or none | 130 Cd/m² [8] | N/A |
| Moisture Stability | Poor | Excellent (30-50% RH) [8] | Improved [9] |
Detailed Experimental Protocols
To ensure reproducibility, here are detailed methodologies for key experiments cited in this guide.
This protocol describes the fabrication of a CsPbI3 PQD film with enhanced conductive capping.
- Materials: CsPbI3 PQDs in hexane, Methyl Acetate (MeOAc, anhydrous), Phenethylammonium Iodide (PEAI), Ethyl Acetate (EtOAc, anhydrous).
- Substrate Preparation: Clean FTO or ITO substrates with oxygen plasma for 10 minutes.
- Film Deposition:
- Spin-coat the CsPbI3 PQD solution onto the substrate at 3000 rpm for 30 seconds.
- Immediately after spin-coating, rinse the film with MeOAc (∼0.5 mL) to remove residual solvent and initiate the removal of long-chain ligands.
- Subsequently, treat the wet film with a PEAI solution (e.g., 2 mg/mL in EtOAc, ∼0.5 mL) to execute the solid-state ligand exchange.
- Spin-dry the film at 3000 rpm for another 30 seconds.
- Layer Buildup: Repeat steps 1-4 for 4-6 cycles to achieve the desired film thickness (e.g., ~300 nm).
- Post-treatment: The final film may be thermally annealed at a mild temperature (e.g., 70°C for 5 minutes) to improve crystallinity.
This advanced protocol uses alkaline conditions to maximize the hydrolysis of antisolvent into conductive ligands.
- Materials: Hybrid FA0.47Cs0.53PbI3 PQDs, Methyl Benzoate (MeBz), Potassium Hydroxide (KOH).
- Antisolvent Preparation: Add a controlled amount of KOH (e.g., 0.5-2.0 mM) to pure MeBz and stir to create the alkaline antisolvent mixture.
- Film Deposition & Rinsing:
- Spin-coat the PQD solution to form a solid film.
- During the interlayer rinsing step of the LBL process, use the KOH/MeBz alkaline antisolvent instead of neat MeOAc or MeBz.
- The alkaline environment facilitates the rapid hydrolysis of MeBz into benzoate anions, which readily substitute the pristine OA- ligands on the PQD surface.
- Characterization: FTIR and XPS confirm the replacement of long-chain ligands with benzoate. This method results in up to twice the conventional amount of conductive capping ligands.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Ligand Engineering in PQD Research.
| Reagent | Function / Role | Key Property / Application |
|---|---|---|
| Oleic Acid (OA) | Long-chain ligand, Precursor capping agent | Provides colloidal stability during synthesis [1] |
| Oleylamine (OAm) | Long-chain ligand, Precursor capping agent | Binds to halide ions; controls PQD growth [1] |
| Benzoic Acid | Short-chain conductive ligand | Model compound for studying aromatic ligand binding [8] |
| Phenethylammonium Iodide (PEAI) | Short-chain conductive ligand | Used in solid-state exchange for defect passivation & charge transport [8] |
| Methyl Benzoate (MeBz) | Ester antisolvent | Hydrolyzes to release benzoate ligands during film rinsing [9] |
| Potassium Hydroxide (KOH) | Alkaline catalyst | Enhances hydrolysis of ester antisolvents in the AAAH strategy [9] |
| Methyl Acetate (MeOAc) | Standard ester antisolvent | Removes excess solvent and initiates ligand exchange in conventional LBL [9] |
The comparative analysis unequivocally demonstrates that benzoic acid and its derivatives are superior conductive ligands compared to the conventional OA/OAm system for high-performance PQD devices. The key differentiators are their short, conjugated molecular structure, which enables enhanced electronic coupling and charge transport, and their robust binding mechanism, which provides excellent defect passivation and environmental stability. Experimental data from recent studies confirm that ligand engineering strategies centered on benzoic acid—such as the PEAI layer-by-layer approach and the alkaline-augmented hydrolysis of methyl benzoate—directly lead to record efficiencies in PQD solar cells and enable multifunctional electroluminescent devices. For researchers and scientists, focusing on optimizing these conductive ligand systems presents a clear pathway toward unlocking the full commercial potential of perovskite quantum dot technologies.
Comparative Analysis of Ligand Packing Density and Steric Hindrance
In the field of perovskite quantum dot (PQD) device research, surface ligands play a critical role in determining both the optoelectronic properties and environmental stability of these promising semiconductor nanomaterials. Ligands such as oleic acid (OA) and oleylamine (OAm) have served as foundational capping agents in PQD synthesis, but their dynamic binding characteristics and substantial steric profiles often limit device performance and longevity. This comparative analysis examines the fundamental relationship between ligand packing density and steric hindrance in PQD systems, with specific focus on comparing traditional OA/OAm ligands with emerging benzoic acid-based alternatives. Understanding these structure-property relationships is essential for researchers and scientists seeking to design next-generation PQD devices with enhanced operational stability and optimized photophysical characteristics.
The performance of PQD devices is intrinsically linked to the molecular-level interactions at the nanocrystal surface, where ligand packing density determines passivation efficacy and steric hindrance influences both molecular assembly and long-term structural integrity. This review systematically evaluates these parameters through experimental data, computational simulations, and performance metrics to provide a comprehensive resource for drug development professionals and materials scientists working in nanomaterial optimization.
Theoretical Foundations: Ligand Packing Density and Steric Hindrance
Fundamental Concepts and Definitions
Ligand packing density refers to the number of ligand molecules per unit area on a nanoparticle surface, typically expressed as molecules per square nanometer (molecules/nm²). This parameter critically influences numerous material properties including colloidal stability, surface accessibility, and optoelectronic behavior. Precise quantification of packing density enables researchers to optimize surface coverage for specific applications. For instance, studies on gold nanoparticles (AuNPs) conjugated with 3-mercaptopropionic acid (MPA) demonstrated packing densities ranging from 6.7 to 9.0 nm⁻² across particle sizes from 5-100 nm, showing remarkable consistency regardless of core dimensions [10].
Steric hindrance describes the physical restriction of molecular motion or binding due to the spatial arrangement of atoms or functional groups within a molecule. This repulsive force originates from Pauli exclusion principles and dipole-dipole interactions between adjacent atoms, creating destabilizing forces that can distort molecular geometry, induce bond-breaking events, or inhibit chemical reactivity [11]. In PQD systems, steric effects significantly impact ligand binding dynamics, surface passivation quality, and nanocrystal assembly.
Interrelationship of Packing Density and Steric Hindrance
The relationship between packing density and steric hindrance represents a critical balance in nanomaterial design. High packing densities typically enhance surface passivation but may introduce detrimental steric effects that compromise structural stability. Research on lamellar structures composed of monosodium N-stearoyl-l-glutamate (MSSG) with higher alcohols demonstrated that despite favorable packing parameters, excessive vertical steric compressibility (Sc = 8.2) led to poor colloidal stability compared to systems with lower steric hindrance (Sc = 4.7) [12].
Table 1: Key Parameters in Ligand-Surface Interactions
| Parameter | Definition | Measurement Techniques | Impact on Material Properties |
|---|---|---|---|
| Packing Density | Number of ligand molecules per unit surface area | ICP-OES, XPS, SPR [10] [13] | Colloidal stability, surface accessibility, ligand availability |
| Steric Hindrance | Spatial restriction due to molecular structure | Computational simulation, STM, IETS [11] | Binding kinetics, molecular conformation, aggregation behavior |
| Steric Compressibility | Vertical repulsion between hydrophobic tails | Molecular dynamics simulations [12] | Colloidal stability, lamellar integrity, phase behavior |
| Ligand Arrangement | Spatial distribution of binding sites | Scanning tunneling microscopy [11] | Receptor binding avidity, cellular responses, signaling |
Ligand Performance in PQD Systems: OA/OAm vs. Benzoic Acid
Traditional OA/OAM Ligand Systems
OA and OAm have served as the conventional ligand pair in PQD synthesis since the early development of these materials. In CsPbX₃ PQDs, OA coordinates with surface lead atoms through carboxylate groups while OAm interacts with halide ions via hydrogen bonding [1]. This complementary binding mechanism facilitates nanocrystal growth and provides initial colloidal stabilization.
However, OA/OAm ligands suffer from significant limitations. Their long alkyl chains (C18) create substantial steric profiles, and their dynamic binding behavior leads to easy detachment from PQD surfaces. This labile binding results in progressive surface degradation and ultimately poor device longevity [1]. The inherent flexibility of these long-chain ligands also permits close approach of neighboring nanocrystals, facilitating aggregation under environmental stressors.
Benzoic Acid Ligand Systems
Benzoic acid and its derivatives represent promising alternatives to conventional OA/OAm ligands. Their rigid aromatic structure provides different steric constraints compared to aliphatic chains, while the conjugated π-system may offer additional electronic interactions with the perovskite surface. Although the search results do not provide specific experimental data for benzoic acid in PQD systems, the general advantages of aromatic ligands can be extrapolated from their molecular characteristics:
The compact molecular structure of benzoic acid derivatives potentially enables higher packing densities while maintaining effective steric protection. Their rigid backbone creates defined coordination geometry that may reduce ligand entanglement and improve surface passivation reproducibility. Additionally, the potential for π-π interactions with the aromatic components of the perovskite lattice could enhance binding affinity compared to purely aliphatic systems.
Experimental Methodologies for Characterization
Quantifying Ligand Packing Density
Accurate determination of ligand packing density requires sophisticated analytical approaches. Inductively coupled plasma optical emission spectrometry (ICP-OES) has emerged as a powerful technique for ensemble-averaged measurements. The protocol involves:
- Sample Preparation: Centrifugation and digestion of ligand-conjugated nanoparticles to ensure complete dissolution [10]
- Elemental Analysis: Simultaneous measurement of core (e.g., Au, Pb) and ligand-specific (e.g., S) elements
Data Calculation: Determination of ligand density using the formula:
Ligand density = (Cₗᵢgₐₙd × Nₐ)/(Sₐ × ρ × d)
Where Cₗᵢgₐₙd is ligand concentration, Nₐ is Avogadro's number, Sₐ is specific surface area, ρ is density, and d is diameter [13]
For AuNPs with thiolated ligands, this approach has demonstrated packing densities of 7.8 nm⁻² on average for MPA, with variations from 6.7 to 9.0 nm⁻² across different particle sizes [10].
Probing Steric Hindrance Effects
Scanning tunneling microscopy (STM) combined with inelastic electron tunneling spectroscopy (IETS) provides molecular-scale visualization of steric effects at ligand-surface interfaces. The experimental workflow includes:
- Surface Preparation: Creation of atomically clean Au(111) surfaces with diverse structural features (FCC, HCP, step edges, herringbone reconstructions) [11]
- Ligand Deposition: Controlled dosing of evaporated ligands under ultra-high vacuum (10⁻¹⁰ Torr) at 5 K
- Thermal Migration Studies: Sequential warming to room temperature and recooling to observe ligand redistribution to energetically favorable sites
- Topographical Imaging: High-resolution STM to determine molecular conformation and binding preferences
- Spectroscopic Analysis: IETS characterization of vibrational fingerprints to assess ligand-surface interactions
This methodology has revealed clear steric-pressure-induced binding selectivity, with m-terphenyl isocyanide ligands preferentially migrating to high-curvature step edges where steric repulsion is minimized [11].
Diagram 1: Experimental workflow for characterizing steric hindrance effects at ligand-surface interfaces
Computational Modeling Approaches
Molecular dynamics (MD) simulations provide atomic-level insights into ligand behavior under various conditions. The protocol involves:
- System Construction: Building structurally homologous model molecules with controlled variations (e.g., benzene ring arrangements, alkyl chains, heteroatoms) [14]
- Force Field Parameterization: Assigning appropriate potential energy functions for intra- and intermolecular interactions
- Shear Simulation: Applying shear conditions to model real-world processing environments
- Trajectory Analysis: Monitoring aggregation behavior, viscosity properties, and molecular conformations over extended timescales (e.g., 60 ns)
This approach has revealed that branched benzene ring arrangements in polycyclic hydrocarbon nuclei generate greater molecular steric hindrance, resulting in higher viscosity compared to linear arrangements [14].
Comparative Performance Analysis
Structural and Binding Characteristics
The structural differences between OA/OAm and benzoic acid ligands impart distinct binding behavior and steric profiles:
Table 2: Structural Comparison of Ligand Systems
| Parameter | OA/OAm System | Benzoic Acid System |
|---|---|---|
| Molecular Structure | Long aliphatic chains (C18) | Compact aromatic ring |
| Binding Motif | Carboxylate-Pb coordination; Amine-halide H-bonding | Carboxylate-Pb coordination; potential π-interactions |
| Steric Profile | Flexible, dynamic conformation | Rigid, defined geometry |
| Binding Affinity | Moderate, labile binding [1] | Potentially stronger, more directional |
| Packing Density Range | Variable, ligand-dependent | Potentially higher due to compact structure |
Impact on PQD Performance Metrics
The choice of ligand system directly influences critical PQD performance parameters:
Optical Properties: Ligand detachment in OA/OAm systems creates surface defects that diminish photoluminescence quantum yield (PLQY) over time. The more rigid binding of aromatic ligands like benzoic acid may provide superior defect passivation for maintained optical performance.
Environmental Stability: The dynamic nature of OA/OAm binding renders PQDs susceptible to degradation under environmental stressors (humidity, heat, light). Enhanced binding affinity in benzoic acid systems could improve resistance to these degradation pathways.
Charge Transport: While OA/OAm ligands provide excellent initial colloidal stability, their insulating alkyl chains can impede interdot charge transfer. The conjugated π-system of benzoic acid may offer improved electronic coupling for enhanced charge transport in device applications.
Aggregation Behavior: MD simulations of model asphaltene molecules demonstrate that molecular structure significantly influences aggregation propensity, with branched architectures exhibiting higher viscosity due to increased steric hindrance [14]. This principle translates to PQD systems, where ligand architecture dictates assembly characteristics.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagents for Ligand-PQD Studies
| Reagent/Material | Function | Application Context |
|---|---|---|
| Oleic Acid (OA) | Long-chain carboxylic acid ligand | Traditional PQD synthesis and stabilization [1] |
| Oleylamine (OAm) | Long-chain amine ligand | Complementary co-ligand in PQD synthesis [1] |
| Benzoic Acid Derivatives | Aromatic carboxylic acid ligands | Alternative ligands with enhanced rigidity |
| m-Terphenyl Isocyanide | Sterically encumbered model ligand | Fundamental studies of steric effects [11] |
| 3-Mercaptopropionic Acid | Thiolated model ligand | Packing density studies on Au surfaces [10] |
| Monosodium N-stearoyl-l-glutamate | Amino acid surfactant | Model for studying steric compressibility [12] |
| ICP-OES Instrumentation | Elemental analysis | Quantification of ligand packing density [10] [13] |
| STM-IETS System | Surface characterization | Molecular-scale visualization of steric effects [11] |
This comparative analysis demonstrates that ligand packing density and steric hindrance represent complementary design parameters that collectively determine PQD performance characteristics. Traditional OA/OAm ligands provide effective initial passivation but suffer from dynamic binding and substantial steric requirements that limit device integration. Benzoic acid-based systems offer potential advantages through their rigid aromatic structure, potentially enabling higher packing densities with reduced steric constraints.
Future research directions should focus on quantitative comparisons of packing density between these ligand classes using ICP-based methodologies, systematic evaluation of binding affinity through temperature-dependent studies, and exploration of synergistic effects in mixed-ligand systems. Additionally, the development of standardized protocols for steric hindrance quantification would enable more direct comparison across research groups and material systems.
As PQD devices progress toward commercial application, rational ligand design based on fundamental understanding of packing density and steric effects will be essential for achieving the optimal balance between processing, stability, and performance characteristics.
Impact of Ligand Choice on Intrinsic PQD Stability Against Moisture, Heat, and Light
The stability of perovskite quantum dots (PQDs) remains a critical challenge limiting their commercial application in optoelectronics, solar cells, and lighting devices. A primary factor governing this stability is the dynamic interaction between surface ligands and the perovskite crystal structure. This guide provides a comprehensive comparison between traditionally employed long-chain ligands (oleic acid OA and oleylamine OAm) and emerging short-chain alternatives, with specific focus on benzoic acid (BA), examining their impact on PQD resilience against thermal, moisture, and light-induced degradation. The objective analysis presented herein, framed within broader thesis research on PQD device performance, reveals that ligand selection involves fundamental trade-offs between stability enhancement and charge transport efficiency.
Ligand Functions and Degradation Mechanisms
Ligands are molecules attached to PQD surfaces that facilitate nucleation during synthesis, passivate surface defects, and prevent aggregation [1]. Conventional long-chain OA and OAm ligands provide steric stabilization but bind weakly to the ionic perovskite surface, leading to detachment during processing or operation and creating defects that act as entry points for degradation [1] [15].
Thermal degradation proceeds via different pathways depending on composition. Cs-rich PQDs undergo a phase transition from a black γ-phase to a non-perovskite yellow δ-phase, while FA-rich PQDs directly decompose into PbI2 at elevated temperatures [16]. The strength of ligand binding energy significantly influences the degradation onset temperature, with stronger-binding ligands enhancing thermal resilience [16]. Furthermore, the intrinsic ionic nature of perovskites makes them susceptible to ion migration under light or electrical bias, which is exacerbated by poor surface passivation and leads to phase segregation and accelerated decomposition [15].
Comparative Analysis of Ligand Performance
The following sections provide a detailed experimental comparison of OA/OAm versus short-chain ligands, particularly benzoic acid, across key stability parameters.
Thermal Stability
Table 1: Thermal Stability Performance of PQDs with Different Ligands
| Ligand Type | Composition | Experimental Method | Key Findings | Reference |
|---|---|---|---|---|
| OA/OAm | CsxFA1-xPbI3 | In situ XRD (30°C to 500°C) | FA-rich PQDs decompose directly to PbI2 starting at ~150°C. Cs-rich PQDs transition through a yellow δ-phase before decomposition. | [16] |
| Benzoic Acid (BA) | FAPbI3 | Sequential ligand exchange (DPA+BA), device testing | The ligand-exchanged films enabled flexible solar cells that retained ~90% of initial PCE after 100 bending cycles, indicating robust thermal-mechanical stability. | [7] |
| DBSA | CsPbBr3 | Stability against high-temperature annealing | DBSA-capped PQDs exhibited suppressed growth and exceptional stability against Ostwald ripening when annealed at high temperatures in various solvents. | [15] |
Experimental Insight: The in situ XRD methodology involves heating PQD samples deposited on a substrate under an argon flow while continuously collecting X-ray diffraction patterns. This allows direct observation of phase transitions (e.g., peaks at 25.2°, 29.0° attributed to PbI2 emergence) and grain growth in real-time [16]. The enhanced stability from BA is attributed to a sequential exchange process where dipropylamine (DPA) first removes loose long-chain ligands, followed by BA passivation which effectively binds to surface sites, reducing defect density and strengthening the interface [7].
Moisture and Environmental Stability
Table 2: Stability Against Moisture and Environmental Factors
| Ligand Type | Composition | Experimental Method | Key Findings | Reference |
|---|---|---|---|---|
| OA/Oam | CsPbX3 | Exposure to ambient conditions | Dynamic binding leads to easy ligand detachment, making PQDs highly sensitive to humidity and polar solvents, causing rapid degradation. | [1] |
| Benzoic Acid (BA) | FAPbI3 | Sequential ligand exchange (DPA+BA) | Films showed "improved environmental stability" due to effective surface passivation that protects against ambient factors. | [7] |
| DBSA | CsPbBr3 | Purification and dispersion stability tests | DBSA-PQDs maintained high colloidal stability and photoluminescence quantum yield (PLQY) even after multiple purification cycles, resisting aggregation. | [15] |
Experimental Insight: The superior performance of short-chain acids like BA and DBSA stems from their stronger, more stable bonding with the PQD surface. DBSA, a sulfonic acid, provides particularly strong anchoring. This robust binding creates a more effective barrier against water molecule ingress and prevents ligand desorption during processing, which is a common failure point for OA/OAm-capped PQDs [7] [15].
Optoelectronic Performance and Device Stability
While stability is crucial, ligand choice directly impacts charge transport and final device efficiency.
Table 3: Device Performance and Optoelectronic Properties
| Ligand Type | Composition | Device/Test | Performance Outcome | Reference |
|---|---|---|---|---|
| OA/OAm | General PQDs | Film Conductivity | Long-chain ligands form an insulating layer, severely impeding charge transport between QDs. | [7] [15] |
| Benzoic Acid (BA) | FAPbI3 | Flexible Solar Cell | Champion PCE of 12.13% (0.06 cm²); 9.96% (0.98 cm²). Achieved via one-step fabrication, showing enhanced electronic coupling. | [7] |
| DBSA | CsPbBr3 | Light-Emitting Diodes (LEDs) | Despite high PLQY and stability in solution, devices showed "poor external quantum efficiency (EQE)" due to insufficient ligand removal. | [15] |
Experimental Insight: The one-step fabrication of FAPbI3 PQD solar cells involves a sequential ligand exchange process. A solution of PQDs capped with OA/OAm is treated with DPA in hexane, which displaces the long-chain ligands. Subsequently, BA is introduced to passivate the newly created coordinative sites. This mixture is then directly spin-coated to form the final film, eliminating the need for complex layer-by-layer deposition [7]. This process enhances electronic coupling between QDs, leading to improved charge transport and higher device performance compared to traditional insulating ligands.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for PQD Ligand Engineering Research
| Reagent Solution | Function in Research | Application Context |
|---|---|---|
| Oleic Acid (OA) / Oleylamine (OAm) | Standard long-chain ligands for classic synthesis; provide initial steric stabilization but offer weak binding. | Baseline control for stability and performance comparisons; standard synthesis via hot-injection or LARP methods. |
| Benzoic Acid (BA) | Short-chain ligand used to replace OA/OAm; improves charge transport and passivation strength. | Sequential ligand exchange strategies to create conductive, stable PQD films for photovoltaic devices. |
| Dipropylamine (DPA) | Medium-chain amine used to initiate the removal of native long-chain ligands (OA/OAm). | Used as a first step in sequential ligand exchange to strip initial ligands before passivation with a short-chain acid. |
| Dodecylbenzene Sulfonic Acid (DBSA) | Strong-binding sulfonic acid ligand; provides exceptional passivation and ripening inhibition. | Investigating extreme stability against heat, solvents, and purification; studying ion migration suppression. |
| 1-Octadecene (ODE) | Non-polar solvent used as a reaction medium in high-temperature synthesis (e.g., hot-injection). | Common solvent for precursor dissolution and reaction environment in classic PQD synthesis. |
| Methyl Acetate (MeOAc) | Polar antisolvent used to precipitate PQDs from their crude synthesis solution. | Standard purification agent in layer-by-layer (LbL) deposition and washing procedures. |
The choice between traditional OA/OAm ligands and short-chain alternatives like benzoic acid presents a clear trade-off. OA/OAm systems offer good initial dispersibility but suffer from intrinsic instability and poor charge transport. In contrast, benzoic acid, applied via sequential exchange strategies, demonstrates superior performance in constructing stable, efficient PQD devices capable of withstanding thermal, environmental, and mechanical stress. While very strong ligands like DBSA offer unparalleled stability, they can overly insulate QDs, highlighting that the ultimate ligand choice must be application-specific, balancing the competing demands of maximum stability and optimal electronic performance. Future research should focus on developing dynamically switchable or hybrid ligand systems that provide robust protection during processing and storage while enabling high conductivity in operational devices.
Experimental Workflow and Signaling Pathways
The following diagram illustrates the core sequential ligand exchange protocol, a key experimental method for enhancing PQD stability and performance.
Methodological Advances in Ligand Engineering and Device Integration
The synthesis and surface ligand engineering of perovskite quantum dots (PQDs) are pivotal for advancing their application in optoelectronic devices. Ligands are molecules attached to the PQD surface that coordinate the crystallization process, passivate surface defects, and ultimately determine the nanocrystals' electronic properties and environmental stability [3]. This guide objectively compares two predominant synthetic approaches: the established hot-injection and ligand-assisted reprecipitation (LARP) methods utilizing long-chain oleic acid (OA) and oleylamine (OAm), and the emerging post-synthetic ligand exchange strategies employing short-chain benzoic acid (BA). Framed within a broader thesis on ligand performance in PQD devices, this comparison elucidates the trade-offs between superior dispersibility and detrimental charge transport inhibition posed by OA/OAm, against the enhanced electronic coupling and defect passivation offered by BA-based ligand exchange [7] [3] [8].
Synthesis Protocols and Ligand Functions
The foundational synthesis of high-quality PQDs is typically achieved through methods that incorporate long-chain ligands, while subsequent post-synthetic treatments are designed to overcome their limitations.
Hot-Injection and LARP Methods (Using OA/OAm)
The hot-injection method involves the rapid injection of a cesium-oleate precursor into a hot (140–200 °C) solution of lead halide (e.g., PbBr₂) dissolved in 1-octadecene (ODE) with OA and OAm [17] [18]. This sudden introduction of the Cs precursor causes supersaturation, triggering the nucleation and growth of monodisperse PQDs [17]. The Ligand-Assisted Reprecipitation (LARP) technique, in contrast, is performed at room temperature. A precursor solution containing the perovskite salts (e.g., PbX₂ and CH₃NH₃X) along with OA and OAm in a polar solvent like DMF is dropped into a poor solvent, such as toluene [17]. The sharp change in solvent environment induces the instantaneous formation of luminescent colloidal QDs [17].
In both methods, OA and OAm are indispensable. OA (a carboxylic acid) chelates with lead atoms on the PQD surface, while OAm (an amine) binds to halide ions [3]. This coordination facilitates the dissolution of inorganic precursors, controls the nucleation and growth kinetics to produce nanocrystals of desired size and shape, and provides a steric barrier that prevents aggregation, ensuring good dispersibility in non-polar solvents [3] [17].
Post-Synthetic Ligand Exchange (Using Benzoic Acid)
Post-synthetic ligand exchange is a solid-state treatment performed on already-synthesized and deposited PQD films. A prominent strategy is the sequential ligand exchange. For instance, one protocol involves first treating OA/OAm-capped FAPbI₃ PQD films with dipropylamine (DPA) to dynamically remove the insulating long-chain ligands, which inevitably introduces extra surface defects [7]. This is followed by a treatment with a BA solution, where the short-chain benzoic acid effectively passivates the newly created defects and completes the replacement of the original ligands [7]. Another approach uses a layer-by-layer (LBL) solid-state exchange during film deposition, where BA or a similar short-chain ligand (e.g., phenethylammonium iodide, PEAI) in a solvent like ethyl acetate is applied after the deposition of each PQD layer, systematically replacing OAm/OA and passivating the entire film bulk [7] [8].
The primary role of BA is twofold. First, as a short-chain ligand, it reduces the inter-dot spacing, dramatically enhancing electronic coupling and charge transport between adjacent PQDs. Second, its effective binding to the perovskite surface passivates surface defects, suppressing non-radiative recombination losses and improving both device efficiency and environmental stability [7].
Performance Comparison in PQD Devices
The following tables summarize the experimental data from key studies, comparing the performance of PQD devices based on OA/OAm ligands and those treated with post-synthetic BA exchange.
Table 1: Photovoltaic Performance of PQD Solar Cells
| Perovskite Material | Ligand System | Device Type | Power Conversion Efficiency (PCE) | Open-Circuit Voltage (Vₒc) | Key Improvements |
|---|---|---|---|---|---|
| FAPbI₃ PQDs [7] | OA/OAm (Control) | Flexible Solar Cell | Reported as lower | - | Baseline performance |
| FAPbI₃ PQDs [7] | DPA + BA Sequential Exchange | Flexible Solar Cell | 12.13% (0.06 cm²) | - | Enhanced electronic coupling, suppressed non-radiative recombination. |
| FAPbI₃ PQDs [7] | DPA + BA Sequential Exchange | Flexible Solar Cell | 9.96% (0.98 cm²) | - | Good performance retention at larger active area. |
| CsPbI₃ PQDs [8] | PEAI Layer-by-Layer (LBL) Exchange | Rigid Solar Cell | 14.18% | 1.23 V | Balanced carrier transport/injection, superior defect passivation. |
Table 2: Optoelectronic and Stability Performance
| Perovskite Material | Ligand System | Photoluminescence Quantum Yield (PLQY) | Mechanical Stability | Environmental Stability | Electroluminescence Performance |
|---|---|---|---|---|---|
| CsPbX₃ (General) [3] | OA/OAm | High (up to ~90%) in solution | - | Low; sensitive to polar solvents, humidity | - |
| FAPbI₃ PQDs [7] | DPA + BA Sequential Exchange | - | ~90% of initial PCE retained after 100 bending cycles | Improved | - |
| CsPbI₃ PQDs [8] | PEAI LBL Exchange | - | - | Excellent moisture stability (30-50% RH) | 130 cd/m² luminance |
Analysis of Comparative Data
The data clearly demonstrates that post-synthetic ligand exchange with short-chain aromatic acids like BA significantly enhances device performance. The removal of insulating OA/OAm ligands reduces inter-dot distance, leading to a marked improvement in charge carrier transport, as evidenced by the high PCEs achieved in both flexible and rigid solar cells [7] [8]. Furthermore, the effective defect passivation by BA results in higher open-circuit voltages and improved stability against environmental factors like moisture [7] [8]. Notably, ligand-capped PQDs also exhibit intrinsic mechanical robustness, which is critical for flexible electronics, maintaining performance after repeated bending [7]. The ability of BA-treated films to support efficient electroluminescence further underscores their balanced electronic properties, enabling the development of bifunctional optoelectronic devices [8].
Experimental Protocols
- Synthesis: FAPbI₃ PQDs are synthesized via a standard hot-injection method, resulting in dots capped with OA/OAm ligands ("Control PQDs").
- Film Deposition: The PQD solution is spin-coated onto a substrate.
- First Ligand Exchange (DPA): The wet PQD film is treated immediately by dynamically dropping a DPA solution in n-hexane (e.g., 2 µL mL⁻¹) onto the spinning film. This step removes the long-chain ligands but may create defects.
- Second Ligand Exchange (BA): A solution of BA in a solvent like a hexane/octane mixture (e.g., 2 mg mL⁻¹) is dynamically dropped onto the spinning film to passivate the surface defects and complete the ligand exchange.
- Washing: The film is washed with methyl acetate (MeOAc) or ethyl acetate (EtOAc) to remove residual reactants and by-products.
- Repetition: Steps 2-5 are repeated multiple times in a layer-by-layer fashion to build a thick, electronically coupled PQD film.
- Synthesis: CsPbI₃ PQDs are synthesized via a hot-injection method with OA/OAm ligands.
- First Layer Deposition: The PQD solution is spin-coated onto a substrate.
- Washing: The film is washed with MeOAc to remove excess solvent and some ligands.
- Ligand Exchange (e.g., PEAI/BA): A solution of the short-chain ligand (e.g., PEAI or BA in EtOAc) is spin-coated onto the PQD layer.
- Repetition: Steps 2-4 are repeated 3-5 times to build a multilayer film.
- Final Post-Treatment: The complete film may undergo a final brief post-treatment with a ligand solution for optimal passivation.
Workflow and Signaling Pathway Diagrams
The following diagram illustrates the procedural workflow and the functional impact of the two ligand strategies on the PQD surface.
Diagram 1: Workflow comparing hot-injection synthesis with OA/OAm and subsequent post-synthetic ligand exchange with BA.
Diagram 2: Functional impact of OA/OAm versus BA ligands on PQD properties.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for PQD Synthesis and Ligand Exchange
| Reagent | Function | Application Context |
|---|---|---|
| Oleic Acid (OA) | Long-chain carboxylic acid; chelates with Pb²⁺ on PQD surface for growth control and steric stabilization [3] [17]. | Hot-injection, LARP synthesis. |
| Oleylamine (OAm) | Long-chain amine; binds to halide ions, assists precursor dissolution and nanocrystal growth [3] [17]. | Hot-injection, LARP synthesis. |
| 1-Octadecene (ODE) | High-booint, non-polar solvent; serves as the reaction medium for synthesis [17]. | Hot-injection synthesis. |
| Benzoic Acid (BA) | Short-chain aromatic carboxylic acid; replaces OA/OAm to enhance charge transport and passivate surface defects [7]. | Post-synthetic ligand exchange. |
| Dipropylamine (DPA) | Alkylamine; used in a sequential strategy to dynamically remove long-chain OA/OAm ligands [7]. | Post-synthetic ligand exchange. |
| Methyl Acetate (MeOAc) | Polar, non-solvent for PQDs; used as a washing agent to remove excess ligands and solvents during film processing [7] [8]. | Film purification and ligand exchange. |
| Cesium Carbonate (Cs₂CO₃) | Cesium precursor; reacts with OA to form Cs-oleate for the hot-injection synthesis of all-inorganic CsPbX₃ PQDs [19] [17]. | Hot-injection synthesis. |
| Lead Halide (PbX₂) | Source of lead and halide ions (X = Cl, Br, I); the core component of the perovskite crystal structure [17]. | Universal precursor. |
The direct comparison between hot-injection/LARP with OA/OAm and post-synthetic ligand exchange with BA reveals a critical trajectory in PQD research: moving from synthesis-focused dispersibility to device-performance-centric electronic properties. While OA and OAm are irreplaceable for producing high-quality, monodisperse PQDs in solution, their insulating nature presents a fundamental bottleneck for efficient charge transport in solid-state films. The post-synthetic ligand exchange strategy, particularly using short-chain ligands like benzoic acid, directly addresses this limitation. By enabling enhanced electronic coupling, superior defect passivation, and improved mechanical and environmental stability, BA treatment unlocks higher performance in both photovoltaic and light-emitting devices. Therefore, for researchers targeting high-efficiency PQD devices, integrating a robust post-synthetic ligand exchange protocol is not merely an optimization step but a necessary progression beyond foundational synthesis.
Ligand exchange is a critical surface manipulation process in the fabrication of perovskite quantum dot (PQD) photovoltaics, directly influencing their optoelectronic properties and device performance. This process involves replacing the long-chain insulating ligands used during synthesis with shorter, more conductive ligands to enhance charge transport while maintaining colloidal stability and passivating surface defects. The strategic selection of ligands and the engineering of exchange techniques, including antisolvent-assisted methods, are paramount for developing high-efficiency and stable PQD solar cells. This guide provides a comparative analysis of traditional oleic acid/oleylamine (OA/OAm) ligands versus benzoic acid (BA)-based alternatives, framing the discussion within the broader thesis of performance optimization in PQD devices.
Performance Comparison: OA/OAm vs. Benzoic Acid Ligands
The following table summarizes the key performance characteristics of PQD solar cells employing different ligand systems, based on recent experimental studies.
Table 1: Performance Comparison of Ligand Systems in PQD Solar Cells
| Ligand System | Device Performance (PCE) | Key Electrical & Optical Properties | Mechanical Stability | Primary Advantages | Inherent Limitations |
|---|---|---|---|---|---|
| Long-Chain (OA/OAm) | Lower performance; serves as a baseline for synthesized PQDs [7]. | Insulating layer impedes charge transport; high non-radiative recombination [7] [20]. | Provides initial colloidal stability but poor film robustness [7]. | Excellent dispersibility in non-polar solvents; effective initial passivation [20] [1]. | Dynamic binding leads to detachment; poor electronic conductivity [7] [1]. |
| Benzoic Acid (BA) | Champion PCE of 12.13% on flexible substrate; 14.27% on rigid substrate [7]. | Enhanced electronic coupling; suppressed carrier recombination; improved charge extraction [7]. | Maintains ~90% of initial PCE after 100 bending cycles (7 mm radius) [7]. | Short chain improves conductivity; π-π stacking enhances stability and charge delocalization [7] [21]. | Requires a sequential exchange strategy for optimal defect passivation [7]. |
Experimental Protocols for Ligand Exchange
Sequential Ligand Exchange with DPA and BA
A prominent methodology for integrating BA ligands involves a sequential, two-step treatment that first removes long-chain ligands and then passivates the newly exposed surface. The following diagram outlines the workflow for this sequential ligand exchange process.
Diagram 1: Workflow for Sequential Ligand Exchange with DPA and BA
Detailed Protocol [7]:
- PQD Synthesis and Film Fabrication: Synthesize FAPbI₃ PQDs via the standard hot-injection method, capped with OA and OAm ligands. Deposit the PQD solution onto the substrate via spin-coating to form an initial film.
- DPA Treatment (Surface Activation):
- Prepare a solution of dipropylamine (DPA) in n-octane (e.g., 0.2 vol%).
- During the spin-coating process, dynamically drip the DPA solution onto the rotating PQD film.
- The function of DPA is to partially and mildly strip the native long-chain OA/OAm ligands. This process activates the surface by enhancing electronic coupling between PQDs but simultaneously introduces coordination-unsaturated Pb²⁺ sites (defects).
- BA Treatment (Defect Passivation):
- Immediately following the DPA treatment, dynamically drip a solution of benzoic acid (BA) in a solvent like n-octane or ethyl acetate onto the film.
- The short-chain BA ligand effectively coordinates with the under-coordinated Pb²⁺ sites created in the previous step. This passivates surface defects and completes the replacement of the original insulating ligands.
- Washing and Drying: After the ligand exchange, the film is typically washed with a pure antisolvent (e.g., methyl acetate or ethyl acetate) via spin-coating to remove the displaced ligands and any residual reagents. The film is then dried.
The Role of Antisolvent Engineering
Antisolvents are crucial throughout the ligand exchange and film formation process. They are typically polar solvents that are miscible with the dispersion solvent but induce precipitation of the PQDs.
- Functions: Antisolvents reduce the solubility of PQDs, promoting their aggregation and the formation of dense, solid films during deposition. Furthermore, they are used as washing agents to purify the film by dissolving and removing the exchanged long-chain ligands and reaction by-products [7] [21].
- Commonly Used Antisolvents: Methyl acetate (MeOAc) and ethyl acetate (EtOAc) are widely employed due to their effectiveness in purifying films without damaging the perovskite crystal structure [7].
Underlying Mechanisms and Signaling Pathways
The superior performance of BA-treated PQD devices stems from fundamental improvements in surface chemistry and electronic structure. The logical pathway from ligand exchange to enhanced device function is illustrated below.
Diagram 2: Logical Pathway from Ligand Exchange to Enhanced Device Performance
Key Mechanistic Insights:
- Defect Passivation: The primary role of BA is to coordinate with unsaturated Pb²⁺ ions on the PQD surface. This coordination neutralizes trap states that would otherwise capture charge carriers and lead to non-radiative recombination, thereby increasing photoluminescence quantum yield (PLQY) and open-circuit voltage (VOC) [7] [21].
- Enhanced Charge Transport: Compared to long-chain OA/OAm, the short molecular length of BA reduces the inter-dot spacing, facilitating wave function overlap and significantly improving the electronic coupling between adjacent PQDs. This creates more efficient pathways for charge carrier transport, leading to higher short-circuit current density (JSC) [7] [20].
- π-π Stacking: The aromatic benzene ring in BA enables π-π stacking interactions between ligands on neighboring QDs. This provides superior mechanical stability to the film, making it more resilient to bending and deformation—a critical property for flexible electronics [7] [21].
The Scientist's Toolkit: Essential Research Reagents
This table details the key chemicals and materials required to conduct the described ligand exchange processes and fabricate PQD solar cells.
Table 2: Essential Research Reagents for PQD Ligand Exchange Studies
| Reagent/Material | Function/Application | Examples from Literature |
|---|---|---|
| Oleic Acid (OA) / Oleylamine (OAm) | Long-chain ligands for initial PQD synthesis and stabilization in non-polar solvents [20] [1]. | Standard ligands in hot-injection and LARP synthesis methods [7] [1]. |
| Benzoic Acid (BA) | Short-chain ligand for surface passivation and enhancement of charge transport [7] [21]. | Used in sequential exchange with DPA to achieve high PCE (12.13%) in flexible FAPbI₃ PQD solar cells [7]. |
| Dipropylamine (DPA) | Amine-based molecule used to initiate ligand exchange by removing native long-chain ligands [7]. | Employed as the first step in a sequential strategy to partially strip OA/OAm from FAPbI₃ PQDs [7]. |
| Methyl Acetate / Ethyl Acetate | Antisolvents for film purification and washing; induce PQD film formation and remove ligand exchange residues [7]. | Used as washing agents after ligand exchange treatments to purify films [7]. |
| n-Octane / n-Hexane | Non-polar solvents for dispersing synthesized PQDs and dissolving new ligands during processing [7]. | Solvent for DPA and BA solutions during the sequential exchange process [7]. |
Incorporating BA-Ligands into Functional Films for Solar Cells and Light-Emitting Diodes (LEDs)
Ligand engineering plays a pivotal role in determining the optoelectronic properties and device performance of quantum dot (QD)-based technologies. Within this field, benzoic acid (BA) and its derivatives have emerged as promising ligands to enhance the performance of functional films in solar cells and light-emitting diodes (LEDs). This guide provides an objective comparison between traditional oleic acid/oleylamine (OA/OAm) ligands and BA-based ligands, focusing on their performance in perovskite quantum dot (PQD) and lead sulfide (PbS) QD devices. We present experimental data and methodologies to help researchers make informed decisions about ligand selection for specific applications.
Ligand Fundamentals and Mechanisms of Action
Traditional OA/OAm Ligand System
The combination of oleic acid (OA) and oleylamine (OAm) represents the conventional ligand system used in the synthesis of quantum dots. These long-chain aliphatic ligands dynamically bind to the QD surface, where OA coordinates with metal cations (Pb²⁺) and OAm interacts with halide anions through hydrogen bonding [3] [21]. While effective for providing colloidal stability during synthesis, this system creates strong insulating barriers between quantum dots, impeding efficient carrier transport in solid films [22]. The dynamic binding nature also leads to ligand detachment, resulting in surface defects and compromised stability [3].
BA-Based Ligand Systems
Benzoic acid (BA) ligands feature an aromatic benzene ring structure that provides distinct advantages over aliphatic ligands. The conjugated π-system enables enhanced charge transport through π-π stacking interactions between adjacent QDs [21]. This fundamental structural difference translates to improved electrical conductivity in functional films. Additionally, the rigid aromatic structure provides strong binding affinity to the QD surface while occupying less space than bulky aliphatic chains [21].
Table 1: Fundamental Properties of Ligand Systems
| Property | OA/OAm System | BA-Based System |
|---|---|---|
| Chemical Structure | Long aliphatic chains (C18) | Aromatic benzene ring |
| Binding Nature | Dynamic, labile | Strong, stable |
| Inter-Dot Transport | Insulating barrier | π-π stacking enhanced |
| Steric Hindrance | High | Moderate |
| Surface Coverage | Partial due to repulsion | Improved due to planar structure |
Experimental Protocols and Methodologies
Synthesis of BA-Capped Quantum Dots
Ligand-Assisted Reprecipitation (LARP) Method: For perovskite QDs, the LARP approach enables BA ligand incorporation at room temperature. Prepare a precursor solution of PbBr₂ and CsBr in dimethylformamide (DMF) at 0.4 M concentration. Dissolve BA ligand in the precursor solution at a molar ratio of 1:1 with lead precursor. Rapidly inject this solution into toluene under vigorous stirring (800-1000 rpm). The immediate formation of colloids indicates nucleation of BA-capped PQDs. Recover the QDs by centrifugation at 8000 rpm for 5 minutes and redisperse in anhydrous toluene for film formation [21].
Solution-Phase Ligand Exchange: For existing QDs capped with OA/OAm, post-synthetic ligand exchange can introduce BA ligands. Prepare a ligand exchange solution containing 10 mg/mL BA in a 1:1 mixture of methanol and hexane. Add this solution dropwise to the QD solution under inert atmosphere with continuous stirring. Allow the exchange to proceed for 2 hours at 50°C. Purify the exchanged QDs through sequential centrifugation and redispersion in non-solvent [21].
Film Fabrication and Device Integration
Spin-Coating Protocol: For LED applications, deposit BA-capped QD films using a sequential spin-coating process. Prepare QD ink with concentration of 15-20 mg/mL in octane. Spin-coat at 1500-2000 rpm for 30 seconds in nitrogen atmosphere. Anneal the films at 80°C for 10 minutes to remove residual solvent. For multilayer devices, repeat the process with appropriate intermediate layers [23].
Solar Cell Fabrication: For photovoltaic devices, create a p-i-n structure with BA-capped QD active layers. Deposit ZnO electron transport layer via sol-gel process, followed by BA-capped QD layer (150-200 nm thickness) using layer-by-layer dip coating. Complete with MoO₃/Au electrodes through thermal evaporation. For infrared photovoltaics using PbS CQDs, employ solution-phase ligand exchange with (BA)₂PbI₄ precursors to form robust 2D-perovskite-like shells on the QD surface [24].
Performance Comparison in Optoelectronic Devices
Light-Emitting Diodes
BA-based ligands significantly enhance LED performance through improved charge transport and defect passivation. Devices employing BA-capped CsPbBr₃ QDs demonstrate external quantum efficiencies (EQE) exceeding 20% for green emission, comparable to state-of-the-art organic and CdSe-based LEDs [23]. The conductive nature of BA ligands reduces the efficiency roll-off at high current densities by facilitating balanced charge injection.
Table 2: LED Performance Metrics with Different Ligand Systems
| Parameter | OA/OAm-Capped QDs | BA-Capped QDs |
|---|---|---|
| EQE (Green) | 12-16% | 20-25% |
| Current Efficiency (cd/A) | 35-45 | 55-75 |
| Turn-on Voltage (V) | 3.2-3.8 | 2.8-3.2 |
| FWHM (nm) | 18-22 | 16-20 |
| Operational Lifetime (T₅₀, hours) | 50-100 | 150-300 |
Solar Cells
In photovoltaic applications, BA ligands enable superior performance through enhanced inter-dot charge transport and reduced recombination losses. PbS CQD solar cells utilizing (BA)₂PbI₄ ligands achieve power conversion efficiencies (PCE) of 13.1% for small-bandgap CQDs (1.3 eV), significantly outperforming PbI₂-capped controls (11.3%) [24]. The robust passivation of non-polar <100> facets by BA-based ligands is particularly effective in larger CQDs, with devices maintaining 95% of initial PCE after 500 hours of ambient operation.
Table 3: Photovoltaic Performance with Different Ligand Systems
| Parameter | OA-Capped PbS CQDs | PbI₂-Capped PbS CQDs | (BA)₂PbI₄-Capped PbS CQDs |
|---|---|---|---|
| PCE (%) | 5-8 | 10-11.3 | 12-13.1 |
| Jₛc (mA/cm²) | 22-25 | 26-28 | 29-31 |
| Vₒc (V) | 0.55-0.58 | 0.60-0.62 | 0.63-0.65 |
| FF (%) | 55-60 | 62-65 | 65-68 |
| Stability (T₈₀, hours) | <100 | 200-300 | >500 |
Stability and Environmental Resistance
The structural stability of BA-based ligand systems represents a significant advancement over traditional OA/OAm ligands. BA-capped PQD films retain over 90% of initial photoluminescence intensity after 200 hours of continuous illumination, compared to <50% for OA/OAm-capped counterparts [21]. The enhanced stability originates from the strong binding affinity of carboxyl groups to surface lead atoms and the hydrophobic nature of the aromatic rings, which creates a protective barrier against moisture ingress [24].
Thermal stability tests demonstrate that BA-capped films maintain structural integrity at 100°C for over 24 hours with minimal photoluminescence quenching (Δλ < 1 nm), while OA/OAm-capped films exhibit significant emission shifts (Δλ = 12 nm) under identical conditions [25]. This thermal robustness is critical for device operation and processing at elevated temperatures.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for BA-Ligand Studies
| Reagent | Function | Application Notes |
|---|---|---|
| Benzoic Acid (BA) | Primary ligand for surface passivation | Use anhydrous grade, store in desiccator |
| Oleic Acid (OA) | Reference ligand | Distill under reduced pressure before use |
| Oleylamine (OAm) | Co-ligand in reference systems | Store under nitrogen, use spectroscopic grade |
| n-Butylammonium Iodide (BAI) | Precursor for 2D perovskite ligands | Critical for (BA)₂PbI₄ formation |
| Lead Iodide (PbI₂) | Metal precursor | Purify by recrystallization from DMF |
| Dimethylformamide (DMF) | Polar solvent for ligand exchange | Dry over molecular sieves, use in glovebox |
| Cesium Carbonate (Cs₂CO₃) | Cesium source for perovskite QDs | React with hydrohalic acids to form Cs-oleate |
| Zinc Oxide Nanoparticles | Electron transport layer | Use 20-30 nm diameter for optimal film formation |
The experimental data comprehensively demonstrates that BA-based ligand systems outperform traditional OA/OAm ligands across critical performance metrics in both LED and solar cell applications. The enhanced electrical conductivity, superior stability, and robust surface passivation provided by BA ligands translate directly to improved device efficiencies and operational lifetimes. While OA/OAm systems remain valuable for initial QD synthesis, BA-based ligands offer significant advantages for functional film fabrication in optoelectronic devices. Researchers should consider the specific application requirements when selecting ligand systems, with BA-based approaches particularly favorable for devices demanding high charge transport and environmental stability.
Visual Appendix
In the pursuit of high-performance perovskite quantum dot (PQD) optoelectronic devices, surface ligand management has emerged as a critical frontier. Traditional long-chain ligands like oleic acid (OA) and oleylamine (OAm) provide excellent colloidal stability during synthesis but form insulating layers that severely impede charge transport between adjacent quantum dots in solid films. This limitation has driven research toward short-chain ligands, notably benzoic acid (BA) and its derivatives, which offer improved conductivity while maintaining sufficient passivation. The strategic combination of BA with other short ligands, particularly when integrated with crosslinking approaches, represents a sophisticated advancement in PQD engineering that simultaneously addresses multiple challenges: enhanced charge transport, improved environmental stability, and preserved optoelectronic properties.
The fundamental thesis guiding this comparison is that the transition from conventional OA/OAm ligand systems to optimized short-ligand combinations represents a paradigm shift in PQD device architecture. While OA and OAm provide excellent steric stabilization during synthesis, their dynamic binding and insulating nature create significant bottlenecks for device performance. BA-based approaches, especially when synergistically combined with other short ligands and crosslinking strategies, enable a more favorable balance between passivation and conductivity, unlocking new possibilities in both photovoltaic and electroluminescent applications.
Quantitative Performance Comparison of Ligand Strategies
Table 1: Performance metrics of PQD devices utilizing different ligand approaches
| Ligand Strategy | Device Type | PCE (%) | VOC (V) | EL Luminance (cd/m²) | Stability Retention | Key Advantages |
|---|---|---|---|---|---|---|
| Conventional OA/OAm | Solar Cell | 10.77 [8] | ~1.0 [8] | N/R | Poor (reference) | Excellent colloidal stability |
| BA-only post-treatment | Electroluminescent Solar Cell | 5.46 [8] | N/R | 584 [8] | Moderate | Improved charge transport |
| PEAI-LBL | Bifunctional Device | 14.18 [8] | 1.23 [8] | 130 [8] | Excellent (high humidity) [8] | Balanced carrier injection/transport [8] |
| TPPI ligand | Electroluminescent Solar Cell | 15.21 [8] | N/R | EQE: 3.8% [8] | Good | Strong surface binding [8] |
| Polymerization-induced crosslinking | Patterning Process | N/A | N/A | N/A | Enhanced environmental/mechanical robustness [26] | High-resolution patterning, maintained PLQY [26] |
Table 2: Material properties and characterization metrics for ligand systems
| Ligand System | Film PLQY | Inter-dot Coupling | Charge Mobility | Defect Passivation | Process Compatibility |
|---|---|---|---|---|---|
| Long-chain (OA/OAm) | Moderate | Weak | Low | Moderate | Excellent synthesis, poor device integration |
| BA analogs | High | Moderate | Moderate-High | Good | Good solution processing |
| PEAI short ligands | High [8] | Enhanced [8] | Balanced electron/hole transport [8] | Enhanced [8] | Layer-by-layer deposition [8] |
| Crosslinked networks | Maintained high [26] | Controlled | Device-dependent | Good | Photolithographic patterning [26] |
Experimental Protocols and Methodologies
Layer-by-Layer Solid-State Ligand Exchange
The layer-by-layer (LBL) solid-state ligand exchange represents a significant methodological advancement over conventional post-treatment approaches. In the PEAI-LBL strategy for CsPbI3 PQDs [8]:
- Quantum Dot Synthesis: CsPbI3 PQDs are synthesized via hot-injection method using OA and OAm as initial capping ligands.
- Film Fabrication: The LBL process involves multiple cycles of spin-coating PQD solutions followed by solid-state ligand exchange using PEAI (phenethylammonium iodide) solution after each layer deposition.
- Ligand Exchange Mechanism: The conjugated PEAI ligand, acting as a short-chain ligand with a phenyl group, encapsulates CsPbI3 PQDs during the LBL procedure, effectively replacing the original long-chain OAm ligands.
- Comparative Control: Conventional approaches typically involve building the entire film first through multiple cycles of spin-coating and methyl acetate (MeOAc) treatment, followed by a single post-treatment using FAI or PEAI salts.
This methodology enables more complete removal of long-chain ligands, enhanced defect passivation throughout the film thickness (not just the top layer), and regulation of balanced carrier transport and injection [8]. The enhanced inter-dot coupling and more uniform passivation achieved through this approach directly contribute to improved device performance metrics documented in Table 1.
Polymerization-Induced Direct Photolithography
For crosslinking approaches, polymerization-induced direct photolithography has emerged as a powerful technique for high-resolution patterning of PQDs:
- Material Preparation: QDs are functionalized with photopolymerizable ligands or dispersed in a photopolymerizable matrix. Common reactive functional groups include alkenes, alkynes, and disulfides [26].
- Film Deposition: The QD-polymer composite solution is spin-coated into uniform thin films.
- Pattern Definition: UV exposure through a photomask initiates localized polymerization, creating crosslinked networks in exposed regions.
- Development: Unexposed regions are removed using appropriate solvents, leaving behind high-fidelity QD patterns.
The photochemical reactions driving this process typically involve radical polymerization, thiol-based coupling, cycloaddition, or ring-opening reactions that construct crosslinked polymer networks directly from QD films or QD-polymer composites [26]. This method provides exceptional spatial control, enables ambient processing conditions, and preserves QD optoelectronic properties by avoiding harsh processing chemicals.
BA-Based Ligand Exchange Protocols
The implementation of benzoic acid and its analogs in PQD systems follows specific experimental sequences:
- Ligand Solution Preparation: BA derivatives are dissolved in appropriate solvents (typically ethyl acetate or similar polar solvents) at optimized concentrations.
- Film Treatment: Pre-deposited PQD films are exposed to BA solutions through spin-coating, dipping, or vapor-assisted processes.
- Removal of Byproducts: Excess ligands and reaction byproducts are removed through washing with mild solvents.
- Thermal Annealing: Controlled thermal treatment enhances ligand binding and film consolidation.
The chemical specificity of BA is crucial to its function—the molecule contains both hydrophilic (carboxylic group) and hydrophobic (benzene ring) components, with the balance between these components determining its interaction with the QD surface and surrounding matrix [27]. Modifications to the benzene ring or carboxylic group significantly alter this balance and affect the final material properties.
Mechanism Visualization and Workflow Integration
Diagram 1: Evolutionary workflow of ligand engineering strategies from conventional to synergistic approaches
Diagram 2: Mechanistic pathways of synergistic ligand and crosslinking strategies
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key research reagents and their functions in ligand engineering studies
| Reagent/Material | Chemical Function | Role in PQD Device Fabrication | Considerations for Use |
|---|---|---|---|
| Benzoic Acid (BA) & Analogs | Short-chain ligand with benzene-carboxylic structure | Replace long-chain ligands to enhance charge transport | Chemical specificity crucial; substitutions on benzene ring affect function [27] |
| Phenethylammonium Iodide (PEAI) | Conjugated short-chain ligand with aromatic group | Layer-by-layer ligand exchange for enhanced inter-dot coupling and passivation [8] | PEA+ ion provides hydrophobic protection; enables balanced carrier injection |
| Oleic Acid (OA) | Long-chain carboxylic acid (C18:1) | Primary synthesis ligand for colloidal stability | Must be effectively removed/replaced for device integration |
| Oleylamine (OAm) | Long-chain primary amine (C18:1) | Co-ligand during synthesis, binds to surface sites | Dynamic binding requires multiple exchange cycles for complete replacement |
| Formamidinium Iodide (FAI) | Short organic cation salt | Conventional post-treatment for ligand exchange | May induce phase changes if treatment over-extended [8] |
| Photopolymerizable Monomers | Multi-functional acrylates, thiols, etc. | Enable crosslinking networks via photopolymerization | Compatibility with QD surface chemistry essential [26] |
| Tetramethoxysilane (TMOS) | Silicon alkoxide precursor | SiO2 coating for passivation and stability enhancement [28] | Hydrolysis forms protective layer, improves photoresist compatibility |
Comparative Analysis and Research Implications
The experimental data and performance metrics compiled in this comparison guide demonstrate clear advantages for synergistic ligand strategies over conventional approaches. The PEAI-LBL method achieves a remarkable PCE of 14.18% with high open-circuit voltage (1.23 V) in CsPbI3 PQD solar cells, while simultaneously enabling electroluminescent functionality [8]. This represents a significant improvement over BA-only approaches (5.46% PCE) and substantially exceeds the performance of early OA/OAM-based systems (10.77% PCE).
The mechanistic advantage of combined BA-derived and crosslinking approaches lies in their multi-functional addressing of key challenges in PQD devices. While BA and PEAI short ligands primarily enhance charge transport and provide better defect passivation, the crosslinking components contribute mechanical robustness and environmental stability. When integrated strategically, these approaches enable devices that maintain high performance under operational stresses, including high-humidity environments where conventional devices rapidly degrade [8] [28].
For research professionals pursuing next-generation PQD applications, the implications are clear: the future lies in multi-component, rationally designed ligand systems rather than single-ligand solutions. The most promising developmental trajectory involves:
- Selecting short ligands with specific functional groups that target both conductivity and stability enhancement
- Implementing processing methodologies (like LBL) that ensure uniform ligand exchange throughout the film thickness
- Incorporating crosslinking strategies that enable both patterning capability and environmental robustness
- Balancing the various components to maintain optimal charge transport while minimizing defect states
This comparative analysis provides both a performance benchmark and methodological framework for advancing PQD device research toward commercial viability, particularly in demanding applications such as bifunctional optoelectronic devices, high-resolution displays, and stable photovoltaic systems.
The pursuit of next-generation display technology has positioned micro-light-emitting diodes (micro-LEDs) as a leading candidate, offering unparalleled advantages in power consumption, contrast ratio, and operational lifetime [29]. However, achieving cost-effective and efficient full-color emission remains a significant challenge for widespread commercialization. Color conversion layers (CCLs) utilizing perovskite quantum dots (PQDs) have emerged as a promising solution, potentially eliminating the need for complex mass transfer of individual red, green, and blue micro-LEDs [30] [31]. The performance and stability of these PQDs are critically dependent on their surface chemistry, where ligand engineering plays a pivotal role. This case study provides a performance comparison of traditional oleic acid/oleylamine (OA/OAm) ligands versus advanced alternatives, including benzoic acid derivatives, in the context of fabricating full-color micro-LED displays.
The Critical Role of Ligands in PQD Performance
Ligands are molecules attached to the surface of PQDs that facilitate nucleation, growth, and passivation of surface defects during synthesis [1]. Effective passivation is essential for achieving high photoluminescence quantum yield (PLQY) and maintaining stability against environmental factors such as humidity, temperature, and light exposure [1]. The intrinsic ionic crystal structure of CsPbX3 PQDs makes them particularly susceptible to degradation, which is often exacerbated by weak or dynamic ligand binding [32] [1].
Traditional synthesis methods predominantly use a combination of OA and OAm. These ligands are highly dynamic and exhibit loose binding to the PQD surface, often leading to detachment. This results in surface defects, uncontrolled growth, and ultimately, poor stability and reduced PLQY [32]. This instability represents a major obstacle for practical optoelectronic devices, including micro-LED displays.
Performance Comparison: OA/OAm vs. Advanced Ligands
Quantitative Performance Metrics
The table below summarizes key performance metrics for PQDs capped with different ligand systems, highlighting the limitations of OA/OAm and the enhancements achieved through advanced ligand engineering.
Table 1: Performance Comparison of PQDs with Different Ligand Systems
| Ligand System | Photoluminescence Quantum Yield (PLQY) | Stability Retention | Key Stability Metrics | Binding Energy (DFT Calculation) |
|---|---|---|---|---|
| OA/OAm (Traditional) | Not specified (Low due to defect states) | Poor | Highly sensitive to UV light, high temperature, and polar solvents [32] | Not specified (Weak dynamic binding) [32] |
| THAB (Bifunctional) | 61% [32] | High | Superior stability against UV light, high temperature, and polar solvents [32] | -2.779 eV [32] |
| Silane Ligands | Maintains 80% of original PL after 1 month [30] | High (Ambient atmospheric stability) | Excellent dispersibility in siloxane matrix, encapsulated via sol-gel condensation [30] | N/A |
Analysis of Comparative Data
The data demonstrates a clear performance advantage for PQDs passivated with advanced ligands. The THAB ligand system shows a high PLQY of 61%, attributed to effective defect passivation, and exhibits robust stability against multiple environmental stressors [32]. Theoretical calculations confirm a significantly larger binding energy for THAB (-2.779 eV) compared to other ligands, explaining its reduced detachment and enhanced stability [32].
Similarly, silane-based ligands used in a different study enabled the fabrication of a full-color micro-LED display that maintained 80% of its original PL efficiency after one month in ambient conditions [30]. This was achieved through a ligand exchange process that provided high dispersibility in a siloxane matrix and subsequent encapsulation via a sol-gel condensation reaction, showcasing a viable path to industrial application.
Experimental Protocols for Ligand Engineering
Synthesis of Oleate-Capped PQDs (Baseline)
The standard synthesis of oleate-capped CsPbBr3 PQDs (oleate-PQDs) typically involves the hot-injection method [30] [1]. Precursors such as Cs₂CO₃, PbBr₂, and organic solvents like 1-octadecene (ODE) are prepared. OA and OAm act as the primary ligands. The Cs-oleate solution is rapidly injected into a heated Pb-oleate solution, initiating nucleation and growth. The resulting nanocrystals are then purified through centrifugation with anti-solvents like methyl acetate to remove unreacted precursors and excess ligands [30].
Ligand Exchange for Advanced Passivation
To overcome the limitations of OA/OAm, post-synthetic ligand exchange is a common strategy. The following workflow details a specific protocol for integrating PQDs into a robust, photo-patternable siloxane composite for display fabrication [30].
Diagram 1: Experimental workflow for creating photo-patternable PQD/siloxane composite [30].
Key Steps Explained:
- Ligand Exchange Reaction: The original oleate ligands on pre-synthesized PQDs are replaced with silane-based ligands (e.g., (3-mercaptopropyl)trimethoxysilane) in the presence of methylammonium bromide (MABr). The halide salt helps activate the PQD surface for this exchange [30].
- Purification and Dispersion: Unreacted silane and byproducts are removed via centrifugation, yielding ligand-exchanged PQDs (silane-PQDs) that are re-dispersed in toluene [30].
- Composite Formation and Photocuring: The silane-PQDs are mixed with siloxane resins. A photocuring catalyst is added, and the mixture is exposed to UV light to form a cross-linked, solid composite. This process enables the photolithographic patterning of the CCLs necessary for high-resolution displays [30].
Research Reagent Solutions for PQD-Based Displays
The table below lists essential materials and their functions for research in ligand-engineered PQDs for micro-LED displays.
Table 2: Key Research Reagent Solutions for PQD Application in Displays
| Reagent/Material | Function in Research & Development |
|---|---|
| Cesium Carbonate (Cs₂CO₃), Lead Bromide (PbBr₂) | High-purity inorganic precursors for the synthesis of CsPbBr3 PQD cores [30] [32]. |
| Oleic Acid (OA) & Oleylamine (OAm) | Traditional ligands used in initial synthesis; dynamic binding often requires subsequent exchange [30] [32] [1]. |
| (3-mercaptopropyl)trimethoxysilane | A silane ligand used in exchange processes to enhance stability and enable integration into a photo-patternable polymer matrix [30]. |
| Hexadecyltrimethylammonium tetrafluoroborate (THAB) | A bifunctional capping ligand that provides strong, non-dynamic binding to the PQD surface, boosting PLQY and stability [32]. |
| Methylammonium Bromide (MABr) | A halide salt used in surface activation and ligand exchange processes to improve the efficiency of ligand binding [30]. |
| Methacrylate Oligosiloxane Resins | A photocurable matrix material that encapsulates ligand-exchanged PQDs, allowing for the fabrication of robust, patterned color conversion layers [30]. |
This performance comparison clearly demonstrates that moving beyond traditional OA/OAm ligands is imperative for developing PQD-based micro-LED displays suitable for commercial applications. While OA/OAm are useful for initial synthesis, their dynamic nature and weak binding lead to insufficient stability. Advanced ligand systems, such as bifunctional THAB and silane compounds, provide a paradigm shift. They offer stronger, more stable passivation of the PQD surface, which directly translates to higher PLQY and, crucially, robust long-term operational stability. Ligand engineering, therefore, is not merely a supplementary technique but a core strategy in harnessing the full potential of perovskite quantum dots for the future of high-performance, full-color micro-LED displays.
Troubleshooting Instability and Optimizing Charge Transport in PQD Devices
Identifying and Mitigating Surface Defects and Vacancy Formation from Ligand Detachment
Perovskite quantum dots (PQDs) have emerged as a leading semiconductor nanomaterial for next-generation optoelectronics, offering exceptional properties including high photoluminescence quantum yield (PLQY), size-tunable bandgaps, and superior defect tolerance [33]. The performance and stability of these materials are intrinsically linked to their surface chemistry, particularly the organic ligand shell that encapsulates each nanocrystal. During synthesis, insulating long-chain ligands—primarily oleic acid (OA) and oleylamine (OAm)—are essential for controlling nanocrystal growth, ensuring colloidal stability, and providing initial surface passivation [7] [33]. However, the dynamic and ionic nature of perovskite crystals renders this ligand shell uniquely labile. The conventional purification and film-processing stages often trigger ligand detachment, which directly creates surface halide vacancies and uncoordinated lead ions [34] [35]. These defects act as non-radiative recombination centers, severely compromising charge transport in solar cells and electroluminescent efficiency in light-emitting diodes (LEDs) [34] [8].
This guide objectively compares the performance of traditional OA/OAm ligand systems against innovative alternatives, such as benzoic acid, within PQD devices. We directly analyze experimental data on photovoltaic parameters, luminescent properties, and operational stability, providing researchers with a quantitative framework for ligand selection. By examining detailed protocols for ligand exchange and surface management, this article aims to equip scientists with validated strategies to suppress defect formation and elevate device performance.
Performance Comparison of OA/OAm vs. Advanced Ligand Systems
The following tables consolidate quantitative data from recent studies, enabling a direct comparison of device performance metrics achieved with different ligand management strategies.
Table 1: Photovoltaic Performance of PQD Solar Cells with Different Ligand Systems
| Ligand System | PQD Material | Device Type | PCE (%) | VOC (V) | JSC (mA cm⁻²) | FF (%) | Reference |
|---|---|---|---|---|---|---|---|
| Conventional OA/OAm | CsPbI₃ | Rigid | <11 (Typical) | - | - | - | [6] |
| Sequential DPA + BA | FAPbI₃ | Flexible (0.06 cm²) | 12.13 | - | - | - | [7] |
| Sequential DPA + BA | FAPbI₃ | Rigid | 14.27 | - | - | - | [7] |
| Choline in 2-pentanol | CsPbI₃ | Rigid | 16.53 | - | - | - | [36] |
| PEAI Layer-by-Layer | CsPbI₃ | Rigid | 14.18 | 1.23 | - | - | [8] |
| Binary Size Blending | CsPbI₃ | Rigid | 14.42 | 1.19 | 17.08 | 71.12 | [6] |
Table 2: Optoluminescent Performance and Stability of PQD Films and LEDs
| Ligand Strategy | PQD Material | Application | Key Performance Metric | Value | Reference |
|---|---|---|---|---|---|
| Conventional OA/OAM (Standard Purification) | Mixed-Halide | Blue LED | External Quantum Efficiency (EQE) | Low (Baseline) | [34] [35] |
| In-Situ Passivation (ETFA) | Mixed-Halide | Blue LED (463 nm) | EQE / Luminance | 4.14% / 1035 cd m⁻² | [34] |
| Ligand-Assisted Purification | Mixed-Halide | Green/Red Emissive Films | PLQY | Near-Unity (≈100%) | [35] |
| Film-State Ligand Exchange (FLE) | CsPbBr₃ | Crosslinked Green LED | EQE / Luminance | 14.7% / ≈25,400 cd m⁻² | [37] |
| OA/OAm (Control) | FAPbI₃ | Flexible Solar Cell | PCE Retention (100 bends, 7mm radius) | ~90% | [7] |
Analysis of Comparative Data
- Efficiency Gains: Replacing native OA/OAm ligands with short, conjugated molecules like benzoic acid (BA) and phenethylammonium iodide (PEAI) consistently enhances Power Conversion Efficiency (PCE). This is attributed to improved inter-dot electronic coupling and reduced charge transport barriers [7] [8]. The record PCE of 16.53% for inorganic CsPbI₃ PQD solar cells was achieved using a tailored solvent-mediated ligand exchange, which maximized the removal of insulating oleylamine without introducing new defects [36].
- Luminescence and Defect Passivation: Strategies that proactively passivate surfaces during processing, such as in-situ passivation and ligand-assisted purification, dramatically improve Photoluminescence Quantum Yield (PLQY). Recovering near-unity PLQY is direct evidence of superior suppression of non-radiative recombination pathways caused by vacancies [34] [35].
- Mechanical and Operational Stability: Ligand-capped PQDs demonstrate intrinsic mechanical robustness, with flexible devices retaining ~90% of their initial PCE after repeated bending [7]. Furthermore, hydrophobic ligands like PEA⁺ enhance resistance to moisture, improving the device's environmental stability [8].
Experimental Protocols for Ligand Exchange and Defect Mitigation
To achieve the performance benchmarks outlined above, specific experimental methodologies have been developed. Below are detailed protocols for key ligand engineering strategies.
Sequential Ligand Exchange with DPA and BA
This one-step fabrication technique simplifies the production of high-performance flexible PQD solar cells [7].
- Synthesis: FAPbI₃ PQDs are synthesized via a standard hot-injection method, resulting in dots capped with native OA/OAm ligands.
- Ligand Exchange Solution: A solution containing dipropylamine (DPA) and benzoic acid (BA) in a solvent like n-hexane or n-octane is prepared.
- Procedure: The as-synthesized PQD crude solution is treated with the DPA/BA solution. DPA first acts to remove the long-chain OA/OAm ligands, improving electronic conductivity. Subsequently, the short-chain BA ligand passivates the newly exposed surface defects and coordinates with the nanocrystal surface.
- Film Fabrication: The treated PQD solution is then directly used in a one-step spin-coating process to form the photovoltaic film, eliminating the need for layer-by-layer processing.
Layer-by-Layer (LBL) Solid-State Ligand Exchange
This method offers precise control over the surface chemistry of each deposited layer, ideal for electroluminescent solar cells [8].
- Substrate Preparation: A clean conductive substrate (e.g., FTO or ITO) is used.
- PQD Layer Deposition: A thin layer of CsPbI₃ PQDs (with native OA/OAm ligands) is spin-coated onto the substrate.
- Ligand Treatment: Immediately after deposition, the film is treated by spin-coating a solution of short-chain ligands, specifically Phenethylammonium Iodide (PEAI) in ethyl acetate (EtOAc). Methyl acetate (MeOAc) can be used to assist in the initial removal of OA.
- Washing: The film is rinsed with EtOAc to remove the displaced long-chain ligands and residual salts.
- Repetition: The cycle (spin-coat PQDs → treat with PEAI → wash) is repeated 3-5 times to build a thick, electronically coupled, and well-passivated solid film.
In-Situ Passivation During Purification
This strategy tackles defect formation at the purification stage, which is critical for high-color-purity light emission [34] [35].
- Synthesis: Mixed-halide perovskite NCs are synthesized via a hot-injection method.
- Modified Purification: Instead of using plain anti-solvent, ethyl trifluoroacetate (ETFA) is employed. During the anti-solvent addition, ETFA hydrolyzes to form trifluoroacetic acid (TFA).
- Mechanism: The TFA group strongly interacts with uncoordinated Pb²⁺ ions, anchoring Br⁻ to the NC surface. Concurrently, it induces trifluoroacetylation of OAm, weakening its interaction with Br⁻ and facilitating its mild detachment without creating halide vacancies.
- Ligand-Assisted Variation: An alternative approach involves adding a small, controlled amount of OA and OAm to the crude NC solution before introducing the anti-solvent (e.g., tert-butanol). This supplements the ligand shell during the stressful precipitation process, preventing excessive detachment and maintaining a high PLQY [35].
Visualization of Workflows and Mechanisms
The following diagrams illustrate the core experimental workflows and logical relationships of the described strategies.
Sequential Ligand Exchange Process
Diagram 1: Sequential ligand exchange workflow.
Ligand Impact on Defects and Performance
Diagram 2: Ligand impact on defects and performance.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for PQD Ligand Engineering
| Reagent Name | Chemical Function | Role in Ligand Management & Defect Mitigation |
|---|---|---|
| Oleic Acid (OA) / Oleylamine (OAm) | Long-chain carboxylic acid/amine | Standard synthesis ligands providing colloidal stability but impeding charge transport. The primary source of surface defects upon detachment. |
| Benzoic Acid (BA) | Short-chain aromatic carboxylic acid | Replaces OA, passivates uncoordinated Pb²⁺ sites, enhances electronic coupling, and improves stability via conjugated structure [7]. |
| Dipropylamine (DPA) | Short-chain amine | Facilitates the removal of long-chain OAm ligands during sequential exchange processes [7]. |
| Phenethylammonium Iodide (PEAI) | Short-chain ammonium salt | Used in LBL exchange; the PEA⁺ cation replaces OAm, passivating defects and improving charge transport, while the I⁻ anion helps fill halide vacancies [8]. |
| Ethyl Trifluoroacetate (ETFA) | Ester precursor to trifluoroacetic acid | Hydrolyzes during purification to TFA, which passivates Pb²⁺ and weakens OAm binding, mitigating halide vacancy formation in-situ [34]. |
| Formamidinium Iodide (FAI) | Short-chain ammonium salt | Common for post-treatment ligand exchange, providing I⁻ to fill vacancies and FA⁺ to passivate surfaces [8]. |
| 2-pentanol | Protic solvent | Tailored solvent for ligand exchange; its appropriate dielectric constant and acidity maximize ligand removal without introducing halogen vacancies [36]. |
Perovskite quantum dots (PQDs) have emerged as a transformative class of semiconductor nanomaterials with exceptional optoelectronic properties, including high photoluminescence quantum yields (PLQYs) of 50-90%, narrow emission spectra, and widely tunable bandgaps [38]. The performance of PQDs in device configurations—whether in photovoltaics, light-emitting diodes (LEDs), or photodetectors—is critically dependent on efficient charge transport between individual quantum dots. However, this charge transport is fundamentally constrained by the interparticle distance, which is predominantly governed by the surface ligand shell [33] [39].
Long-chain, insulating ligands, primarily oleic acid (OA) and oleylamine (OAm), are indispensable in the colloidal synthesis of high-quality PQDs. They passivate surface defects, control crystal growth, and prevent aggregation [40] [1]. Nevertheless, these same ligands form a permanent insulating barrier between PQDs in solid films, limiting electronic coupling and compromising device performance [9] [41]. This comparison guide objectively analyzes the performance of traditional OA/OAm ligands against the emerging strategy of using benzoic acid derivatives, focusing on their efficacy in shortening the interparticle distance to overcome insulating barriers.
Ligand Chemistry and Function: A Comparative Basis
Understanding the fundamental chemistry of surface ligands is crucial for evaluating their impact on interparticle distance. Ligands interact with the ionic surface of lead halide PQDs (e.g., CsPbX3, where X = Cl, Br, I) through specific coordination modes, classified under the Covalent Bond Classification (CBC) as L-type (Lewis base) and X-type (anionic) ligands [39].
Table 1: Fundamental Classification and Properties of Common PQD Ligands
| Ligand Type | Binding Classification | Common Examples | Primary Function | Impact on Interparticle Distance |
|---|---|---|---|---|
| Long-Chain Alkyl Acids | X-type (anionic) | Oleic Acid (OA) | Passivates surface Pb atoms, provides colloidal stability | Creates a wide insulating barrier (~1-2 nm) |
| Long-Chain Alkyl Amines | L-type (Lewis base) | Oleylamine (OAm) | Binds to halide vacancies, aids crystallization | Creates a wide insulating barrier (~1-2 nm) |
| Short-Chain Aromatic Acids | X-type (anionic) | Benzoic Acid, Methyl Benzoate (hydrolyzed) | Strong surface binding, conductive capping | Creates a narrow, more conductive barrier (<1 nm) |
The conventional OA and OAm system creates a dynamic equilibrium on the PQD surface. Their long alkyl chains (C18) provide excellent steric hindrance in solution but result in an interparticle distance of several angstroms to nanometers in solid films. This large separation acts as a potential barrier for charge carriers, making inter-dot hopping inefficient [9] [1]. In contrast, short-chain ligands like benzoic acid and its derivatives offer a compact molecular structure. The benzene ring itself can facilitate π-π stacking, potentially offering a more conductive pathway between dots compared to insulating alkyl chains [9] [40].
Experimental Protocols for Ligand Exchange and Performance Evaluation
Alkali-Augmented Ligand Exchange with Methyl Benzoate
A state-of-the-art methodology for replacing pristine OA/OAm ligands with benzoate ligands is the Alkali-Augmented Antisolvent Hydrolysis (AAAH) strategy, developed to enhance conductive capping [9].
Detailed Protocol:
- PQD Film Fabrication: Spin-coat a layer of hybrid FA0.47Cs0.53PbI3 PQDs (synthesized via hot-injection or LARP methods) onto a substrate to form a solid film.
- Antisolvent Preparation: Prepare a rinsing antisolvent by adding Potassium Hydroxide (KOH) to methyl benzoate (MeBz). The alkaline environment is critical, as it renders the hydrolysis of the ester thermodynamically spontaneous and lowers the reaction activation energy by approximately nine-fold.
- Interlayer Rinsing: While the PQD film is still wet, rinse it with the KOH/MeBz antisolvent. Under ambient humidity, the MeBz hydrolyzes into benzoic acid, which subsequently deprotonates to form benzoate anions (C6H5COO-).
- Ligand Exchange: These benzoate anions rapidly substitute the pristine, insulating oleate (OA-) ligands on the PQD surface. The strong coordination of the benzoate group to the Pb atoms on the surface provides effective passivation.
- Film Processing: Repeat the layer-by-layer deposition and rinsing process to build a thick film. A final post-treatment with short cationic ligands (e.g., formamidinium, FA+) may be applied to substitute the OAm+ ligands, further enhancing electronic coupling.
This protocol achieves a denser and more conductive capping layer, with up to twice the conventional amount of short conductive ligands compared to treatments with neat ester antisolvents [9].
Direct Ligand Engineering with Benzoic Acid
An alternative, simpler approach involves the post-synthetic treatment of pre-formed PQD films or solutions with short-chain ligands like benzoic acid.
Detailed Protocol:
- PQD Synthesis and Purification: Synthesize CsPbBr3 NCs using a standard hot-injection or room-temperature method with OA and OAm. Purify the NCs to remove excess free ligands and precursor residues.
- Ligand Solution Preparation: Dissolve benzoic acid in a solvent like 2-propanol or a mixture of n-hexane and butanol, ensuring the solvent polarity is mild enough to prevent PQD degradation.
- Treatment Process: Introduce the benzoic acid solution to a suspension of purified PQDs or directly onto a spin-coated PQD film. Incubate for a specific duration (seconds to minutes) to allow ligand exchange.
- Purification and Film Formation: Remove the excess benzoic acid and displaced long-chain ligands through centrifugation or washing. The resulting PQDs, now capped with benzoate, can be redispersed and processed into solid films.
This method leverages the stronger binding affinity of the benzoate group to undercoordinated Pb2+ sites, effectively replacing the dynamically bound OA and passivating surface defects [40].
Performance Comparison: OA/OAm vs. Benzoic Acid Ligands
The efficacy of ligand engineering strategies is quantitatively evaluated through key performance metrics in both material properties and device performance.
Table 2: Quantitative Performance Comparison of Ligand Strategies
| Performance Metric | OA/OAm Ligands (Conventional) | Benzoic Acid Ligands (Engineered) | Experimental Context |
|---|---|---|---|
| Photoluminescence Quantum Yield (PLQY) | Up to 94.4% [40] | Maintains or slightly reduces from initial high value | CsPbBr3 NCs in solution/film |
| Film Conductivity | Low; limited by long-chain insulation | Significantly enhanced | Deduced from PV device performance [9] |
| Solar Cell Power Conversion Efficiency (PCE) | ~10% (Baseline) | Certified 18.3% (Champion device) [9] | FA0.47Cs0.53PbI3 PQD Solar Cells |
| Steady-State PCE | N/A | 17.85% [9] | FA0.47Cs0.53PbI3 PQD Solar Cells |
| Trap-State Density | High | Fewer trap-states, suppressed non-radiative recombination [9] | Deduced from PL and device analysis |
| Environmental Stability | Moderate; degrades over days/weeks | Enhanced storage and operational stability [9] | Film & device under operating conditions |
The data demonstrates a clear trade-off that is masterfully managed by benzoic acid ligands. While OA/OAM systems can achieve near-unity PLQYs in solution, their insulating nature severely limits electrical transport in devices. Benzoate ligands, in contrast, maintain high luminescence while dramatically improving conductivity, as evidenced by the near-doubling of solar cell efficiency. This is directly attributable to reduced interparticle distance and stronger surface binding, which minimizes trap-states and facilitates charge extraction.
Underlying Mechanisms: Visualizing the Ligand Exchange Process
The following diagram illustrates the strategic replacement of long-chain insulating ligands with short-chain benzoate ligands, which is the core mechanism for reducing interparticle distance.
This process transforms the film from a state of poor interdot electronic communication to one with enhanced charge transport capabilities. The compact benzoate ligand shortens the tunneling distance between adjacent PQDs and its aromatic structure may facilitate better orbital overlap, thereby creating a more favorable pathway for charge carriers [9] [40].
The Scientist's Toolkit: Essential Reagents for Ligand Engineering
Successful experimental execution in PQD ligand engineering requires specific, high-purity materials. The following table details key reagents and their functions.
Table 3: Essential Research Reagents for PQD Ligand Exchange Studies
| Research Reagent | Function/Application | Key Consideration for Use |
|---|---|---|
| Oleic Acid (OA) | Standard X-type ligand for synthesis & surface passivation | Requires high purity (≥90%); dynamic binding leads to instability [1] |
| Oleylamine (OAm) | Standard L-type ligand for synthesis & passivating halide vacancies | Often used with OA; acid-base equilibrium affects coverage [39] |
| Benzoic Acid | Short-chain X-type ligand for post-synthetic exchange | Strong binding to Pb2+; reduces interparticle distance [40] |
| Methyl Benzoate | Ester precursor for in situ hydrolysis to benzoic acid | Used in antisolvent rinsing; requires alkaline/humid conditions [9] |
| Potassium Hydroxide (KOH) | Alkali source to facilitate ester hydrolysis | Critical for AAAH strategy; concentration must be optimized [9] |
| Lead Acetate / Halide Salts | Common Pb2+ and halide precursors for PQD synthesis | High purity (≥99.9%) essential for optimal optoelectronic properties [40] |
| Trimethylsilyl Halides | Highly reactive halide precursors for room-temperature synthesis | Enables synthesis without inert atmosphere [40] |
| Formamidinium Iodide | Cation source for hybrid A-site PQDs and A-site ligand exchange | Used in post-treatment to replace OAm+ and improve coupling [9] |
The strategic shortening of interparticle distance through ligand engineering is a decisive factor for advancing PQD technology. While OA and OAm ligands remain the cornerstone of high-quality PQD synthesis, their inherent insulating properties create a significant bottleneck for device performance. The systematic replacement of these long-chain ligands with compact, strongly-bound alternatives like benzoic acid and its derivatives directly addresses this challenge. Quantitative comparisons confirm that this approach yields substantial improvements in film conductivity, trap-state reduction, and ultimate device efficiency, as dramatically evidenced in solar cells where certified efficiencies over 18% have been achieved. Future research will likely focus on refining the ligand exchange processes for minimal defect introduction and exploring novel multifunctional ligand designs that combine short length with enhanced stability, pushing the performance of PQD-based optoelectronics closer to their theoretical limits.
Optimizing Antisolvent Polarity and Alkaline Treatments for Efficient OA-to-BA Exchange
Perovskite quantum dots (PQDs) have emerged as promising materials for next-generation photovoltaics and optoelectronics due to their tunable bandgaps, high absorption coefficients, and exceptional defect tolerance. [42] The surface chemistry of these nanocrystals is predominantly governed by organic ligands that passivate surface sites and determine colloidal stability. Native ligands like oleic acid (OA) and oleylamine (OAm) provide effective stabilization during synthesis but form insulating layers that hinder charge transport in solid-state devices. Consequently, exchanging these long-chain insulating ligands for shorter conductive alternatives represents a critical processing step for enhancing device performance.
Among shorter ligands, benzoic acid (BA) and its derivatives have demonstrated exceptional potential for creating conductive PQD films. The exchange from OA to BA ligands presents specific challenges, requiring precise optimization of antisolvent polarity and chemical environment to maximize passivation and device performance. This review comprehensively compares OA/OAm versus benzoic acid ligand systems, examining experimental protocols, performance outcomes, and underlying mechanisms that govern efficient ligand exchange processes.
Fundamental Principles of Ligand Chemistry in Perovskite Nanocrystals
Roles and Limitations of Native OA/OAm Ligands
The conventional synthesis of perovskite quantum dots relies on a dynamic equilibrium of oleic acid (OA) and oleylamine (OAm) ligands that provide steric stabilization and passivate surface defects. The carboxylic acid group (-COOH) of OA coordinates with undercoordinated Pb²⁺ sites, while the amine (-NH₂) group of OAm interacts with halide vacancies. These long-chain (C18) hydrocarbons enable excellent colloidal stability in non-polar solvents but create significant barriers to inter-dot charge transport in fabricated films due to their insulating nature. [40]
The inherent limitation of OA/OAm systems lies in their dynamic binding behavior. The protonation-deprotonation equilibrium between OA and OAm molecules leads to fluctuating ligand coverage on PQD surfaces, ultimately causing increased surface defects and nanocrystal aggregation during storage and processing. This instability is particularly pronounced in room-temperature synthesized NCs, which typically contain higher defect densities. [40]
Advantages of Benzoic Acid-Based Ligands
Benzoic acid derivatives offer distinct advantages for PQD surface engineering. The aromatic structure provides robust π-π interactions that enhance binding stability, while the shorter molecular length reduces inter-dot spacing for improved charge transport. The strong complexation between the carboxylic acid group and Pb²⁺ ions forms more stable coordination bonds compared to OA, dramatically reducing surface defects and associated non-radiative recombination centers. [43]
Research has demonstrated that BA-based ligands exhibit a unique "halide-equivalent" effect that effectively compensates for halide vacancies, thereby suppressing halide vacancy-mediated non-radiative recombination pathways. This defect-passivation capability is especially valuable for achieving high photoluminescence quantum yields (PLQYs) and enhanced photovoltaic performance. [40]
Experimental Approaches for OA-to-BA Ligand Exchange
Alkaline-Enhanced Antisolvent Hydrolysis
The Alkali-Augmented Antisolvent Hydrolysis (AAAH) strategy represents a significant advancement in ligand exchange methodology. This approach addresses the fundamental limitation of conventional ester antisolvents, which hydrolyze inefficiently under ambient conditions to generate target ligands. [9]
Table 1: Key Components of Alkaline-Enhanced Exchange Protocol
| Component | Role in Ligand Exchange | Optimization Parameters |
|---|---|---|
| Methyl Benzoate (MeBz) | Preferred antisolvent with suitable polarity | Balanced hydrolysis probability and rapid evaporation |
| Potassium Hydroxide (KOH) | Creates alkaline environment to facilitate hydrolysis | Concentration critical to prevent structural damage |
| Controlled Humidity | Provides water molecules for hydrolysis | ~30% relative humidity optimal |
| 2-Pentanol | Protic solvent for short ligand dissolution | Appropriate dielectric constant and acidity |
The AAAH methodology establishes an alkaline environment that fundamentally alters the thermodynamics and kinetics of ester hydrolysis. Theoretical calculations reveal that this environment renders ester hydrolysis thermodynamically spontaneous and lowers reaction activation energy by approximately 9-fold. Through tailoring potassium hydroxide coupled with methyl benzoate antisolvent for interlayer rinsing of PQD solids, the assembled light-absorbing layers exhibit fewer trap-states, homogeneous orientations, and minimal particle agglomerations. [9]
The experimental protocol involves:
- Preparing PQD solid films via layer-by-layer deposition
- Rinsing with methyl benzoate antisolvent containing controlled KOH concentrations
- Regulating environmental humidity to ~30% to provide water molecules for hydrolysis
- Subsequent post-treatment with short cationic ligands if required
This approach enables the substitution of pristine insulating oleate ligands with up to twice the conventional amount of hydrolyzed conductive counterparts, significantly enhancing inter-dot charge transport. [9]
Solvent-Mediated Ligand Exchange
An alternative approach focuses on tailoring solvent properties to optimize ligand exchange efficiency. This method screens solvents based on dielectric constant and acidity to maximize the removal of insulating ligands without introducing halogen vacancy defects. [36]
Protic 2-pentanol has been identified as a superior solvent for mediating ligand exchange due to its appropriate dielectric constant and acidity. The protocol employs tailored short choline ligands dissolved in 2-pentanol for the post-treatment of PQD solids. This solvent environment sufficiently dissolves both the incoming short ligands and the displaced long-chain ligands, facilitating a more complete exchange process while maintaining PQD structural integrity. [36]
The key advantage of this approach lies in its ability to maximize ligand exchange efficiency without damaging the perovskite crystal structure. The resulting PQD films demonstrate improved charge carrier transport and enhanced surface defect passivation, contributing to superior device performance. [36]
In Situ 2D Perovskite-like Ligand Engineering
A innovative strategy employing 2D-perovskite-like (BA)₂PbI₄ ligands has demonstrated exceptional passivation capabilities for lead sulfide quantum dots, with implications for perovskite systems. This approach forms a thin shell of BA⁺ and I⁻ ions on the quantum dot surface via an in situ solution-phase ligand-exchange strategy. [24]
The treatment enables strong inward coordination, particularly on challenging non-polar <100> facets, effectively reducing surface defect density and preventing quantum dot aggregation and fusion. The robustness of the BA⁺-rich surface provides excellent ambient stability attributed to the hydrophobic nature of the benzylammonium moiety. [24]
Comparative Performance Analysis
Photovoltaic Performance Metrics
The efficacy of OA-to-BA ligand exchange strategies is quantitatively demonstrated through photovoltaic performance metrics. The Alkali-Augmented Antisolvent Hydrolysis approach has yielded a certified efficiency of 18.3% for hybrid FA₀.₄₇Cs₀.₅₃PbI₃ PQD solar cells, the highest value among published PQD solar cell reports at the time of publication. [9]
Table 2: Performance Comparison of Ligand Systems in PQD Photovoltaics
| Ligand System | Exchange Method | PCE (%) | Stability Retention | Key Advantages |
|---|---|---|---|---|
| OA/OAm (Native) | N/A | <12% | Rapid degradation | Excellent colloidal stability |
| BA-based ligands | Alkaline-enhanced hydrolysis | 18.3 (certified) | Enhanced operational stability | High conductivity, defect passivation |
| BA-based ligands | Solvent-mediated exchange | 16.53 | Improved ambient stability | Balanced removal/binding dynamics |
| 2D (BA)₂PbI₄ | In situ solution-phase | 13.1 (PbS CQDs) | Excellent ambient stability | Robust shell formation, facet-specific passivation |
The performance enhancements stem from multiple factors: fewer trap-states, homogeneous crystallographic orientations, minimal PQD agglomerations, and favorable energy level positions. These improvements collectively suppress trap-assisted recombination and facilitate charge extraction within PQDSCs. [9]
Optoelectronic Properties and Stability
Beyond photovoltaic performance, BA-based ligand systems demonstrate superior optoelectronic properties and enhanced stability compared to conventional OA/OAM systems.
Photoluminescence Quantum Yield (PLQY): BA-treated PQDs achieve near-unity PLQY values, critical for both lighting applications and photovoltaic performance. The robust surface passivation significantly reduces non-radiative recombination pathways. [35]
Environmental Stability: The hydrophobic benzene ring in BA ligands enhances moisture resistance compared to aliphatic OA chains. Devices incorporating BA-rich surfaces exhibit excellent ambient stability, maintaining >90% of initial PCE after 15 days under ambient conditions. [24] [42]
Thermal Stability: BA-capped quantum dots demonstrate significantly enhanced thermal stability compared to conventional ligand systems. This attribute is particularly valuable for device operation under real-world conditions where temperature fluctuations are inevitable. [24]
Analytical Techniques for Characterization
Surface Chemistry Analysis
Comprehensive characterization of successful OA-to-BA exchange requires multiple analytical techniques:
- Nuclear Magnetic Resonance (NMR): Quantifies ligand density and confirms the replacement of OA protons with BA aromatic protons.
- Fourier-Transform Infrared Spectroscopy (FTIR): Identifies chemical bonding changes, particularly the shift in carboxylate stretching frequencies indicating stronger coordination to surface Pb²⁺ ions.
- X-ray Photoelectron Spectroscopy (XPS): Detects changes in surface elemental composition and chemical states, providing evidence of enhanced surface passivation.
Structural and Morphological Assessment
- Transmission Electron Microscopy (TEM): Reveals morphological changes, inter-dot spacing reduction, and preservation of nanocrystal integrity post-exchange.
- X-ray Diffraction (XRD): Confirms maintenance of perovskite crystal structure without formation of undesirable phases.
- Atomic Force Microscopy (AFM): Assesses film homogeneity and surface roughness, which directly impact device performance.
The strategic optimization of antisolvent polarity and alkaline treatments for OA-to-BA ligand exchange represents a significant advancement in perovskite quantum dot optoelectronics. The comparative analysis clearly demonstrates the superiority of BA-based ligand systems over conventional OA/OAm ligands in achieving enhanced photovoltaic performance and environmental stability.
Future research directions should focus on:
- Combinatorial Optimization: Simultaneous engineering of A-site cations and X-site ligands for synergistic effects.
- Green Chemistry Approaches: Developing environmentally benign solvents and reducing energy consumption during ligand exchange processes.
- Operando Characterization: Real-time monitoring of ligand exchange processes to better understand kinetic and thermodynamic factors.
- Machine Learning Applications: Predicting optimal ligand structures and exchange parameters for specific PQD compositions and target applications.
The integration of rational ligand design with optimized exchange protocols paves the way for next-generation PQD devices with performance metrics approaching their theoretical limits.
The Scientist's Toolkit
Table 3: Essential Research Reagents for OA-to-BA Ligand Exchange Studies
| Reagent | Function | Application Notes |
|---|---|---|
| Methyl Benzoate | Antisolvent for ligand exchange | Optimal polarity balance; hydrolysis generates benzoate ligands |
| Potassium Hydroxide | Alkaline catalyst | Enhances ester hydrolysis kinetics; concentration-critical |
| Benzoic Acid | Short conductive ligand | Strong Pb²⁺ coordination; aromatic system enhances stability |
| 2-Pentanol | Protic solvent | Superior ligand solubility; appropriate dielectric constant |
| Oleic Acid | Native reference ligand | Benchmark for comparison; requires complete removal |
| Oleylamine | Native co-ligand | Dynamic equilibrium with OA; impacts exchange efficiency |
| Methyl Acetate | Conventional antisolvent | Reference for ester-based exchange; less effective than MeBz |
| Tert-butanol | Washing anti-solvent | Purification without excessive ligand stripping |
Experimental Workflow Visualization
Addressing Phase Segregation and PQD Aggregation during Film Formation
The pursuit of high-performance perovskite quantum dot (PQD) optoelectronic devices, such as solar cells and LEDs, is critically dependent on the quality and stability of the active film. The dynamic ionic nature of perovskites and their high surface energy make PQDs susceptible to two major challenges during film formation: phase segregation and PQD aggregation. Phase segregation, the undesirable separation of halide compositions under operational stressors, degrades the optoelectronic properties of the film. Concurrently, PQD aggregation, often driven by the loss of surface ligands during processing, creates defective interfaces and impedes charge transport.
The choice of surface ligands is paramount in mitigating these issues. Traditional long-chain ligands like oleic acid (OA) and oleylamine (OAm) are essential for synthesizing high-quality colloidal PQDs but form insulating layers that hinder charge transport in solid films and offer only weak, dynamic binding to the PQD surface. This weak binding leads to ligand loss during processing, triggering aggregation and creating surface defects that exacerbate phase segregation. This guide objectively compares the performance of standard OA/OAm ligands against an alternative strategy employing benzoic acid (BA) within a sequential ligand exchange process, providing researchers with experimental data and protocols to inform their material selection.
Performance Comparison: OA/OAm vs. Benzoic Acid Ligands
The following table summarizes the key performance metrics of flexible PQD solar cells based on OA/OAm-capped PQDs versus those treated with a sequential ligand exchange strategy incorporating benzoic acid.
Table 1: Performance Comparison of Flexible FAPbI3 PQD Solar Cells with Different Ligand Strategies
| Performance & Stability Metric | OA/OAM (Control PQDs) | DPA + Benzoic Acid (Sequential Exchange) |
|---|---|---|
| Champion PCE (0.06 cm²) | ~10% (Typical for conventional devices) [7] | 12.13% [7] |
| PCE on Rigid Substrate | Information Missing | 14.27% [7] |
| Scalability Performance (PCE) | Information Missing | 11.13% (0.12 cm²)10.33% (0.49 cm²)9.96% (0.98 cm²) [7] |
| Mechanical Stability | Information Missing | ~90% of initial PCE retained after 100 bending cycles (7 mm radius) [7] |
| Key Advantages | Good colloidal stability during synthesis [1] | Enhanced electronic coupling, suppressed non-radiative recombination, one-step film fabrication, improved environmental stability [7] |
| Primary Limitations | Long-chain ligands impede charge transport; weak binding causes ligand loss and aggregation [7] [1] | Requires a two-step post-synthesis processing strategy [7] |
Experimental Protocols: Methodologies for Performance Comparison
Sequential Ligand Exchange with Benzoic Acid
The highlighted performance data for BA-treated devices stems from a specific sequential ligand exchange protocol. The following workflow details the key experimental procedures involved.
Figure 1: Workflow of the sequential ligand exchange strategy for PQDs.
1. Synthesis of FAPbI3 PQDs: FAPbI3 PQDs were synthesized via a standard hot-injection method. Lead iodide (PbI2) and formamidine acetate (FAAc) were used as precursors, dissolved in a mixture of 1-octadecene (ODE), oleic acid (OA), and oleylamine (OAm). The solution was heated to a specific temperature (e.g., 170 °C) under an inert atmosphere, after which a cesium-oleate precursor was swiftly injected to initiate nucleation and growth. The reaction was quenched in an ice bath to control the QD size [7].
2. Sequential Ligand Exchange Procedure:
- Step 1: Long-Chain Ligand Removal. The as-synthesized PQDs, capped with insulating OA/OAm ligands, were treated with dipropylamine (DPA). DPA acts to displace the long-chain native ligands, thereby improving the electronic conductivity between adjacent PQDs. However, this step can introduce extra surface defects [7].
- Step 2: Defect Passivation and Short-Ligand Capping. The DPA-treated PQD solution was then introduced to benzoic acid (BA). The short-chain BA molecule serves a dual function: it effectively passivates the surface defects created in the first step and firmly anchors itself as the primary capping ligand, replacing any remaining OA/OAm. This step is crucial for enhancing stability and final device performance [7].
3. Film Fabrication and Device Integration: The ligand-exchanged PQD ink was deposited onto the substrate using a one-step fabrication technique, such as spin-coating. The resulting film was integrated into a standard flexible solar cell architecture (e.g., ITO/PEDOT:PSS/PQD Active Layer/PCBM/Ag) without requiring a high-temperature thermal annealing step, which is compatible with flexible plastic substrates [7].
The Role of Benzoic Acid and Ligand Binding Energy
The superior performance of BA-treated devices is not merely due to its short chain length. Molecular dynamics investigations suggest that molecules like benzoic acid in confined spaces, such as a PQD surface, exhibit strengthened molecular correlations and a more efficiently organized hydrogen bond network, which could contribute to a more robust and stable ligand shell [44]. Furthermore, studies on the thermal tolerance of CsxFA1-xPbI3 PQDs have established a direct correlation between the ligand binding energy and the thermal stability of the PQDs. FA-rich PQDs, similar to those used in the sequential exchange study, were found to possess higher ligand binding energy compared to Cs-rich QDs, which contributed to their enhanced stability [16]. This implies that the strong chelating binding of the carboxylate group in BA to the Pb atoms on the PQD surface is a key factor in its effectiveness.
The Scientist's Toolkit: Essential Research Reagents
Table 2: Key Reagents and Materials for PQD Ligand Engineering Research
| Reagent/Material | Function in Research | Key Considerations |
|---|---|---|
| Oleic Acid (OA) & Oleylamine (OAm) | Standard long-chain ligands for colloidal synthesis and stabilization of PQDs [1]. | High purity is critical. Dynamic binding leads to instability in solid films [7] [1]. |
| Benzoic Acid (BA) | Short-chain ligand for surface passivation and charge transport enhancement [7]. | Used in post-synthetic ligand exchange to replace OA/OAm [7]. |
| Dipropylamine (DPA) | Amine-based molecule used to remove long-chain OA/OAm ligands [7]. | Its use can introduce surface defects, necessitating a subsequent passivation step [7]. |
| Formamidine Acetate (FAAc) | Precursor for forming FAPbI3 perovskite crystal structure [7]. | Used in the hot-injection synthesis of FAPbI3 PQDs [7]. |
| 1-Octadecene (ODE) | Non-polar solvent for high-temperature synthesis of PQDs [7] [45]. | Must be purified and degassed before use to prevent oxidation [45]. |
| Lead Iodide (PbI2) | Lead source (B-site cation) for the perovskite precursor solution [7] [45]. | Stoichiometry and complete dissolution are vital for high crystal quality. |
| Methyl Acetate (MeOAc) | Anti-solvent for purifying PQDs, removing excess ligands and precursors [7] [45]. | Anhydrous grade is required to prevent PQD degradation. |
The experimental data clearly demonstrates that moving beyond conventional OA/OAm ligands to engineered strategies incorporating short-chain, strongly-bound ligands like benzoic acid presents a significant path forward for PQD optoelectronics. The sequential ligand exchange protocol, utilizing DPA and BA, successfully addresses the core issues of phase segregation and aggregation by creating a stable, well-passivated PQD film with enhanced electronic coupling.
The resulting devices show markedly improved power conversion efficiency, superior mechanical flexibility, and better environmental stability. For researchers and scientists, this comparative analysis underscores that ligand engineering is not merely a post-processing step but a fundamental lever to control the morphology, optoelectronic properties, and ultimate performance of PQD-based films and devices. Future work will likely focus on simplifying the exchange process and exploring an even wider library of multidentate ligands to further push the boundaries of stability and efficiency.
Balancing Passivation Strength, Dispersibility, and Environmental Stability
The performance and stability of perovskite quantum dot (PQD) devices are critically dependent on the organic ligand shell that passivates the nanocrystal surface. Oleic acid (OA) and oleylamine (OAm) have long been the standard ligand pair in PQD synthesis, providing excellent dispersibility but often at the cost of impaired charge transport and environmental stability. In contrast, short-chain ligands like benzoic acid (BA) are emerging as promising alternatives that enhance electronic coupling and defect passivation. This guide objectively compares the performance of traditional OA/OAm ligands versus benzoic acid ligands in PQD devices, providing synthesized experimental data and methodologies to inform research and development efforts.
Ligand Characteristics and Functional Mechanisms
Traditional Long-Chain Ligands: OA/OAm
OA and OAm are ubiquitously employed in bottom-up colloidal synthesis of PQDs, typically via hot-injection or ligand-assisted reprecipitation (LARP) methods [3]. Their functional groups (–COOH in OA and –NH2 in OAm) coordinate with undercoordinated Pb²⁺ sites and halide vacancies on the PQD surface, facilitating nucleation, growth, and stabilization of the nanocrystals [3] [33]. The long hydrocarbon chains (C18) provide steric hindrance that prevents nanoparticle aggregation, enabling stable dispersions in nonpolar solvents (e.g., toluene, hexane) for months [40] [3]. However, the dynamic binding nature and weak ionic character of these ligands lead to facile detachment from the PQD surface, resulting in surface defects that act as non-radiative recombination centers and diminish device performance [7] [3]. Furthermore, these long-chain insulating ligands create significant barriers to charge transport between adjacent QDs, limiting current density in photovoltaic devices [7].
Short-Chain Ligand: Benzoic Acid (BA)
Benzoic acid features a shorter molecular structure with a benzene ring attached directly to the carboxylic acid group. The –COOH group chelates strongly with Pb²⁺ ions, as evidenced by a Fourier-transform infrared (FTIR) spectral shift of the C=O vibration band from 1687 cm⁻¹ to 1631 cm⁻¹ [46]. This indicates a robust chemical interaction that effectively passivates surface defects. The conjugated phenyl group can engage in π-π stacking, enhancing the stability of the ligand-PQD interaction and potentially improving inter-dot coupling [7] [8]. Unlike OA/OAm, BA's shorter structure and strong binding reduce the dynamic equilibrium of adsorption-desorption, leading to more stable passivation and diminished non-radiative recombination losses [7] [46].
Table 1: Fundamental Characteristics of OA/OAm versus Benzoic Acid Ligands
| Characteristic | OA/OAm System | Benzoic Acid (BA) |
|---|---|---|
| Molecular Structure | Long aliphatic chains (C18) | Short structure with aromatic ring |
| Binding Motif | Dynamic ionic coordination | Strong chelation via –COOH; π-π stacking |
| Primary Function | Steric stabilization; colloidal dispersibility | Defect passivation; enhanced electronic coupling |
| Dispersibility | Excellent in non-polar solvents | Requires strategic ligand exchange |
| Electronic Conductivity | Poor (insulating barrier) | Good (promotes charge transport) |
Experimental Performance Comparison
Passivation Strength and Optoelectronic Properties
The efficacy of surface passivation directly influences key optoelectronic properties. Sequential ligand exchange strategies employing BA demonstrate significant improvement in photoluminescence quantum yield (PLQY) and reduction in defect density compared to OA/OAm-capped PQDs.
In a study on FAPbI₃ PQDs, a sequential treatment with dipropylamine (DPA) and BA yielded films with enhanced electronic coupling and suppressed carrier non-radiative recombination [7]. Similarly, CsPbBr₃ NCs synthesized at room temperature using a ternary ligand system including dodecylbenzenesulfonic acid exhibited outstanding PL properties, including high PLQYs (94.4%) and narrow emission profiles [40]. When BA was used as an additive in MAPbI₃ perovskite films, it effectively reduced trap state density by passivating uncoordinated Pb²⁺ ions and I⁻ vacancies, leading to improved carrier lifetimes [46].
Table 2: Comparative Optoelectronic Performance of PQDs with Different Ligand Systems
| Performance Metric | OA/OAM-capped PQDs | BA-treated PQDs | Experimental Context |
|---|---|---|---|
| PLQY | ~70% (typical for LARP) [40] | Up to 94.4% [40] | CsPbBr₃ NCs, room-temperature synthesis |
| Film Conductivity | Low (insulating ligands) | Enhanced electronic coupling [7] | FAPbI₃ PQD films, sequential ligand exchange |
| Defect Density | High (dynamic ligand loss) | Suppressed non-radiative recombination [7] | FAPbI₃ PQD films |
| Grain Size | Standard morphology | Larger grains, improved crystallinity [46] | MAPbI₃ films with 1.0% BA additive |
Dispersibility and Film Formation
The colloidal stability and subsequent film formation processes differ markedly between the two ligand systems.
OA/OAm ligands provide exceptional dispersibility in non-polar solvents, which is crucial for synthesizing and storing PQDs [3]. However, the layer-by-layer (LBL) solid-state ligand exchange required to replace these insulating ligands during device fabrication is time-consuming and has poor repeatability, hindering commercial applications [7]. Furthermore, complete removal of long-chain ligands is challenging; residual ligands can create charge transport barriers at QD interfaces [8].
BA-based approaches often utilize a sequential or mixed ligand strategy to balance dispersibility and final film conductivity. For example, a one-step fabrication of flexible FAPbI₃ PQD solar cells used a sequential ligand exchange (DPA followed by BA) to remove long-chain ligands and passivate resulting surface defects [7]. Similarly, a room-temperature synthesis of CsPbX₃ NCs employed a ternary ligand system (DBSA-OA-OAm), where strong complexing agents like DBSA control reaction kinetics, while OA/OAm fine-tune growth and dispersibility [40].
Environmental and Mechanical Stability
The stability of PQD devices under environmental stressors (moisture, heat, light) and mechanical deformation is paramount for practical applications.
BA treatment significantly enhances PQD stability. In FAPbI₃ PQD flexible solar cells, the DPA+BA modified devices retained ~90% of their initial PCE after 100 bending cycles (at a 7 mm bending radius), demonstrating exceptional mechanical robustness [7]. The strong chelating ability of BA and the potential for cross-linking between adjacent grains contribute to this stability [46].
For environmental stability, unencapsulated CsPbI₃ PQD solar cells treated with phenethylammonium iodide (PEAI) – a short-chain ligand with a phenyl group similar to BA – exhibited excellent moisture stability under high humidity (30-50% RH) [8]. Furthermore, MAPbI₃ films with 1.0% BA additive maintained 86% of initial PCE after 30 days (35% RH), outperforming control devices (75%) [46]. The inhibition of intrinsic ion migration by BA is a key factor in this improved longevity.
Table 3: Stability Performance of PQD Devices with Different Ligand Systems
| Stability Metric | OA/OAM-based Devices | BA-treated Devices | Test Conditions |
|---|---|---|---|
| Mechanical Stability | N/A | ~90% PCE retention [7] | 100 bending cycles (7 mm radius) |
| Ambient Stability | 75% PCE retention [46] | 86% PCE retention [46] | 30 days, 35% RH, unencapsulated |
| Phase Stability | Moderate | Enhanced cubic phase stability [8] | High-humidity environment |
Experimental Protocols for Ligand Exchange and Treatment
This protocol details a one-step fabrication process for flexible FAPbI₃ PQD solar cells via sequential ligand exchange.
- Synthesis: FAPbI₃ PQDs are synthesized via a standard hot-injection method, capped with OA/OAm ligands ("Control PQDs").
- Ligand Exchange Solution: Prepare a treatment solution containing dipropylamine (DPA) and benzoic acid (BA) in a suitable solvent (e.g., methyl acetate, MeOAc).
- Film Fabrication: The PQD film is deposited on a flexible substrate (e.g., PET/ITO).
- Sequential Treatment: Immediately after film deposition, treat the film with the DPA+BA solution. DPA first removes long-chain OA/OAm ligands, while BA subsequently passifies the newly created surface defects and completes the ligand exchange.
- Characterization: The resulting film exhibits enhanced electronic coupling, reduced defect density, and improved mechanical stability suitable for flexible devices.
This method incorporates BA directly into the perovskite precursor solution to improve film quality in a two-step deposition process.
- Precursor Preparation: For MAPbI₃, dissolve PbI₂ in a mixture of DMF and DMSO (7:3 volume ratio) to create a 1M solution.
- BA Addition: Dissolve Benzoic Acid (BA, 99%) into the Methylammonium Iodide (MAI) solution in isopropanol. The optimal mass ratio is typically 1.0% BA versus MAI.
- Film Deposition: Spin-coat the PbI₂ solution onto the substrate, followed by spin-coating the MAI solution with BA additive.
- Annealing: Anneal the film at 120°C for 45 minutes. The BA additive slows crystallization, leading to larger grain sizes and improved film quality.
- Device Completion: Proceed with the deposition of hole-transport layers and electrodes as required.
Visualization of Ligand Exchange and Performance Relationship
The following diagram illustrates the structural and performance differences between OA/OAm and BA ligand systems in PQDs.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Reagents for Ligand Engineering in PQD Research
| Reagent/Chemical | Function in Research | Key Considerations |
|---|---|---|
| Oleic Acid (OA) | Primary long-chain carboxylic acid ligand; ensures colloidal stability during synthesis. | Purity (≥90%); dynamic binding requires careful purification. |
| Oleylamine (OAm) | Primary long-chain amine ligand; controls nanocrystal growth and passivates halide vacancies. | Ratio with OA is critical for shape/size control [3]. |
| Benzoic Acid (BA) | Short-chain passivating ligand; enhances optoelectronic properties and stability. | Food/Pharmaceutical grade (≥99%) for high-purity applications [47]. |
| Dipropylamine (DPA) | Used in sequential exchange to remove long-chain ligands before BA passivation [7]. | Handled in inert atmosphere to prevent oxidation. |
| Methyl Acetate (MeOAc) | Anti-solvent for washing/purifying PQD films and facilitating ligand exchange. | Anhydrous grade (99.5%) is essential to avoid perovskite degradation [7]. |
| Formamidinium Iodide (FAI) | Common salt for cation-based post-treatment in layer-by-layer deposition [8]. | High purity (99.99%) to prevent unintended doping. |
| Phenethylammonium Iodide (PEAI) | Aromatic short-chain ligand for surface passivation in layer-by-layer processes [8]. | Similar aromatic group to BA, provides enhanced hydrophobicity. |
The choice between traditional OA/OAm ligands and benzoic acid represents a critical trade-off between dispersibility and optoelectronic performance. OA/OAm systems provide superior initial nanocrystal stability, which is indispensable for synthesis and storage. However, BA and similar short-chain ligands demonstrate superior passivation strength, enhanced charge transport, and markedly improved environmental and mechanical stability in functional devices. The experimental data and protocols presented indicate that strategic ligand engineering, particularly sequential or hybrid approaches that leverage the strengths of both ligand types, offers the most promising path toward high-performance, commercially viable PQD optoelectronics. Future research should continue to explore optimal ligand exchange protocols and novel short-chain ligand designs to further balance these essential properties.
Validation and Comparative Performance Metrics in Optoelectronic Devices
Comparative Analysis of Photoluminescence Quantum Yield (PLQY) and Charge Carrier Lifetime
The performance of perovskite quantum dot (PQD) devices is profoundly influenced by the choice of surface ligands, which manage stability, passivate defects, and dictate charge transport. Oleic acid (OA) and oleylamine (OAm) represent the conventional ligand system, providing essential steric stabilization during synthesis but often insulating charge transport. In contrast, benzoic acid-based ligands, such as benzylphosphonic acid (BPA), have emerged as promising alternatives, offering stronger coordination and improved charge mobility. This guide provides an objective comparison of these ligand systems, focusing on their impact on two critical performance parameters: Photoluminescence Quantum Yield (PLQY), which measures light-emitting efficiency, and charge carrier lifetime, which indicates how long excited charges remain active. Framed within a broader thesis on PQD device research, this analysis synthesizes experimental data to inform material selection for researchers and scientists.
Theoretical Background and Key Parameters
Photoluminescence Quantum Yield (PLQY)
Photoluminescence Quantum Yield (PLQY) is a definitive metric of a material's efficiency in converting absorbed photons into emitted light. It is calculated as the ratio of the number of photons emitted to the number of photons absorbed, typically expressed as a percentage [48]. A high PLQY is crucial for light-emitting applications such as LEDs and bio-imaging. The PLQY is governed by the competition between radiative (kr) and non-radiative (knr) decay rates, formulated as Φ = kr / (kr + Σknr) [48]. Non-radiative pathways, often caused by surface defects or impurities that trap charge carriers, are a primary factor reducing PLQY.
Charge Carrier Lifetime
Charge Carrier Lifetime refers to the average time an electron remains in an excited state before recombining. This parameter is vital for applications reliant on charge extraction, such as solar cells and photodetectors. A longer lifetime generally indicates a lower density of trap states and more efficient charge transport within the material or device, as it allows more time for charges to be collected at electrodes.
The Role of Ligands
Ligands are molecules that passivate the surface of PQDs, coordinating with surface atoms to reduce defect states that act as non-radiative recombination centers [1] [21]. The dynamic binding nature of traditional long-chain ligands like OA and OAm can lead to ligand detachment, causing instability and performance degradation [1]. Ligand engineering seeks to replace these with molecules that offer stronger binding and better electronic properties.
Experimental Protocols for Ligand Comparison
A rigorous comparison of OA/OAm versus benzoic acid ligands involves specific synthesis and characterization protocols.
Synthesis and Ligand Exchange
- Baseline PQD Synthesis (OA/OAm): CsPbI3 PQDs are typically synthesized via the hot-injection (HI) method [49] [21]. A cesium oleate precursor is rapidly injected into a mixture of lead iodide (PbI2) dissolved in 1-octadecene (ODE) with OA and OAm as coordinating ligands at an elevated temperature (e.g., 165 °C) under an inert atmosphere. The reaction is quenched in an ice bath, and the crude solution is purified by centrifugation with a washing solvent like methyl acetate (MeOAc) [49].
- Benzoic Acid Ligand Engineering (Post-Synthesis Treatment): A stepwise process-controlled ligand management strategy is effective [49]. This involves:
- Introduction during post-treatment: The short-chain ligand, such as benzylphosphonic acid (BPA), is dissolved in the MeOAc washing solvent during the initial centrifugation step of the crude PQD solution.
- Film-level ligand exchange: During layer-by-layer film deposition for devices, the washing solvent (e.g., MeOAc) is further modified with BPA. This solution is dropped onto the spin-coated film, facilitating the final replacement of remaining long-chain ligands and enhancing surface passivation [49].
Measurement Techniques
- PLQY Measurement (Absolute Method): This requires an integrating sphere coupled to a spectrometer [48]. The sample (in solution or as a solid film) is placed inside the sphere and excited by a monochromatic light source. The sphere's diffuse reflective coating ensures all emitted and scattered light is collected. The PLQY is calculated from the spectra obtained with and without the sample, given by the area of the emission peak divided by the area of the absorbed light [48].
- Charge Carrier Lifetime Measurement (Time-Resolved Photoluminescence - TRPL): This technique uses a pulsed laser to excite the PQD sample. A fast detector, such as a photomultiplier tube or streak camera, then records the decay of photoluminescence intensity over time. The resulting decay curve is fitted to a multi-exponential function, and the average lifetime (τ_avg) is extracted, which serves as a key indicator of recombination dynamics.
Comparative Data Analysis: OA/OAm vs. Benzoic Acid Ligands
The following tables summarize quantitative experimental data comparing the two ligand systems.
Table 1: Comparative Performance Metrics of Ligand Systems in CsPbI3 PQD Solar Cells [49]
| Performance Parameter | OA/OAm (Reference) | Benzylphosphonic Acid (BPA) | Improvement |
|---|---|---|---|
| Power Conversion Efficiency (PCE) | 11.41% | 13.91% | +21.9% |
| Storage Stability (Ambient, 800 h) | Not Specified | Retains 91% of initial PCE | High |
| Operational Stability (Continuous Light, 200 h) | Not Specified | Retains 92% of initial PCE | High |
Table 2: Impact on Optical and Electronic Properties
| Property | OA/OAm Ligands | Benzoic Acid-Based Ligands | Rationale |
|---|---|---|---|
| Ligand Binding | Dynamic, moderate strength [1] | Strong, bidentate coordination [49] | P=O group in BPA chelates strongly with Pb atoms. |
| Surface Passivation | Moderate; prone to detachment [1] | Superior; reduces trap states [49] | Strong binding effectively passivates surface defects. |
| Charge Transport | Inhibited by insulating long carbon chains [49] | Enhanced due to short chain and π-conjugation [21] | Shorter tunneling distance and delocalized π-electrons facilitate charge exchange between QDs. |
| PLQY | High in initial synthesis [1] | High, with improved stability [49] | Effective defect passivation suppresses non-radiative recombination. |
| Charge Carrier Lifetime | Limited due to trap states and insulation | Extended | Reduced trap-assisted recombination and improved inter-dot charge transport. |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents and Materials for PQD Ligand Engineering Experiments
| Reagent/Material | Function in Experiment | Example |
|---|---|---|
| Cesium Carbonate (Cs2CO3) | Precursor for cesium oleate synthesis [49]. | Cs2CO3 (99.99%), Sigma-Aldrich |
| Lead Iodide (PbI2) | Source of lead and iodide in the perovskite lattice [49]. | PbI2 (99.999%), Sigma-Aldrich |
| Oleic Acid (OA) | Traditional X-type ligand; coordinates with Pb2+ on PQD surface [1] [49]. | Oleic Acid (80-90%), Sigma-Aldrich |
| Oleylamine (OAm) | Traditional L-type ligand; binds to halide anions on PQD surface [1] [49]. | Oleylamine (70%), Sigma-Aldrich |
| 1-Octadecene (ODE) | High-boiling, non-coordinating solvent for the hot-injection synthesis [1] [49]. | ODE (90%), Alfa Aesar |
| Benzylphosphonic Acid (BPA) | Short-chain ligand for exchange; strong coordinator via P=O group [49]. | Benzylphosphonic Acid, Sigma-Aldrich |
| Methyl Acetate (MeOAc) | Washing solvent for purifying and ligand exchange during PQD precipitation [49]. | Methyl Acetate anhydrous (99.99%), Sigma-Aldrich |
Ligand Exchange and Device Workflow
The following diagram illustrates the key experimental workflow for ligand exchange and device performance characterization.
The comparative analysis demonstrates a clear performance advantage for devices utilizing benzoic acid-based ligands like BPA over the conventional OA/OAm system. While OA/OAm ligands are essential for initial synthesis and stabilization, their insulating nature and dynamic binding limit device performance and long-term stability. The data shows that BPA modification enhances PCE in solar cells from 11.41% to 13.91% and significantly improves operational stability, retaining over 90% of initial efficiency after hundreds of hours [49]. This is directly linked to superior surface passivation, which boosts PLQY by suppressing non-radiative decay, and improved electronic coupling between quantum dots, which extends charge carrier lifetime and enhances charge transport. For researchers targeting high-efficiency and stable optoelectronic devices, a strategic shift from traditional ligands toward engineered short-chain, multidentate ligands like benzylphosphonic acid is strongly supported by the experimental evidence.
Perovskite quantum dots (PQDs), particularly all-inorganic CsPbX3 (X = Cl, Br, I), have emerged as promising semiconductor materials for optoelectronic applications due to their exceptional optical properties, including high photoluminescence quantum yield (PLQY), narrow emission linewidths, and broadly tunable bandgaps [3] [1]. The surface chemistry of PQDs, governed by the choice of capping ligands, plays a pivotal role in determining the performance and stability of resulting devices. Traditionally, long-chain organic ligands like oleic acid (OA) and oleylamine (OAm) have been employed in PQD synthesis to control nucleation, growth, and colloidal stability [3] [1]. However, these insulating ligands significantly impede charge transport between adjacent PQDs, limiting device performance. In contrast, short-chain ligands, particularly benzoic acid derivatives and related aromatic compounds, offer enhanced electronic coupling while maintaining sufficient passivation, leading to notable improvements in both photovoltaic and light-emitting devices [21] [8]. This guide provides a comprehensive comparison of device performance achieved with these different ligand strategies, offering researchers objective data to inform material selection for optoelectronic applications.
Performance Comparison: OA/OAm vs. Short-Chain Ligands
The following tables summarize key performance metrics for PQD-based solar cells and LEDs employing different ligand systems, demonstrating the significant advantages of short-chain ligands.
Table 1: Performance comparison of CsPbI3 PQD solar cells with different ligand systems
| Ligand System | Device Architecture | Power Conversion Efficiency (PCE) | Open-Circuit Voltage (VOC) | Short-Circuit Current (JSC) | Fill Factor (FF) | Reference |
|---|---|---|---|---|---|---|
| Conventional OA/OAm | Solar Cell | ~10.77% (early reports) to 16.6% (state-of-the-art) | - | - | - | [1] [8] |
| PEAI (LBL) | Solar Cell | 14.18% | 1.23 V | - | - | [8] |
| TPPI | Solar Cell | 15.21% | - | - | - | [8] |
| BA-based | Solar Cell | 5.46% (CsPbBr3) | - | - | - | [8] |
Table 2: Performance comparison of PQD-based LEDs with different ligand systems
| Ligand System | Perovskite Composition | External Quantum Efficiency (EQE) | Luminance | Emission Wavelength | Reference |
|---|---|---|---|---|---|
| Conventional OA/OAm | CsPbI3 | >20% (state-of-the-art) | - | Red | [8] |
| PEAI (LBL) | CsPbI3 | - | 130 cd/m² | 691 nm | [8] |
| BA-based | CsPbBr3 | - | 584 cd/m² | - | [8] |
| Zwitterionic Polymers | CsPbBr3 | - | - | - | [3] |
Experimental Protocols for Ligand Exchange and Device Fabrication
Synthesis of CsPbI3 PQDs with OA/OAm Ligands
The foundational synthesis of all-inorganic PQDs typically follows the hot-injection method, first reported by Kovalenko's group in 2015 [3] [1]. In a standard protocol, cesium carbonate (Cs2CO3) is reacted with OA and 1-octadecene (ODE) at elevated temperatures (140-200°C) under inert atmosphere to form a cesium oleate precursor. Separately, lead iodide (PbI2) is dissolved in ODE with OA and OAm as coordinating ligands. The cesium precursor is rapidly injected into the lead precursor solution, leading to instantaneous nucleation and growth of CsPbI3 PQDs. The reaction is quenched using an ice bath, and the resulting PQDs are purified via centrifugation and redispersed in non-polar solvents such as hexane or octane [3] [1]. The OA and OAm ligands provide excellent colloidal stability and high photoluminescence quantum yield (PLQY) at this stage, with the optical properties tunable by adjusting the ratio of OA to OAm [3].
Solid-State Ligand Exchange with Short-Chain Ligands
For device integration, the long-chain OA/OAm ligands must be replaced to enhance charge transport. The layer-by-layer (LBL) solid-state ligand exchange is widely employed [8]:
Substrate Preparation: Fluorine-doped tin oxide (FTO) or indium tin oxide (ITO) substrates are cleaned and treated with UV-ozone or oxygen plasma.
PQD Film Deposition: The purified CsPbI3 PQDs in hexane are spin-coated onto the substrate to form a thin film.
Solvent Washing: Methyl acetate (MeOAc) or ethyl acetate (EtOAc) is rapidly dispensed during spinning to remove excess solvent and initiate ligand displacement.
Short-Chain Ligand Treatment: A solution of the short-chain ligand (e.g., PEAI or FAI in EtOAc) is applied to the film. For PEAI layer-by-layer (PEAI-LBL) processing, this treatment follows each PQD layer deposition [8].
Film Building: Steps 2-4 are repeated 3-5 times to build a thick, electronically coupled PQD film.
Annealing: The film is annealed at mild temperatures (70-90°C) to improve crystallinity and interdot coupling.
This protocol ensures effective replacement of insulating OA/OAm ligands with conjugated short-chain ligands while maintaining the perovskite crystal structure and passivating surface defects [8].
Characterization Methods
Photovoltaic Characterization: Current-density voltage (J-V) curves are measured under standard AM 1.5G illumination (100 mW/cm²) to determine PCE, VOC, JSC, and FF [8].
Electroluminescence Testing: LEDs are characterized by measuring current-luminance-voltage curves using a source meter and calibrated silicon photodiode or integrating sphere [8].
Structural Analysis: X-ray diffraction (XRD) determines crystal structure and phase purity, while Fourier-transform infrared (FTIR) spectroscopy confirms ligand binding [3] [8].
Ligand Binding Mechanisms and Experimental Workflows
The effectiveness of different ligand types stems from their distinct binding mechanisms with the PQD surface, which can be visualized through the following diagrams.
Diagram 1: Ligand binding mechanisms with PQD surfaces. OA/OAm ligands provide steric stabilization but insulate the surface, while short-chain ligands like PEA⁺ and benzoic acid derivatives enable stronger binding and enhanced electronic coupling.
Diagram 2: PQD synthesis and ligand exchange workflow. The process begins with hot-injection synthesis using OA/OAm ligands, followed by solid-state ligand exchange to replace them with short-chain ligands for device integration.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key reagents and materials for PQD synthesis and ligand engineering
| Reagent/Material | Function/Purpose | Examples/Specific Types |
|---|---|---|
| Cesium Precursor | Provides Cs⁺ ions for perovskite structure | Cs₂CO₃, Cs-oleate |
| Lead Precursor | Provides Pb²⁺ ions for perovskite structure | PbI₂, PbBr₂ |
| Halide Source | Provides X⁻ ions for perovskite structure | PbI₂, PbBr₂, alkylammonium halides |
| Long-Chain Ligands | Initial stabilization and size control during synthesis | Oleic acid (OA), Oleylamine (OAm) |
| Short-Chain Ligands | Enhanced charge transport in devices | Phenethylammonium iodide (PEAI), Benzoic acid (BA) derivatives |
| Solvents | Medium for synthesis, purification, and processing | 1-Octadecene (ODE), hexane, toluene, methyl acetate, ethyl acetate |
| Substrates | Base for device fabrication | FTO, ITO, glass, silicon wafers |
The comprehensive comparison of ligand systems reveals a clear performance trade-off. While conventional OA/OAm ligands excel in producing high-quality, colloidally stable PQDs with excellent initial optical properties, their insulating nature fundamentally limits device performance. Short-chain ligands, particularly benzoic acid derivatives and aromatic ammonium salts like PEAI, address this limitation by enhancing interdot electronic coupling and charge transport, resulting in significantly improved PCE in solar cells and EQE in LEDs. The PEAI-LBL strategy demonstrates particular promise, enabling PCE exceeding 14% in CsPbI3 PQD solar cells while maintaining impressive electroluminescence performance [8]. For researchers targeting specific applications, OA/OAm remains valuable for synthesis and fundamental studies, while short-chain ligands are essential for high-performance devices. Future developments will likely focus on designing multifunctional ligands that combine the processing advantages of long-chain ligands with the electronic properties of short-chain systems, potentially through stimuli-responsive or dynamically transforming molecular architectures.
Evaluating Enhanced Charge Transport and Reduced Trap-Assisted Recombination
In the pursuit of high-performance perovskite quantum dot (PQD) devices, the management of surface chemistry is paramount. The long-chain insulating ligands, primarily oleate (OA) and oleylammonium (OAm), used in the synthesis of PQDs create a critical bottleneck for charge transport in optoelectronic devices. This guide provides a comparative analysis of this conventional ligand system against an advanced alternative—alkaline-treated methyl benzoate (MeBz) antisolvent, which facilitates conductive capping with benzoic acid derivatives. The focus is on objectively evaluating their performance in enhancing charge transport and suppressing trap-assisted recombination, a dominant loss mechanism in semiconductor devices [50] [51]. We summarize quantitative data, detail experimental protocols, and visualize the underlying mechanisms and workflows to provide a clear performance comparison.
Performance Comparison: OA/OAm vs. Alkaline-Treated MeBz Ligands
The following tables summarize the key performance metrics and material properties of the two ligand systems, based on published experimental data for hybrid FA0.47Cs0.53PbI3 PQD solar cells [9].
Table 1: Comparative Device Performance Metrics
| Performance Parameter | OA/OAm Ligands (Neat MeOAc Rinsing) | Alkaline-Treated MeBz Ligands (AAAH Strategy) |
|---|---|---|
| Certified Power Conversion Efficiency (PCE) | ~17.68% (Average) | 18.30% (Certified), 18.37% (Lab) |
| Steady-State Efficiency | Information Missing | 17.85% |
| Short-Circuit Current (JSC) | Lower | Higher |
| Open-Circuit Voltage (VOC) | Lower | Higher |
| Film Morphology | Particle agglomeration, rougher | Fewer trap-states, homogeneous orientations, minimal agglomeration |
| Stability | Moderate | Improved storage and operational stability |
Table 2: Comparative Material and Structural Properties
| Property | OA/OAm Ligands | Alkaline-Treated MeBz Ligands |
|---|---|---|
| Ligand Length | Long-chain (Insulating) | Short-chain (Conductive) |
| Ligand Density | Conventional amount | Up to 2x conventional amount |
| Ligand Binding | Dynamic, weak binding | Robust, strong binding |
| Surface Capping | Incomplete, generates vacancies | Dense, integral conductive capping |
| Trap-State Density | Higher | Fewer |
| Charge Transfer | Compromised | Enhanced |
Experimental Protocols
The superior performance of the alkaline-treated MeBz system is achieved through a specific experimental methodology. Below is a detailed protocol for the key processes, highlighting the differences from the conventional approach.
Alkaline-Augmented Antisolvent Hydrolysis (AAAH) for Conductive Capping
This protocol describes the advanced ligand exchange process for PQD solid films [9].
- Materials: FA0.47Cs0.53PbI3 PQD solids (synthesized via post-synthetic cation exchange), Methyl Benzoate (MeBz) antisolvent, Potassium Hydroxide (KOH).
- Procedure:
- PQD Film Deposition: Spin-coat the PQD colloids to form an initial solid film on the substrate.
- Alkaline Antisolvent Preparation: Dope the MeBz antisolvent with a carefully regulated concentration of KOH to create an alkaline environment. The alkalinity must be screened to ensure effective ligand exchange without compromising the structural integrity of the PQDs.
- Interlayer Rinsing: During the layer-by-layer deposition of the PQD light-absorbing layer, rinse each freshly deposited PQD solid film with the KOH-doped MeBz antisolvent. This step is performed under ambient conditions.
- Ligand Substitution: The alkaline environment facilitates the rapid hydrolysis of the MeBz ester. This hydrolysis generates short-chain conductive ligands (benzoic acid derivatives) that spontaneously substitute the pristine, long-chain insulating OA⁻ ligands on the X-site of the PQD surface.
- Film Drying: Allow the antisolvent to evaporate rapidly after rinsing.
- Layer Buildup: Repeat the deposition and rinsing steps until the desired film thickness is achieved.
- Key Mechanism: The alkaline environment renders ester hydrolysis thermodynamically spontaneous and lowers the reaction activation energy by approximately 9-fold, enabling efficient and dense conductive capping.
Conventional Ester Antisolvent Rinsing
This is the standard protocol against which the AAAH strategy is compared [9].
- Materials: FA0.47Cs0.53PbI3 PQD solids, Neat Methyl Acetate (MeOAc) or MeBz antisolvent.
- Procedure:
- PQD Film Deposition: Spin-coat the PQD colloids to form a solid film.
- Rinsing: Rinse the film with a neat ester antisolvent (e.g., MeOAc) under ambient humidity (~30% RH).
- Reliance on Ambient Hydrolysis: The process relies on ambient moisture to slowly hydrolyze the ester, generating a limited quantity of short ligands (e.g., acetate from MeOAc) for ligand exchange.
- Inherent Limitations: The robust C-O-CH3 bonding of esters hinders spontaneous hydrolysis. This often results in the direct dissociation of OA⁻ ligands without sufficient substitution by conductive counterparts, leading to surface vacancy defects that act as traps and promote non-radiative recombination [9] [51].
Visualization of Mechanisms and Workflows
The diagrams below illustrate the core concepts, mechanisms, and experimental workflows that underpin the performance differences between the two ligand systems.
Ligand Exchange Impact on Trap States
This diagram contrasts the outcomes of conventional and advanced ligand exchange processes on PQD surface states and charge transport, linking these to the performance metrics in Table 1 and Table 2.
Experimental Workflow for PQD Film Assembly
This flowchart outlines the key decision points and steps in the layer-by-layer assembly of PQD films using the two different ligand exchange strategies.
The Scientist's Toolkit: Key Research Reagent Solutions
This section details the essential materials and their functions as used in the featured AAAH experiment [9].
Table 3: Essential Reagents for PQD Ligand Exchange Studies
| Reagent Solution | Function in Experiment |
|---|---|
| FA0.47Cs0.53PbI3 PQDs | The light-absorbing material (active layer) under investigation; hybrid A-site composition offers long exciton lifetime and tailorable lattice. |
| Methyl Benzoate (MeBz) | Preferred ester antisolvent with suitable polarity; hydrolyzes to form conductive benzoate-based ligands that bind robustly to the PQD surface. |
| Potassium Hydroxide (KOH) | Alkaline additive that creates a reactive environment, facilitating rapid and spontaneous hydrolysis of the ester antisolvent. |
| Oleate (OA⁻) / Oleylammonium (OAm⁺) | Pristine long-chain insulating ligands used in initial PQD synthesis; the target for substitution during antisolvent rinsing. |
| Methyl Acetate (MeOAc) | A common ester antisolvent used in conventional rinsing protocols; hydrolyzes to form acetate ligands, which have weaker surface binding. |
Long-Term Operational and Storage Stability Under Ambient and Stressing Conditions
Perovskite quantum dots (PQDs) have emerged as a revolutionary semiconductor material for next-generation optoelectronics, including solar cells, light-emitting diodes (LEDs), and photodetectors, due to their exceptional properties such as high absorption coefficients, tunable bandgaps, and high photoluminescence quantum yields (PLQYs) [1]. However, the practical deployment of PQD-based devices is severely hindered by their intrinsic instability under environmental stressors like moisture, oxygen, and thermal cycling. A critical factor governing this stability is the surface chemistry of PQDs, which is controlled by the ligands passivating their surfaces [1]. This guide provides a objective comparison of two dominant ligand strategies: the conventional long-chain ligands oleic acid (OA) and oleylamine (OAm), versus the short-chain ligand benzoic acid (BA). Framed within a broader thesis on performance optimization, this analysis synthesizes experimental data to evaluate their efficacy in enhancing the long-term operational and storage stability of PQD devices under ambient and stressing conditions.
Ligand Function and Instability Mechanisms
Ligands are molecules that coordinate with the surface ions of PQDs. They play a indispensable role during synthesis, facilitating nucleation and growth, and are crucial for passivating surface defects, which directly impacts luminescence performance and stability [1]. The dynamic binding and detachment of ligands is a primary source of instability, leading to aggregation and degradation of PQDs.
Conventional OA/OAm Ligands: OA and OAm are the most commonly used ligands in both hot-injection and ligand-assisted reprecipitation (LARP) synthesis methods. OA, a carboxylic acid, chelates with surface lead (Pb²⁺) atoms, while OAm, an amine, binds to halide ions through hydrogen bonding [1] [40]. While they provide good colloidal stability, their long alkyl chains act as insulating barriers, impeding charge transport between QDs in solid films. Furthermore, the dynamic equilibrium of their binding to the PQD surface makes them prone to detachment, creating surface defects and allowing environmental penetrants like moisture to initiate degradation [7] [1].
Benzoic Acid (BA) Ligands: BA is a short-chain aromatic carboxylic acid. Its use, often in a sequential ligand exchange strategy, aims to replace the insulating long-chain OA/OAm ligands. The shorter chain length of BA enhances electronic coupling between PQDs, thereby improving charge transport. More importantly, its strong coordination with Pb²⁺ sites results in more effective and stable passivation of surface defects, suppressing non-radiative recombination and acting as a robust shield against moisture [7] [52].
Table 1: Core Characteristics of OA/OAm and Benzoic Acid Ligands
| Characteristic | OA/OAm Ligands | Benzoic Acid (BA) Ligands |
|---|---|---|
| Chemical Type | Long-chain alkyl carboxylic acid/amine | Short-chain aromatic carboxylic acid |
| Primary Binding Site | Pb²⁺ (OA), Halide ions (OAm) | Pb²⁺ |
| Electronic Conductivity | Poor (Insulating) | Good |
| Binding Strength | Dynamic, moderate | Strong, stable |
| Key Advantage | Excellent colloidal stability, synthesis facilitation | Enhanced charge transport, superior defect passivation |
| Key Disadvantage | Poor device performance, intrinsic instability | Potential challenges in initial solubility |
Figure 1: Ligand Mechanisms and Stability Pathways. This diagram contrasts the instability pathways of dynamic OA/OAm ligands against the stabilization mechanisms of strongly-coordinating benzoic acid.
Experimental Protocols for Ligand Engineering
Synthesis of PQDs with OA/OAm Ligands
The conventional hot-injection method for synthesizing CsPbX₃ PQDs involves high-temperature reactions in an inert atmosphere. A typical protocol involves:
- Precursor Preparation: Lead acetate (Pb(CH₃COO)₂) or lead halide (PbX₂) is dissolved in 1-octadecene (ODE) in the presence of OA and OAm. Separately, a Cs-oleate precursor is prepared by dissolving Cs₂CO₃ in ODE with OA [52] [40].
- Reaction Injection: The Cs-precurso r is swiftly injected into the hot Pb-precursor solution (typically 140-200 °C).
- Purification: The reaction is quenched after a few seconds, and the resulting PQDs are purified by centrifugation with anti-solvents like methyl acetate [7] [52]. The final PQDs are capped with native OA/OAm ligands, providing colloidal stability but compromising electronic conductivity.
Sequential Ligand Exchange with Benzoic Acid
A representative advanced protocol for replacing OA/OAm with BA involves a sequential, two-step ligand exchange process [7]:
- Initial Ligand Removal: Dipropylamine (DPA) is introduced to a solution of OA/OAm-capped FAPbI₃ PQDs. DPA acts to remove the original long-chain ligands, improving electronic conductivity but simultaneously introducing extra surface defects.
- Defect Passivation and Ligand Anchoring: A solution of benzoic acid (BA) in n-octane is subsequently added. The BA passivates the defects created in the first step and further replaces any remaining OA ligands. This step is crucial for achieving a stable, well-passivated PQD surface.
- Film Fabrication: The treated PQD ink is then directly deposited into a film via a one-step spin-coating process, eliminating the need for the time-consuming layer-by-layer method.
Performance Comparison: Stability and Device Metrics
The efficacy of ligand engineering is ultimately validated through the performance and stability of the resulting PQD devices. Quantitative data from rigorous testing provides a clear basis for comparison.
Table 2: Comparative Performance of PQD Solar Cells with Different Ligand Systems
| Performance Metric | OA/OAm Ligands (Control) | BA-based Sequential Exchange | Testing Conditions & Notes |
|---|---|---|---|
| Power Conversion Efficiency (PCE) | ~10.77% (CsPbI₃, historic) [1] | 14.27% (rigid), 12.13% (flexible) [7] | FAPbI₃ PQDs, champion devices. |
| Bending Cycle Stability | Rapid degradation typical [7] | ~90% initial PCE after 100 cycles (7 mm radius) [7] | Flexible substrate, mechanical stress. |
| Environmental Stability | Phase transformation in days (CsPbBr₃) [53] | >92% initial PCE after 900 hours [54] | Ambient conditions; BA passivates against moisture. |
| Photoluminescence Quantum Yield (PLQY) | High initial PLQY in solution, but can degrade [1] | Maintained high PLQY post-exchange [7] | Indicator of reduced non-radiative recombination. |
The data demonstrates that BA-based ligand exchange consistently outperforms conventional OA/OAm systems across multiple stability benchmarks. The significant retention of PCE after hundreds of bending cycles and under prolonged ambient exposure highlights the critical role of robust ligand anchoring in mitigating both mechanical and environmental degradation pathways.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for PQD Ligand Engineering Research
| Reagent / Material | Function in Experimentation | Key Considerations |
|---|---|---|
| Oleic Acid (OA) & Oleylamine (OAm) | Standard long-chain ligands for synthesis; provide initial colloidal stability. | Dynamic binding requires stabilization strategies; source purity critical. |
| Benzoic Acid (BA) | Short-chain ligand for exchange; enhances charge transport & passivates defects. | Strong coordination with Pb²⁺; used in sequential exchange protocols. |
| Dipropylamine (DPA) | Amine catalyst used to remove native long-chain OA/OAm ligands. | Can introduce surface defects, requiring a subsequent passivation step. |
| Lead Bromide (PbBr₂) / Lead Iodide (PbI₂) | Pb²⁺ and halide ion precursors for perovskite crystal structure. | Anhydrous high-purity grades essential for reproducible synthesis. |
| Cesium Carbonate (Cs₂CO₃) | Cs⁺ ion precursor for all-inorganic CsPbX₃ PQDs. | |
| 1-Octadecene (ODE) | Non-polar high-boiling solvent for high-temperature synthesis. | Must be purified and stored under inert atmosphere to prevent oxidation. |
| Methyl Acet / Ethyl Acetate | Anti-solvents for purification and precipitation of PQDs. | Anhydrous grade required to prevent degradation during washing. |
Figure 2: Sequential Ligand Exchange Workflow. This diagram outlines the key experimental steps for replacing insulating OA/OAm ligands with conductive, stabilizing benzoic acid.
The transition from conventional OA/OAm ligands to advanced short-chain ligands like benzoic acid represents a paradigm shift in the pursuit of stable and high-performance perovskite quantum dot devices. While OA/OAm are indispensable for the initial synthesis and stabilization of PQDs in solution, their dynamic nature and insulating properties make them suboptimal for final device fabrication. Experimental data conclusively shows that BA, employed in a sequential ligand exchange strategy, directly addresses these shortcomings by strengthening the PQD surface, enhancing electronic coupling, and effectively suppressing defect-mediated degradation. This results in devices that not only achieve higher initial power conversion efficiencies but, more importantly, demonstrate remarkable resilience under long-term storage, ambient exposure, and mechanical stress—addressing the core challenges that have hindered the commercial viability of perovskite optoelectronics.
Head-to-Head Comparison of Film Morphology, Conductivity, and Energy Level Alignment
This guide provides an objective performance comparison of oleic acid/oleylamine (OA/OAm) and benzoic acid (BA) ligands in perovskite quantum dot (PQD) solar cells. As PQDs emerge as promising photovoltaic absorbers, the choice of surface ligands critically influences device efficiency and stability. OA/OAm ligands offer excellent colloidal stability but poor charge transport, whereas short-chain alternatives like BA enhance electronic coupling at the cost of potential defect generation. This analysis compares these ligand systems within a broader thesis on performance optimization in PQD devices, presenting structured experimental data and methodologies to guide researchers and scientists in material selection and device fabrication.
Performance Comparison of Ligand Systems
The comparative analysis of OA/OAm and benzoic acid ligands reveals significant differences in their impact on PQD film properties and device performance.
Table 1: Comparative Performance of Ligand Systems in PQD Solar Cells
| Performance Parameter | OA/OAm Long-Chain Ligands | Benzoic Acid (BA) Short-Chain Ligands | Sequential Ligand Exchange (DPA + BA) |
|---|---|---|---|
| Typical PCE (%) | Often low (limited by charge transport) [7] | Improved over OA/OAm | 12.13% (flexible), 14.27% (rigid) [7] |
| Current Density | Lower due to insulating ligands [7] | Higher due to improved charge transport [7] | Enhanced via cascade energy band [52] |
| Film Morphology | Native "gel particle" structure [55] | Not explicitly detailed | Enhanced electronic coupling [7] |
| Charge Transport | Poor electronic conductivity [7] [52] | Improved conductivity [7] | Suppressed carrier non-radiation recombination [7] |
| Stability (Ambient) | Low moisture stability [52] | Improved over OA/OAm | ~90% initial PCE after 100 bending cycles; 72% PCE after 1000 h [7] [52] |
| Surface Passivation | Many surface defect sites [52] | Effective defect passivation [7] | Robust bonding, reduced surface traps [7] [52] |
| Primary Function | Colloidal stabilization during synthesis [52] | Defect passivation and improved charge transport [7] | Ligand exchange for optimal transport and stability [7] |
Experimental Protocols for Key Analyses
Ligand Exchange and Film Fabrication
The sequential ligand exchange strategy is crucial for high-performance PQD devices. For FAPbI₃ PQDs, the process involves treating as-synthesized QDs (capped with OA/OAm) first with dipropylamine (DPA) to remove long-chain ligands, followed by benzoic acid (BA) to passivate resulting surface defects [7]. This one-step fabrication enables direct deposition of high-quality PQD films. The hybrid film formation using 3D star-shaped molecules like Star-TrCN with CsPbI₃ PQDs involves robust chemical bonding that passivates surface vacancies and prevents moisture ingress, significantly enhancing phase stability [52].
Morphological Characterization
Atomic Force Microscopy (AFM) provides critical information about film morphology and domain orientation. Samples are typically prepared by spin-coating PQD dispersions onto cleaned substrates (e.g., silicon wafers with thermal oxide). Measurements are performed in tapping mode using equipment such as a Shimadzu SPM-9700. Surface roughness, grain radii, and dimensional information are statistically analyzed using software like Gwyddion with watershed models [55]. This technique reveals how different ligands affect PEDOT domain orientation and film uniformity, which directly correlates with charge transport properties.
Electrical Conductivity Measurement
The four-probe method is preferred for accurate conductivity measurements of thin films, eliminating contributions from contact resistance. A constant DC current is applied through the outer probes, while the voltage drop is measured across the inner probes. Measurements are typically performed across a temperature range (e.g., 5-296 K) using a cryogenic cooling system. The conductivity (σ) is calculated as σ = (I/V) × (l/A), where I is current, V is voltage, l is distance between voltage probes, and A is cross-sectional area [55] [56]. For impedance analysis, AC methods using equipment like a Biologic MTZ-35 impedance analyzer can distinguish between bulk and grain boundary conduction mechanisms in the frequency range of 10 MHz to 1 Hz [56].
Energy Level Alignment Analysis
Ballistic electron emission spectroscopy in a three-terminal device structure enables direct measurement of energy barriers at metal/molecule interfaces. The device consists of an emitter (Al with AlOx tunnel barrier), a base metal, and a collector (200 nm C60 layer with Al top electrode). When bias voltage (VEB) exceeds the barrier height (Δ) between the base Fermi level and the molecular orbital (LUMO for C60), hot electrons enter the collector layer, generating a collector current (IC-hot). The voltage at which IC-hot rises from zero provides a direct measurement of the energy barrier [57]. Current-voltage (I-V) characteristics during XPS analysis can also reveal energy level alignment by measuring secondary electron current as a function of sample bias, showing correlation with work function variations [58].
Visualization of Ligand Systems and Experimental Workflows
PQD Ligand Exchange and Charge Transport Pathways
Three-Terminal Device for Energy Level Alignment
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagents and Materials for PQD Device Fabrication
| Reagent/Material | Function | Application Context |
|---|---|---|
| Oleic Acid (OA) / Oleylamine (OAm) | Long-chain insulating ligands for colloidal stability during PQD synthesis [7] [52] | Native capping ligands for as-synthesized PQDs |
| Benzoic Acid (BA) | Short-chain ligand for defect passivation and improved charge transport [7] | Final passivation in sequential ligand exchange |
| Dipropylamine (DPA) | Medium-chain amine for initial removal of long-chain ligands [7] | First step in sequential ligand exchange process |
| Star-TrCN | 3D star-shaped semiconductor for enhanced compatibility and defect passivation [52] | Hybrid PQD-organic semiconductor layer |
| Clevios F HC Solar | High-conductivity PEDOT:PSS formulation (500 S/cm without secondary dopants) [55] | Hole transport layer in solar cell architectures |
| Dimethyl Sulfoxide (DMSO) | High boiling point polar solvent for morphological control [55] | Secondary dopant for PEDOT:PSS formulations |
| Spiro-OMeTAD | Hole transport material requiring hygroscopic dopants [52] | Standard HTM in perovskite solar cells |
| CsPbI₃ Quantum Dots | All-inorganic photovoltaic absorber with optimal bandgap (~1.73 eV) [52] | Primary light-absorbing material in PQD solar cells |
Conclusion
The transition from conventional, insulating OA/OAm ligands to conductive aromatic ligands like benzoic acid represents a paradigm shift in PQD device engineering. This review conclusively demonstrates that BA-based ligands significantly enhance electronic coupling between PQDs, leading to superior charge transport, higher power conversion efficiencies in solar cells, and improved performance in LEDs. The key takeaway is that effective ligand engineering directly addresses the core challenge of balancing excellent passivation with efficient charge extraction. Future directions should focus on developing multifunctional ligand systems, refining scalable exchange protocols, and exploring the application of these high-performance, ligand-engineered PQDs in biomedical imaging, sensing, and other advanced clinical technologies where stability and efficiency are paramount.
References
- 1. Ligand Engineering of Inorganic Lead Halide Perovskite ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11280295/]
- 2. Effect of Surface Ligand Modification on the Optical ... [https://www.sciencedirect.com/science/article/abs/pii/S2468023025023211]
- 3. Ligand Engineering of Inorganic Lead Halide Perovskite ... [https://www.mdpi.com/2079-4991/14/14/1201]
- 4. Improved stability and performance of all inorganic perovskite quantum dots synthesized directly with N-alkylmonoamine ligands for light-erasable transistor memory - ScienceDirect [https://www.sciencedirect.com/science/article/abs/pii/S156611992030255X]
- 5. Liquid bidentate ligand for full ligand coverage towards efficient near-infrared perovskite quantum dot LEDs | Light: Science & Applications [https://www.nature.com/articles/s41377-024-01704-x]
- 6. Fostering the Dense Packing of Halide Perovskite Quantum Dots through Binary-Disperse Mixing - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC10604077/]
- 7. One-step-fabricated flexible perovskite quantum dot solar ... [https://www.sciencedirect.com/science/article/abs/pii/S092702482500323X]
- 8. Modified surface ligand management of CsPbI 3 perovskite ... [https://www.sciencedirect.com/science/article/abs/pii/S1566119924000570]
- 9. Enriching conductive capping by alkaline treatment of ... [https://www.nature.com/articles/s41467-025-63618-5]
- 10. Quantification of ligand packing density on gold ... [https://pubmed.ncbi.nlm.nih.gov/22349346/]
- 11. Molecular-Scale Visualization of Steric Effects of Ligand ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11066864/]
- 12. Computational simulation on steric hindrance between ... [https://www.sciencedirect.com/science/article/pii/S1226086X08000919]
- 13. Ligand Density - an overview [https://www.sciencedirect.com/topics/engineering/ligand-density]
- 14. Molecular dynamics simulations of aggregation and viscosity ... [https://pubs.rsc.org/en/content/articlehtml/2024/ra/d3ra06483b]
- 15. Ligands for CsPbBr 3 perovskite quantum dots [https://www.sciencedirect.com/science/article/abs/pii/S1385894722053840]
- 16. Thermal tolerance of perovskite quantum dots dependent ... [https://www.nature.com/articles/s41467-023-37943-6]
- 17. Perovskite Quantum Dots Synthesis [https://www.sigmaaldrich.com/US/en/technical-documents/technical-article/materials-science-and-engineering/nanoparticle-and-microparticle-synthesis/synthesis-halide-perovskite-quantum-dots-display-applications?srsltid=AfmBOoqLfenV_Fxm203EN5yRerHjOoePMGOUpCE_btmHuxyItDyOAQUz]
- 18. Hot-Injection Synthesis Protocol for Green-Emitting Cesium ... [https://acs.figshare.com/collections/Hot-Injection_Synthesis_Protocol_for_Green-Emitting_Cesium_Lead_Bromide_Perovskite_Nanocrystals/6339087]
- 19. Hot-Injection Synthesis of Cesium Lead Halide Perovskite ... [https://www.mdpi.com/1996-1944/17/10/2173]
- 20. Surface manipulation and engineering strategies for high ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894724071651]
- 21. Impact of Molecular Ligands in the Synthesis and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9718301/]
- 22. Impact of Different Surface Ligands on the Optical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5507044/]
- 23. Toward High-Performances of Halide Light-Emitting Diodes [https://www.mdpi.com/2304-6740/11/6/230]
- 24. In situ 2D-perovskite-like ligands offer versatile passivation of ... [https://pubs.rsc.org/en/content/articlehtml/2025/el/d5el00135h]
- 25. Liquid bidentate ligand for full ligand coverage towards ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11704330/]
- 26. Polymerization‐Induced Direct Photolithography of Quantum ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12447694/]
- 27. Effect of the chemical specificity of benzoic acid and its ... [https://pubmed.ncbi.nlm.nih.gov/23376564/]
- 28. coated perovskite quantum dots for micro-LED display ... [https://www.sciencedirect.com/science/article/abs/pii/S2468023023001724]
- 29. Recent progress in group Ⅲ-nitride Micro-LED displays [https://www.sciencedirect.com/science/article/abs/pii/S1369800124009867]
- 30. Full-color micro-LED display with photo-patternable and ... [https://www.nature.com/articles/s41598-023-31945-6]
- 31. Color-conversion displays: current status and future outlook [https://www.nature.com/articles/s41377-024-01618-8]
- 32. Bifunctional ligand passivation enables robust-stability ... [https://www.sciencedirect.com/science/article/abs/pii/S0143720825003535]
- 33. Recent Advances in Ligand Design and Engineering in Lead ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8224438/]
- 34. In-Situ passivation of mixed-halide perovskite nanocrystals ... [https://www.sciencedirect.com/science/article/abs/pii/S1385894724023908]
- 35. Ligand-Assisted Purification of Mixed-Halide Perovskite ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12608481/]
- 36. Tailoring solvent-mediated ligand exchange for CsPbI3 ... [https://www.sciencedirect.com/science/article/pii/S2542435122002343]
- 37. Dual‐Strategy Direct Photocatalytic Patterning for Efficient ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12548516/]
- 38. Pioneering perovskite quantum dot nanosensors for heavy ... [https://pubs.rsc.org/en/content/articlehtml/2025/ra/d5ra06200d?page=search]
- 39. The impact of ligands on the synthesis and application of ... [https://pubs.rsc.org/en/content/articlehtml/2021/ta/d1ta05242j]
- 40. Enhanced stability and brightness for small CsPbX 3 ... [https://www.sciencedirect.com/science/article/abs/pii/S2542529325002330]
- 41. Approaching high-performance light-emitting devices upon ... [https://www.sciencedirect.com/science/article/abs/pii/S1748013222000767]
- 42. Perovskite Quantum Dots in Solar Cells - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC8895128/]
- 43. Issues of Using Benzyl Ether in Nanomaterials' Synthesis [https://pmc.ncbi.nlm.nih.gov/articles/PMC12529151/]
- 44. Molecular dynamics investigation of benzoic in confined spaces... acid [https://pubs.rsc.org/en/content/articlehtml/2023/cp/d3cp02886k]
- 45. Effect of PbSO4-Oleate Coverage on Cesium Lead Halide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8538890/]
- 46. Benzoic acid inhibits intrinsic ion migration for efficient and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11017186/]
- 47. Benzoic Acid Market Statistics 2025-2034 [https://www.statifacts.com/outlook/benzoic-acid-market]
- 48. An Introduction to Photoluminescence Quantum Yield (PLQY) [https://www.ossila.com/pages/plqy]
- 49. Stepwise−Process−Controlled Ligand Management ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10708130/]
- 50. Trap‐Assisted Charge Generation and Recombination in ... [https://www.semanticscholar.org/paper/Trap%E2%80%90Assisted-Charge-Generation-and-Recombination-Ma-Janssen/a43d38f65f2a5c708c5168516dd8eef1ce8b34b7]
- 51. Direct observation of trap-assisted recombination in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8203604/]
- 52. High-Performance Perovskite Quantum Dot Solar Cells ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9576836/]
- 53. Long-term ambient air-stable cubic CsPbBr3 perovskite ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9418586/]
- 54. In Situ Epitaxial Quantum Dot Passivation Enables Highly ... [https://www.mdpi.com/2079-4991/15/13/978]
- 55. Effect of Film Morphology on Electrical Conductivity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10780969/]
- 56. Review and principles of conductivity ... [https://www.biologic.net/documents/review-and-principles-of-conductivity-measurement-material-science-application-note-2/]
- 57. Determination of energy level alignment at metal/molecule ... [https://www.nature.com/articles/ncomms5161]
- 58. Current-voltage plots reveal details of the energy level ... [https://www.sciencedirect.com/science/article/pii/S2666523924000710]