Surface Morphology and Chemical Composition of Dental Biomaterials: A Comparative Analysis for Advanced Research
This article provides a comprehensive comparative analysis of the surface morphology and chemical composition of contemporary dental biomaterials, tailored for researchers, scientists, and drug development professionals.
Surface Morphology and Chemical Composition of Dental Biomaterials: A Comparative Analysis for Advanced Research
Abstract
This article provides a comprehensive comparative analysis of the surface morphology and chemical composition of contemporary dental biomaterials, tailored for researchers, scientists, and drug development professionals. It explores the fundamental principles governing material-tissue interactions, details advanced spectroscopic and microscopic characterization methodologies, and addresses challenges in material performance and biofilm formation. A systematic comparison of metallic, ceramic, and polymeric biomaterials highlights their distinct surface properties and biological behaviors. By integrating foundational knowledge with applied research, this review serves as a critical resource for guiding the development and clinical validation of next-generation dental biomaterials.
The Blueprint of Interaction: How Surface Properties Govern Biological Response
Defining Surface Morphology, Topography, and Chemical Composition in Dental Biomaterials
In dental biomaterials research, the surface properties of a material are critical determinants of its clinical performance and biological response. Surface morphology and topography refer to the physical structure and geometric features of a surface, encompassing characteristics such as roughness, pattern, and texture at the micro- and nano-scale. Chemical composition defines the molecular and elemental makeup of the surface, which directly influences its bioactivity, corrosion resistance, and interfacial interactions [1] [2] [3]. These surface parameters collectively govern fundamental biological processes including protein adsorption, cellular adhesion, proliferation, differentiation, and bacterial colonization [2] [3]. A comprehensive understanding of these properties is therefore essential for developing advanced dental biomaterials with optimized functionality and improved clinical outcomes.
This guide provides a comparative analysis of surface characteristics across major dental biomaterial classes, detailing standardized experimental protocols for their characterization, and presenting quantitative data to inform research and development efforts.
Comparative Surface Analysis of Dental Biomaterials
The biological performance of dental biomaterials is intrinsically linked to their surface properties. The following section compares key material classes based on quantitative measurements of surface topography, wettability, and chemical composition.
Surface Roughness and Hardness
Table 1: Surface Roughness and Hardness of Denture Base Resins
| Material Type | Fabrication Method | Surface Roughness (Ra in µm) | Vickers Hardness | Key Characteristics |
|---|---|---|---|---|
| PMMA [4] | CAD/CAM Milled | 0.16 ± 0.014 | Highest values | Superior surface smoothness and hardness |
| PMMA [4] | Conventional Heat-polymerized | 0.21 ± 0.019 | Intermediate values | Roughest surface among PMMA groups |
| PMMA [4] | 3D-Printed | Intermediate roughness | Lowest values | Higher hydrophobicity |
| 3D-Printed Soft Liner [5] | DLP 3D Printing | Increased roughness | Significantly higher Shore A hardness | Rougher than conventional silicone-based liners |
| Silicone-based Soft Liner [5] | Conventional | Maintains smoothest surface | Lower Shore A hardness | Maintains greatest color stability |
Surface Wettability and Energy
Table 2: Wettability and Surface Energy of Dental Biomaterials
| Material Class | Specific Material | Contact Angle (°) | Surface Characteristics | Biological Correlation |
|---|---|---|---|---|
| PMMA Denture Base [4] | 3D-Printed PMMA | 73.94 ± 2.29 | Highest hydrophobicity | Potential for reduced plaque adhesion |
| PMMA Denture Base [4] | CAD/CAM PMMA | 73.26 ± 2.37 | Hydrophobic | - |
| PMMA Denture Base [4] | Conventional PMMA | 68.38 ± 1.93 | Hydrophobic | - |
| Modified Surfaces [6] | Laser-Processed 'Grid' | 158.63 ± 1.26 | Super-hydrophobic | Minimal cell/culture medium spread |
| Modified Surfaces [6] | Laser-Processed 'Line' | Significantly lower | Super-hydrophilic | Cell culture medium spread across nearly entire surface |
| Acrylic Resins [2] | Various (after aging) | 65.5 - 84.7 | High critical surface energy | Zone of good bacterial adhesion |
| Composites [2] | Bis-GMA based | ~49.1 - 58.8 | Lower critical surface energy | Zone of poor bacterial adhesion |
Zirconia vs. Titanium: A Surface-Centric Comparison
Table 3: Surface and Biological Properties of Titanium and Zirconia Implants
| Property | Titanium Implants | Zirconia Implants |
|---|---|---|
| Base Material | Commercially pure Ti (cpTi) or Ti-6Al-4V alloy [3] | Zirconium dioxide (ZrO₂), often Yttria-stabilized (YSZ) [1] |
| Surface Oxide | TiO₂ (Titanium dioxide) [3] | ZrO₂ (Zirconium dioxide) [1] |
| Aesthetics | Grayish, potentially visible through gingiva [1] | Tooth-like color, superior aesthetics [1] |
| Corrosion Behavior | Susceptible to galvanic corrosion in saliva [1] | Excellent long-term corrosion resistance, dense oxide layer [1] |
| Biofilm Formation | Higher bacterial colonization [1] | Resists bacterial colonization more effectively [1] |
| Surface Aging | Hydrocarbon accumulation, loss of hydrophilicity over time [3] | Highly stable surface, no significant aging reported [1] |
| Surface Activation | UV photofunctionalization reverses aging, restores bioactivity [3] | UV treatment enhances hydrophilicity and reduces surface carbon [1] |
| Typical BIC | 45-65% with modern microrough surfaces [3] | Comparable to titanium; 48-53% in animal studies [1] |
Experimental Protocols for Surface Characterization
Standardized methodologies are crucial for obtaining reliable and comparable data on biomaterial surfaces. The following sections detail common experimental protocols.
Protocol 1: Surface Roughness and Hardness Testing
Methodology for Denture Base Resins (Based on [4])
- Specimen Preparation: Prepare 120 standardized specimens (25 × 25 × 3 mm). Divide into groups (n=40 per group) for different materials (e.g., conventional, CAD/CAM, 3D-printed PMMA). Finish, polish, and store in distilled water.
- Aging: Subject specimens to thermocycling and immersion in artificial saliva to simulate oral conditions.
- Surface Roughness Measurement: Use a contact profilometer (e.g., Mitutoyo Surftest SJ-210). Traverse the stylus across the specimen surface and record the average roughness (Ra) in micrometers.
- Surface Hardness Measurement: Use a Vickers hardness tester (e.g., Mitutoyo HM-200). Apply a load for a specific dwell time and measure the diagonal of the indentation to calculate hardness.
Protocol 2: Surface Wettability and Energy Analysis
Methodology for Polymer-Based Dental Materials (Based on [2])
- Specimen Preparation: Prepare beam-shaped samples (approx. 30 × 10 × 5 mm) of materials (acrylic resins, composites, etc.) per manufacturers' instructions.
- Aging/Incubation: Incubate specimens in simulated oral environments (e.g., 0.9% NaCl, orange juice) at 37°C for up to 21 days.
- Contact Angle Measurement: Use the sessile drop method with a goniometer. Deposit a droplet of purified water on the tested surface and measure the contact angle. Lower angles indicate higher wettability/hydrophilicity.
- Surface Free Energy (SFE) Calculation: Estimate SFE using models like Owens–Wendt, van Oss–Chaudhury–Good, or Zisman's method, based on contact angle data with multiple liquids.
Protocol 3: Surface Modification and Biological Evaluation
Methodology for Laser-Modified Biomaterials (Based on [6])
- Surface Modification: Use a High-Focus Laser Scanning (HFLS) system to create precise micro-patterns ('Line' or 'Grid') on the biomaterial surface.
- Surface Characterization: Perform morphological analysis via microscopy and surface wettability via contact angle measurements.
- Cell Response Assays:
- Cell Adhesion: Seed cells on modified surfaces, incubate, fix, and count adhered cells.
- Cell Proliferation: Use assays (e.g., MTT) to measure metabolic activity after specific time periods.
- Cell Migration: Perform scratch/wound healing assays to observe cell movement.
- Drug Delivery Functionality:
- Drug Holding & Release: Load a model drug (e.g., Prednisolone), immerse in buffer, and measure drug release over time via UV-Vis spectroscopy.
- Anti-inflammatory Effect: Analyze conditioned media from drug-release studies using Western Blot to assess inflammatory cytokine expression.
The following diagram illustrates the logical workflow and key interactions between surface properties, characterization techniques, and biological outcomes in dental biomaterials research.
Relationship between surface properties, characterization methods, and biological outcomes in dental biomaterials.
Essential Reagents and Research Tools
Table 4: Key Reagent Solutions and Materials for Surface Science Research
| Reagent/Material | Function in Research | Application Example |
|---|---|---|
| Artificial Saliva [7] | Simulates oral electrolyte environment for aging and wear testing. | Corrosion studies, wear testing under simulated oral conditions. |
| Stainless Steel Antagonists [7] | Standardized counterface material for wear simulation. | Ball-on-disc, block-on-ring, and reciprocating wear tests. |
| Silicon Carbide Polishing Paper [7] | Creates standardized surface finishes on specimen materials. | Preparing specimens with defined surface roughness before testing. |
| AgVO₃ Nanoparticles [8] | Biofunctionalization additive to enhance antimicrobial properties. | Modifying alginate impression materials to create self-disinfecting surfaces. |
| UV Light Source [1] [3] | Surface photofunctionalization to increase hydrophilicity and bioactivity. | Treating titanium or zirconia surfaces to reverse aging and enhance osteoconductivity. |
| Instant Coffee Granules [5] | Staining agent for color stability testing of polymeric materials. | Evaluating color change (ΔE) of denture base resins and soft liners. |
| PET-G Material [2] | Polymer used for removable orthodontic retainers. | Studying surface properties and aging of orthodontic devices. |
| Bone Morphogenetic Protein-2 (BMP-2) [1] | Growth factor used to biofunctionalize material surfaces. | Coating on sandblasted Y-TZP to enhance osteoblast differentiation. |
Discussion and Clinical Implications
The data reveals critical relationships between surface properties and biological performance. Surface topography directly influences cellular and microbial responses. Roughness values (Ra) below 0.2 μm are theoretically desirable to minimize plaque accumulation [2], a threshold that CAD/CAM-milled PMMA reliably meets [4]. Increased roughness generally promotes osteoblastic differentiation on implant surfaces but can simultaneously suppress cellular proliferation, highlighting a fundamental trade-off in implant design [3].
Surface wettability and energy are pivotal for biointegration. Hydrophilic surfaces (low contact angle) typically enhance protein adsorption and cell attachment [6] [3]. The phenomenon of "biological aging" of titanium, where hydrocarbon contamination over time increases hydrophobicity, significantly reduces osteoconductivity. This degradation is reversible via UV photofunctionalization, which restores super-hydrophilicity and bioactivity [3]. Similarly, UV treatment of zirconia enhances its hydrophilicity and reduces surface carbon, improving osteoblast response [1].
Chemical composition underpins corrosion resistance and biocompatibility. Zirconia's dense, stable oxide layer (ZrO₂) provides exceptional long-term corrosion resistance, even in acidic environments, minimizing metal ion release [1]. Titanium's biocompatibility also stems from its passive TiO₂ layer, though it remains susceptible to galvanic corrosion in the oral environment [1] [3]. Recent strategies focus on biofunctionalization, such as incorporating AgVO₃ nanoparticles into alginates to create antimicrobial surfaces [8] or applying BMP-2 to zirconia to boost osteogenesis [1].
These surface properties are not independent; they function synergistically. The 3D Theory of Osseointegration conceptualizes this synergy, identifying material composition (1), surface topography (2), and time-dependent physicochemical properties (3) as interdependent dimensions governing implant integration [3]. A holistic approach that optimizes all three dimensions is essential for advancing the next generation of dental biomaterials.
The Critical Role of Surface Properties in Osseointegration and Biofilm Formation
The long-term success of dental implants is fundamentally governed by the complex interplay between the implant surface and the biological environment. While implant design and surgical protocol are crucial, the surface properties ultimately determine the ability to achieve osseointegration while resisting biofilm formation. This biological response is directed by a triad of surface characteristics: topography (physical structure and roughness), chemical composition (material and surface chemistry), and physicochemical properties (wettability and charge) [3]. The emerging "3D Theory of Osseointegration" conceptualizes these factors as interdependent dimensions that dynamically influence biological outcomes [3]. A profound clinical challenge persists: despite advancements, bone-implant contact (BIC) with modern microrough titanium implants typically reaches only 45-65%, leaving substantial surface area vulnerable to soft tissue intervention or microbial colonization [3]. This review systematically compares how specific surface modifications simultaneously influence osteogenic integration and microbial resistance, providing a framework for developing next-generation dental implant surfaces.
Surface Properties and Osseointegration: Mechanisms and Comparative Performance
The Dimensional Framework of Osseointegration
Osseointegration is mediated through three interconnected dimensions:
- Dimension 1 (Material): Commercially pure titanium (cpTi) and titanium alloys (Ti-6Al-4V) remain the gold standard due to their biocompatibility and self-passivating oxide layer (TiO₂) [3].
- Dimension 2 (Topography): Surface roughness at the micro- and nano-scale directly influences cellular response. While increased roughness promotes osteoblastic differentiation, it can simultaneously suppress cellular proliferation [3].
- Dimension 3 (Physicochemical Properties): Time-dependent biological aging of titanium surfaces leads to hydrocarbon accumulation and loss of hydrophilicity, significantly diminishing osteoconductivity. This degradation is reversible through techniques like UV photofunctionalization [3].
Comparative Performance of Surface Modifications Under Challenging Conditions
Surface modifications must maintain efficacy under biologically challenging conditions such as diabetes, which alters bone metabolism and impairs healing. The following table summarizes experimental data on surface performance under diabetic conditions:
Table 1: Surface Modification Efficacy Under Diabetic Conditions
| Surface Modification | Key Osteogenic Markers | Experimental Model | Reported Enhancement |
|---|---|---|---|
| Biomimetic Coatings | ALP, OPN, OCN | In vitro high-glucose microenvironment | Increased expression of osteogenic biomarkers [9] |
| UV Photofunctionalization | Not Specified | In vitro high-glucose microenvironment | Enhanced osteogenic response [9] |
| Sandblasted & Acid-Etched (SLA) | ALP, OPN, OCN | Systematic review of in vitro studies | Consistent increase in osteogenic factors [9] |
The data indicates that surface modifications can partially counteract the inhibitory effects of diabetic conditions on osseointegration. The biomimetic coatings and UV photofunctionalization show particular promise in enhancing the expression of critical osteogenic markers including alkaline phosphatase (ALP), osteopontin (OPN), and osteocalcin (OCN) [9].
Experimental Protocols for Assessing Osseointegration
Standardized methodologies are essential for evaluating the osseointegration potential of modified surfaces:
In Vitro Osteogenic Marker Analysis:
- Purpose: Quantify the expression of osteogenic differentiation markers.
- Methodology: Culture osteoblast precursor cells on test surfaces under high-glucose conditions (simulating diabetes). Measure ALP activity via enzymatic assays, and quantify OPN and OCN expression using techniques like ELISA or immunofluorescence at specific time points (e.g., 7, 14, 21 days) [9].
- Output: Quantitative data on osteogenic differentiation potential.
Surface Wettability and Contamination Assessment:
- Purpose: Evaluate the physicochemical surface state critical for bioactivity.
- Methodology: Measure contact angle to determine hydrophilicity/hydrophobicity. Analyze surface carbon content using techniques like X-ray Photoelectron Spectroscopy (XPS/ESCA) to assess hydrocarbon contamination from biological aging [3].
- Output: Quantitative thresholds for optimal surface bioactivity (e.g., carbon content <30%, contact angle <30° indicating super-hydrophilicity) [3].
Surface Properties and Biofilm Formation: A Complex Interrelationship
Dynamics of Early Biofilm Formation on Dental Implants
Biofilm formation on dental implants is a dynamic, patient-specific process that begins within minutes of implantation [10]. Advanced sequencing technologies have revealed that early biofilm communities on implant abutments are remarkably complex, diverse, and subject-specific, with studies identifying 371 different bacterial species in developing biofilms [10]. The biofilm structure evolves significantly over time, with mean biofilm volume increasing by 44% between the first and second week, followed by a further 30% increase between the second and third week [10]. These early communities include numerous disease-associated species, which can comprise up to 21.7% of the microbial community in some samples, highlighting the critical importance of the early post-operative period for infection prevention [10].
The succession of bacterial colonization follows a defined sequence:
- Early colonizers (minutes to hours): Predominantly Streptococcus and Actinomyces species [10].
- Secondary colonizers (days to weeks): Genera including Fusobacterium, Neisseria, Veillonella, and Prevotella [10].
- Mature biofilms (weeks to months): Characterized by fully developed extracellular polymer matrix with various microcolonies, voids, and channels [10].
Surface Characteristics Influencing Biofilm Adhesion
Multiple surface parameters influence the initial attachment and development of biofilms:
Table 2: Surface Properties and Their Influence on Biofilm Formation
| Surface Property | Impact on Biofilm Formation | Experimental Evidence |
|---|---|---|
| Roughness | Increased roughness generally enhances bacterial adhesion and biofilm stability by providing protected niches | Clinical studies show complex, three-dimensional biofilm structures developing on implant surfaces [10] |
| Hydrophobicity/Hydrophilicity | Hydrophobic surfaces typically promote initial bacterial attachment; hydrophilic surfaces may reduce adhesion | UV photofunctionalization creating super-hydrophilic surfaces reduces bacterial adhesion [3] |
| Chemical Composition | Surface chemistry influences protein adsorption which subsequently affects bacterial adhesion; antimicrobial elements can disrupt biofilm formation | Silver-containing coatings (e.g., TiN:Ag) show potential for antimicrobial activity [11] |
| Surface Charge | Negative surface charges may repel negatively charged bacterial cells | Closely related to physicochemical properties modified by biological aging and treatments [3] |
Methodologies for Biofilm Characterization
Comprehensive biofilm analysis requires multimodal approaches:
Confocal Laser Scanning Microscopy (CLSM) for 3D Structure:
- Purpose: Characterize three-dimensional biofilm architecture, volume, viability, and surface coverage.
- Methodology: Stain biofilms with fluorescent viability markers (e.g., SYTO 9/propidium iodide). Capture Z-stack images at multiple locations on the implant surface. Reconstruct 3D models and quantify parameters using image analysis software [10].
- Output: Quantitative data on biofilm volume, viability, and covered surface area; visualization of microcolonies, voids, and channels.
Full-Length 16S rRNA Gene Sequencing:
- Purpose: Achieve high-resolution taxonomic characterization of biofilm communities down to Amplicon Sequence Variants (ASVs).
- Methodology: Extract DNA from biofilm samples, amplify full-length 16S rRNA gene, and sequence using long-read platforms (e.g., PacBio). Process sequences using bioinformatics pipelines to resolve ASVs and perform taxonomic assignment [10].
- Output: Comprehensive, high-resolution microbial community composition; identification of early colonizers and disease-associated species; analysis of patient-specific variations.
The Researcher's Toolkit: Essential Methods for Surface Characterization
Advanced Techniques for Surface Analysis
Comprehensive characterization of dental implant surfaces requires multiple complementary analytical techniques:
Table 3: Essential Surface Characterization Methods
| Technique | Primary Application | Key Parameters Measured | Considerations |
|---|---|---|---|
| Scanning Electron Microscopy (SEM) | Surface morphology visualization | High-resolution topographical images | Limited to 2D without 3D reconstruction; requires specialized software for roughness quantification [12] |
| ImageJ Software with Plugins | Surface roughness quantification from SEM images | Ra (average roughness), Rq (root mean square roughness) | Requires methodology standardization; different plugins (SurfCharJ, roughness/waviness) yield different values [12] |
| Contact Angle Goniometry | Surface wettability assessment | Contact angle measurements (hydrophilicity/hydrophobicity) | Critical for evaluating biological aging and UV photofunctionalization efficacy [3] |
| X-ray Photoelectron Spectroscopy (XPS/ESCA) | Surface chemical composition | Elemental and chemical state composition; hydrocarbon contamination levels | Detects biological aging through carbon content quantification [3] [11] |
| Atomic Force Microscopy (AFM) | Nanoscale topography and roughness | High-resolution 3D surface topography | Provides superior resolution but higher cost and operational complexity [12] |
| Confocal Laser Scanning Microscopy (CLSM) | 3D biofilm structure analysis | Biofilm volume, viability, surface coverage | Enables live imaging of biofilms without disruption [10] |
Standardized Protocol for Surface Roughness Analysis Using ImageJ
To ensure consistency in surface roughness measurements, the following standardized protocol is recommended:
- Image Acquisition: Capture SEM images at standardized magnifications (e.g., 500×, 1000×, 2000×, 4000×) and export in TIFF format [12].
- Image Pre-processing: Import images into ImageJ and convert to 32-bit. Apply median filter (radius 0.2) to reduce noise [12].
- Scale Calibration: Adjust pixel scale to micrometers based on SEM image magnification and scale bar.
- Roughness Analysis:
- For SurfCharJ plugin: Draw a diagonal line across the image and run the plugin with appropriate sampling lengths (90µm for 500×, 40µm for 1000×, 20µm for 2000×, 10µm for 4000×) [12].
- For roughness/waviness separation: Use a cutoff value of 4 to separate waviness and roughness components before measurement [12].
- 3D Visualization: Generate 3D surface plots using Surface Plot or interactive 3D Surface Plot plugins.
- Data Reporting: Report both Ra and Rq values with specification of the plugin and methodology used.
The critical role of surface properties in governing both osseointegration and biofilm formation presents a fundamental challenge in implant dentistry: surface characteristics that enhance bone integration may simultaneously promote microbial colonization. The current evidence indicates that microrough surfaces, while promoting osteoblastic differentiation, can also provide protected niches for biofilm development [3] [10]. This paradox underscores the need for sophisticated surface engineering approaches that maximize osteoconductivity while minimizing microbial adhesion.
Future directions should focus on smart surface designs that incorporate:
- Temporal functionality: Surfaces that are initially hydrophilic to promote cell attachment but can modulate properties over time.
- Zone-specific topography: Different surface characteristics at transgingival versus bone-integrated regions.
- Antimicrobial strategies: That do not compromise osteogenesis, such as topographical features that mechanically disrupt biofilm formation without inhibiting bone cell adhesion.
The integration of advanced characterization methodologies with standardized protocols will enable more precise correlations between specific surface parameters and biological outcomes. As the field moves toward personalized implant solutions, understanding the complex interplay between surface properties and host response will be paramount in developing next-generation implants that achieve the elusive goal of 100% bone-implant contact while resisting microbial colonization.
The selection of appropriate materials is fundamental to advancements in scientific and industrial fields, from drug development to medical device manufacturing. Understanding the intrinsic properties of the three major material classes—metals, ceramics, and polymers—enables researchers to make informed decisions tailored to specific application requirements. This comparative guide provides an objective analysis of these material classes, with particular emphasis on their performance in dental materials research, where surface morphology and chemical composition critically determine biological interactions and clinical outcomes [13]. The increasing demand for biocompatible solutions in restorative dentistry and medical implants has accelerated research into optimizing these materials for enhanced performance, durability, and tissue compatibility [14] [13].
Fundamental Properties Comparison
The distinctive characteristics of metals, ceramics, and polymers stem from their atomic bonding and microstructural differences. Metals possess metallic bonding, enabling excellent electrical and thermal conductivity. Ceramics feature ionic/covalent bonding, providing high hardness and thermal stability. Polymers have covalent bonding with weak secondary forces, resulting in low density and high flexibility [15].
Table 1: Comparative Properties of Major Material Classes
| Material Property | Metals | Ceramics | Polymers |
|---|---|---|---|
| Biocompatibility | - | + | o |
| Hardness | - | + | - |
| Wear Resistance | - | + | - |
| Density / Weight | - | + | + |
| Dimensional Stability | - | + | - |
| Chemical Resistance | - | + | - |
| High Temperature Resistance | o | + | - |
| Electrical Insulation | Poor | Excellent | Good |
| Corrosion Resistance | Low | High | Variable |
Note: + indicates superior performance; o moderate performance; - inferior performance relative to other classes [15]
Ceramics demonstrate exceptional performance in most categories, particularly hardness, wear resistance, and chemical stability, making them invaluable for demanding applications. Technical ceramics exhibit exceptional corrosion resistance, maintaining performance in aggressive chemical environments where metals corrode and plastics degrade [15]. Their high hardness and wear resistance extend service life in mechanical applications, outperforming conventional metals and plastics in abrasive environments [15].
Metals provide superior strength and toughness but suffer from higher density and susceptibility to corrosion. Polymers offer the advantages of low weight and ease of processing but are limited by poor temperature resistance and mechanical properties compared to ceramics and metals [15].
Performance in Dental Applications
Dental Material Requirements
The oral environment presents unique challenges for materials, including constant exposure to moisture, temperature fluctuations, mechanical stresses from chewing, and a diverse microbiome [13]. Dental materials must withstand these conditions while maintaining structural integrity and biocompatibility. The seamless integration with biological tissues is paramount for long-term success in dental applications [13].
Table 2: Dental Material Applications and Performance
| Material Class | Specific Materials | Dental Applications | Key Advantages | Limitations |
|---|---|---|---|---|
| Metals | Titanium alloys, Gold, Cobalt-Chromium, Nickel-Chromium | Implants, Crowns, Bridges, Partial dentures [13] | Superior strength, durability, osseointegration (Ti) [13] | Potential corrosion, metal sensitivity, poor aesthetics [13] |
| Ceramics | Zirconia, Porcelain, Lithium disilicate, Alumina | Crowns, Veneers, Bridges, Implants, Inlays/Onlays [13] [16] | Excellent aesthetics, biocompatibility, wear resistance, hypoallergenic [13] | Brittleness, processing challenges [13] |
| Polymers | PMMA, Composite resins, Polyurethane | Dentures, Temporary restorations, Fillings, Clear aligners [13] | Aesthetics, ease of fabrication, bonding to tooth structure [13] | Wear susceptibility, degradation over time [13] |
Biocompatibility and Clinical Performance
Metals represent a cornerstone of dental materials, with titanium and its alloys being particularly valuable for dental implants due to their ability to osseointegrate—forming a direct bond with bone [13]. This property provides a stable foundation for prosthetic teeth. However, concerns about potential nickel sensitivity from some alloys have led to increased preference for titanium or metal-free alternatives [13].
Ceramics have gained significant popularity in dentistry due to their excellent aesthetic properties and biocompatibility. Zirconia, in particular, has emerged as a leading material with exceptional strength, wear resistance, and hypoallergenic properties, making it suitable for patients with metal sensitivities [13]. Dental ceramics are chemically inert, reducing the risk of adverse tissue reactions, and can be engineered to mimic the translucency and color of natural teeth [13].
Polymers and composite resins are widely used in restorative dentistry for direct fillings and in orthodontics for clear aligners. These materials bond well to tooth structure and can be color-matched to natural teeth [13]. Recent advancements in polymer chemistry have led to more durable and wear-resistant formulations, improving their performance in high-stress oral environments [13].
Experimental Analysis and Methodologies
Research Reagent Solutions for Material Testing
Table 3: Essential Research Reagents and Materials for Dental Material Analysis
| Reagent/Material | Function/Application | Research Context |
|---|---|---|
| Aluminum Oxide (Al₂O₃) | Ceramic reinforcement material for composites [17] | Structural analysis and compression testing of ceramic specimens [17] |
| Photosensitive Composite Resin | Matrix for vat photopolymerization of ceramic parts [17] | Additive manufacturing of ceramic components via stereolithography [17] |
| Acetone | Solvent for extraction of bonding agent from green ceramic parts [17] | Post-processing of additively manufactured ceramic components [17] |
| Isopropyl Alcohol | Rinsing agent for uncured resin from printed parts [17] | Post-processing of specimens fabricated via vat photopolymerization [17] |
| Zirconia (ZrO₂) Suspension | Feedstock for additive manufacturing of dental restorations [16] | Fabrication of crowns, bridges, and implants via AM technologies [16] |
| Polyethylene Glycol | Additive to minimize "coffee staining" defects in inkjet printing [16] | Improvement of ceramic ink formulation for additive manufacturing [16] |
Key Experimental Protocols
Additive Manufacturing of Ceramic Components
Specimen Preparation: Research specimens are typically designed according to international standards (e.g., EN 843-4:2005) using CAD software, then exported as STL files for additive manufacturing [17].
Material Extrusion (MEX) Protocol:
- Printer: Creality CR-10S with modified filament feeding system
- Nozzle temperature: 175°C
- Layer height: 0.2 mm
- Print speed: 5 mm/s
- Infill density: 100%
- Post-processing: Solvent extraction in acetone at 42°C for 24 hours (mass loss should not exceed 9.8%) [17]
Vat Photopolymerization (VPP) Protocol:
- Printer: Form 2 by Formlabs
- Layer thickness: 0.1 mm
- Resin type: Photopolymer methacrylate resin
- Exposure time: 15 seconds
- Post-processing: Rinsing with isopropyl alcohol, no additional UV exposure required [17]
Sintering Process:
- VPP specimens: Maximum temperature of 1271°C using controlled heating program [17]
- MEX specimens: Temperature of 1540°C in furnace with Kanthal Super heating elements [17]
Mechanical and Structural Characterization
Compression Testing: Cylindrical specimens with diameters of 10 mm and varying heights (5, 10, 15 mm) are tested to determine compression strength [17]. MEX-fabricated Al₂O₃ specimens demonstrate compression strength up to 168 MPa, while VPP specimens reach 81 MPa [17].
Microstructural Analysis: Specimens are cut along printed layers (Z-axis), mounted in resin, and ground using abrasive water paper with grit sizes from 240 to 1200 [17]. Analysis reveals distinct microstructural differences:
- MEX specimens: Similar microstructures before and after heat treatment, with sintering primarily affecting grain size [17]
- VPP specimens: Irregular grain shapes before sintering that fuse during heat treatment, forming solid structures where individual grains become indistinguishable [17]
Density Measurement: Using automatic helium pycnometer (AccuPyc 1340 II) with cylindrical measurement chamber, conducting measurements in two stages (10 purges and 700 measurement cycles) at filling pressure of 0.13 MPa [17].
Figure 1: Experimental workflow for additive manufacturing and analysis of ceramic materials
Advanced Ceramic Composites and Surface Morphology
Recent research has focused on developing advanced ceramic composites with enhanced properties through reinforcement strategies. In-situ synthesis of reinforcement phases represents a novel approach for improving tribological performance and surface characteristics [18].
Reinforced Ceramic Matrix Composites
The in-situ synthesis of BN-Si₃N4 reinforced SiC-Al₂O₃ composites demonstrates significant advancements in ceramic matrix technology. This approach involves:
Fabrication Process:
- Powder metallurgy approach using high-energy ball milling
- In-situ formation of BN and Si₃N₄ through nitridation of Si metal and B₂O₃
- Pressure-less sintering process for consolidation [18]
Microstructural Characteristics:
- SEM images reveal well-dispersed BN and Si₃N₄ in the SiC matrix
- Strong bonding observed at the Al₂O₃-SiC interface
- Enhanced surface morphological characteristics [18]
Performance Outcomes:
- Synergistic interaction between in-situ formed BN and Si₃N₄ phases
- BN provides solid lubrication effect
- Si₃N₄ contributes to hardness and toughness
- Significant improvement in tribological performance [18]
Surface Morphology in Dental Ceramics
Surface morphology plays a critical role in the clinical performance of dental materials. For zirconia-based dental restorations, additive manufacturing techniques present both opportunities and challenges:
Surface Quality Parameters:
- SLA-fabricated ZrO₂ exhibits Vickers hardness of 1398 HV and flexural strength of 200.14 MPa [16]
- Surface roughness measures approximately 2.06 μm for SLA-produced zirconia [16]
- DLP-fabricated ZrO₂ demonstrates higher flexural strength of 831 ± 74 MPa [16]
Additive Manufacturing Challenges:
- Porosity remains a significant concern in AM-fabricated ceramics
- Reduced mechanical strength compared to conventionally processed materials
- Variability in sintering outcomes affects final properties [16]
Figure 2: Relationship between processing parameters, microstructure, and final properties of ceramic materials
This comparative analysis demonstrates that each material class offers distinct advantages and limitations for specific applications. Ceramics excel in environments demanding high hardness, wear resistance, and chemical stability, while metals provide superior strength and toughness. Polymers offer advantages in processing flexibility and weight-sensitive applications. In dental materials research, the trend toward metal-free restorations has accelerated the development of advanced ceramics and composites with improved aesthetics and biocompatibility. The emergence of additive manufacturing technologies has further expanded design possibilities for complex dental restorations, though challenges remain in achieving consistent mechanical properties comparable to conventionally manufactured materials. Future research directions include developing nanoscale composites, optimizing in-situ reinforcement strategies, and refining additive manufacturing protocols to enhance the performance and reliability of materials across all three classes.
The adhesion of bacteria to solid surfaces represents the critical initial step in biofilm formation, a process with profound implications across environmental microbiology, biomedical device integration, and dental material performance [19] [20]. While multiple factors influence bacterial adhesion, the thermodynamic parameter of Surface Free Energy (SFE) has emerged as a primary predictor of cellular attachment behavior [19] [21]. SFE, defined as the work required to increase the surface area of a solid phase, directly influences a material's wettability and ultimately governs its interactions with biological systems [22]. In dental materials research, understanding and controlling SFE provides a powerful approach to designing surfaces that either promote tissue integration or resist microbial colonization [20] [23]. This guide objectively compares how SFE and wettability across different material classes influence bacterial adhesion, providing researchers with experimental data and methodologies to inform material selection and surface engineering strategies.
Thermodynamic Principles: SFE as a Predictor of Bacterial Adhesion
Fundamental Relationship Between SFE and Wettability
Surface Free Energy and surface tension are physically equivalent concepts, typically applied to solids and liquids respectively [22]. Both are measured in mJ/m² (millijoule per square meter) or equivalently mN/m (millinewton per meter). The SFE of a material directly determines its wettability, which is quantitatively measured by the contact angle (θ) formed when a liquid droplet rests on the solid surface [22]. This relationship is described by Young's equation:
γlvcosθ = γsv - γsl
where γlv is the liquid-vapor surface tension, γsv is the solid-vapor surface free energy, and γsl is the solid-liquid interfacial tension [24] [22]. A lower contact angle indicates better wettability and typically corresponds to a higher SFE solid surface [22].
SFE Difference Theory for Bacterial Adhesion
Research has demonstrated that bacterial adhesion is fundamentally mediated by the SFE difference between bacterial cells and substrate surfaces [19]. The thermodynamic adhesion energy (ΔFadh), which predicts whether adhesion will occur spontaneously, can be calculated using Neumann's equation of state [19]:
ΔFadh = 2√(γbvγlv)e-β(γbv-γlv)2 + 2√(γsvγlv)e-β(γsv-γlv)2 - 2√(γbvγsv)e-β(γbv-γsv)2 - 2γlv
where γbv, γsv, and γlv represent the SFEs of bacteria, solid substrate, and liquid medium, respectively, and β is a constant (0.0001247 (mJ/m²)⁻²) [19]. The central finding is that smaller SFE differences between bacteria and substrate correlate with increased adhesion [19]. This principle provides a thermodynamic foundation for predicting and controlling bacterial adhesion across material systems.
Table 1: Surface Free Energy Values for Common Dental Materials and Bacterial Cells
| Material / Microorganism | Surface Free Energy (mJ/m²) | Surface Treatment | Contact Angle (°) |
|---|---|---|---|
| Zirconia (polished) | - | No treatment | - |
| Zirconia (GCSD-coated) | Significantly increased | Glass-ceramic spray deposition | Reduced vs. polished |
| 3D-printed resin (untreated) | Low | None | High |
| 3D-printed resin (treated) | Increased | Sandblasting + silane | Reduced |
| Clean glass | 70 ± 1 | - | - |
| Silanized glass | 38 ± 1 | - | - |
| Pseudomonas putida KT2440 | 49.8 | - | - |
| Escherichia coli DH5α | 46.9 | - | - |
| Staphylococcus epidermidis | 45.2 | - | - |
Experimental Comparisons: Bacterial Adhesion Across Material Classes
Metallic and Ceramic Dental Materials
Zirconia's biological inertness and low SFE contribute to suboptimal protein adsorption and cell attachment, despite its advantageous mechanical properties for dental restorations [23]. Surface modification techniques like Glass-Ceramic Spray Deposition (GCSD) significantly enhance hydrophilicity and SFE, improving osteoblast viability but potentially creating surfaces more susceptible to bacterial adhesion [23]. Studies on zirconia abutments demonstrate that smoother, polished surfaces with lower SFE reduce bacterial accumulation in the transmucosal region, critical for peri-implant health [23]. This creates a design paradox: higher SFE surfaces promote tissue integration but may increase bacterial adhesion risk, necessitating balanced surface engineering approaches.
Polymeric and Resin-Based Materials
Resin composites, commonly used in dental restorations, demonstrate variable bacterial adhesion depending on their composition and surface energy [20]. 3D-printed resins present particular challenges due to layer-by-layer polymerization, incomplete monomer conversion, and oxygen inhibition layers that create heterogeneous surface energies [25]. Surface treatments like alumina air abrasion (sandblasting) combined with silane coupling agents significantly increase SFE and improve bond strength while potentially influencing bacterial adhesion patterns [25]. Research shows that untreated resin surfaces typically exhibit higher bacterial adhesion, while controlled surface modifications can reduce microbial colonization [20] [25].
Table 2: Bacterial Adhesion Response to Material Wettability
| Material Type | Surface Energy | Wettability | Bacterial Adhesion Trend |
|---|---|---|---|
| Superhydrophobic textiles | Very low | Contact angle >150° | Significantly reduced adhesion |
| Moderate hydrophobic surfaces | Low | Contact angle 90-150° | Immense adherence, especially Gram-negative |
| Hydrophilic surfaces | High | Contact angle <90° | Research shows conflicting results |
| High SFE metals/ceramics | Very high | Near-zero contact angle | Generally increased adhesion |
| 3D-printed resins (treated) | Medium-high | Reduced contact angle | Variable by bacterial species |
Textile and Industrial Materials
Studies on electrospun fibrous webs of polystyrene (PS) and polylactic acid (PLA) demonstrate that wettability is the primary factor influencing bacterial adhesion, where hydrophilic surfaces typically result in considerably higher adhesion [21]. Beyond SFE, pore characteristics including total pore volume and pore size significantly affect bacterial adherence and retention, with compact spatial fiber distribution limiting cell intrusion [21]. Superhydrophobic textiles with reduced total pore volume and smaller pore size effectively circumvent bacterial adhesion through combined surface energy and topological effects [21].
Methodological Approaches: Measuring SFE and Bacterial Adhesion
Determining Surface Free Energy
Multiple established methodologies exist for experimental SFE determination:
Contact Angle Goniometry: The most common approach involves measuring contact angles with multiple liquids of known surface tension components [26] [24]. The Owens-Wendt-Rabel-Kaelble (OWRK) method is frequently used, calculating SFE from contact angles with at least two test liquids [26] [22].
Spectrophotometric Method: A novel approach determines bacterial SFE by analyzing colloidal stability through spectrophotometric measurements, avoiding technical challenges associated with contact angle measurements on bacterial lawns [19].
Pendant Drop Method: Used for determining liquid surface tension when measuring solid SFE through contact angle approaches [26].
Table 3: Essential Research Reagents and Equipment for SFE and Adhesion Studies
| Category | Specific Items | Research Function |
|---|---|---|
| Test Liquids | Water, diiodomethane, ethylene glycol, glycerol | Contact angle measurement with varying polarity |
| Culture Media | Luria-Bertani (LB) broth, phosphate buffer solutions (PBS) | Bacterial cultivation and suspension preparation |
| Model Bacteria | E. coli (Gram-negative), S. aureus (Gram-positive) | Representative bacterial strains for adhesion studies |
| Surface Treatment | O2 plasma, C4F8 plasma, silane coupling agents | Surface energy modification |
| Analysis Equipment | Contact angle goniometer, spectrophotometer, scanning electron microscope | Quantifying wettability, bacterial concentration, and visualization |
Quantifying Bacterial Adhesion
Standardized methodologies for bacterial adhesion assessment include:
Adhesion Assays: Bacterial suspensions are incubated with material samples, followed by rinsing and quantification of adhered cells through colony-forming unit (CFU) counting, spectrophotometric methods, or microscopy [19] [21].
Advanced Imaging: Scanning electron microscopy (SEM) and confocal laser scanning microscopy provide visual confirmation of adhesion patterns and biofilm formation [21] [23].
Molecular Analysis: DNA sequencing of adhered microbial communities reveals material-specific differences in bacterial abundance and diversity [20].
Research Implications and Material Design Strategies
The relationship between SFE and bacterial adhesion provides a thermodynamic foundation for designing next-generation dental materials with controlled biological responses. Surface engineering approaches can be strategically employed to either minimize bacterial adhesion on critical surfaces or enhance tissue integration when needed [23] [25]. The conflicting requirements for different regions of dental implants highlight the need for zone-specific surface engineering - where transmucosal areas benefit from low-SFE, smooth surfaces to resist bacterial adhesion, while bone-integration regions may require higher-SFE, roughened surfaces to promote osteogenesis [23]. Future material development should focus on intelligent dental material surfaces that can dynamically adjust their properties in response to the biological environment, potentially through stimuli-responsive polymers or nanoscale surface modifications [20]. Additionally, standardized protocols for SFE measurement and bacterial adhesion assessment are crucial for generating comparable data across research studies and accelerating the development of advanced dental materials with optimized surface properties.
Influence of Surface Roughness Parameters (Sa, Sds, Sdr) on Cellular Response
The biomaterial-cell interface is a critical determinant of the success of medical implants and tissue-engineered constructs. Surface topography, quantified through specific three-dimensional roughness parameters, directly influences cellular adhesion, proliferation, and differentiation—processes fundamental to osseointegration and tissue regeneration. Among the most influential parameters are Sa (arithmetic mean height), which describes the average roughness; Sds (density of summits), which quantifies peak frequency; and Sdr (developed interfacial ratio), which expresses the surface complexity beyond a flat plane [27]. Understanding how these parameters direct cellular response provides a scientific basis for engineering implant surfaces with predictable biological outcomes, a crucial consideration in dental and orthopedic biomaterials research [28] [27].
Key Surface Roughness Parameters and Their Biological Significance
The quantitative characterization of surface topography moves beyond simple two-dimensional profiling to provide a three-dimensional understanding of the surface landscape.
- Sa (Arithmetic Mean Height): This parameter represents the arithmetic mean of the absolute height deviations from a mean plane. It provides a general overview of surface roughness. Surfaces with a moderately rough Sa (approximately 1-2 µm) have been shown to improve bone-implant contact compared to smoother or rougher surfaces [27].
- Sds (Density of Summits): This parameter describes the number of peaks per unit area. A higher Sds indicates a denser distribution of surface features, which can influence the number of potential anchoring points for cell adhesion. Research on dental implants has reported Sds values ranging from approximately 155,000 to 175,000 peaks per mm² [27].
- Sdr (Developed Interfacial Ratio): This hybrid parameter represents the percentage of additional surface area contributed by the texture compared to a perfectly flat reference plane. It is a measure of surface complexity and wettability. A higher Sdr indicates a more complex surface that can enhance protein adsorption and mechanical interlocking with cells [27].
Quantitative Comparison of Roughness Parameters and Cellular Outcomes
The following tables summarize experimental data from key studies, linking specific roughness parameters to observed cellular and clinical responses.
Table 1: Roughness Parameters of Commercially Available Dental Implants
| Implant Brand | Sa (µm) | Sds (peaks/mm²) | Sdr (%) | Surface Classification |
|---|---|---|---|---|
| Oneplant | 1.01 ± 0.20 | 168,931 ± 21,201 | 73.20 ± 37.28 | Moderately Rough |
| Titaniumfix | 0.90 ± 0.23 | 164,673 ± 10,265 | 57.08 ± 27.10 | Minimally Rough |
| Neodent | 0.67 ± 0.16 | 155,725 ± 15,727 | 52.33 ± 48.12 | Minimally Rough |
| Biomet 3i | 0.53 ± 0.12 | 174,877 ± 18,432 | 45.15 ± 22.45 | Minimally Rough |
Source: Adapted from [27]. Implants were treated with blasting and acid etching.
Table 2: Influence of Hydroxyapatite (HA) Surface Roughness on Human Bone Marrow Cell Response
| Surface Roughness (Sa) | Cell Adhesion | Cell Proliferation | Alkaline Phosphatase (ALP) Activity | Detachment Strength |
|---|---|---|---|---|
| Smooth (Polystyrene) | Baseline | High | Low | Low |
| 0.7 µm (Smooth HA) | Higher than baseline | High | Low | Medium |
| 2.1 µm (Rough HA) | Significantly Higher | Lower than smooth surfaces | Significantly Higher | Significantly Higher |
Source: Summarized from [28]. Rougher HA surfaces promoted osteoblastic differentiation but suppressed proliferation compared to smoother surfaces.
Table 3: Combined Effect of Surface Energy and Roughness on Cell Adhesion and Growth
| Surface Type | Roughness Ratio (r) | Surface Energy (mJ m⁻²) | Cell Adhesion & Growth Efficiency |
|---|---|---|---|
| Superhydrophobic | ~3 | ~21 | Suppressed |
| Low Energy / Intermediate Roughness | ~2 | ~70 | Most Favorable |
| Superhydrophilic | ~1.05 | ~100 | Suppressed |
Source: Derived from [29]. The study on PDMS surfaces with HeLa and MDA-MB-231 cells revealed that moderate surface energy and intermediate roughness ratio constitute the optimal conditions for cell culture.
Experimental Protocols for Investigating Roughness and Cellular Response
Protocol: Surface Modification and Characterization of Titanium Implants
This methodology is used to create and analyze surfaces with controlled roughness [27].
- Surface Treatment: Subject machined titanium implants to a combination of blasting (using TiO₂ or Al₂O₃ particles) followed by acid etching (e.g., with HCl+H₂SO₄ or HNO₃+HF solutions). Variations in particle size, acid concentration, temperature, and exposure time generate different topographies.
- Surface Characterization:
- Quantitative Analysis: Use a light interferometer to measure the topographical parameters (Sa, Sds, Sdr) on the tops, valleys, and flanks of the implant threads. Analyze a minimum of three samples per implant with three measurements each.
- Qualitative Analysis: Perform Scanning Electron Microscopy (SEM) at various magnifications (e.g., 350X to 5,000X) to visually assess the surface morphology and the uniformity of the treatment.
- Statistical Analysis: Analyze data using one-way ANOVA with post-hoc tests to determine significant differences between implant surfaces.
Protocol: Evaluating Osteoblast Response to Hydroxyapatite Roughness
This in vitro protocol assesses the biological response to varied surface textures [28].
- Substrate Preparation: Fabricate hydroxyapatite (HA) discs with different surface roughness (e.g., Sa of 0.7 µm and 2.1 µm) through sintering and grinding. Use polished polystyrene as a smooth control.
- Cell Culture: Seed human bone marrow cells onto the HA discs and maintain under standard culture conditions (e.g., DMEM with 10% FCS, 37°C, 5% CO₂).
- Biological Assays:
- Cell Adhesion: Quantify the number of cells attached to the surface after a predetermined time (e.g., 4 hours).
- Cell Proliferation: Measure cell viability and growth over time using assays like MTT at days 3, 7, and 14.
- Cell Differentiation: Assess alkaline phosphatase (ALP) activity, a key early marker of osteoblastic differentiation.
- Detachment Strength: Use a fluid shear stress apparatus to determine the force required to detach cells from the surface.
The logical workflow for investigating surface roughness and cellular response, from surface preparation to data analysis, is summarized in the following diagram:
Signaling Pathways and Cellular Mechanosensing
The cellular perception of surface topography is a complex process involving mechanotransduction pathways. While the exact signaling cascades are beyond the scope of the cited morphological studies, the overarching mechanism involves the integrin-mediated adhesion of cells to the surface. Rougher, more complex surfaces (with higher Sdr) influence cell shape and spreading, which in turn affects cytoskeletal tension and nuclear signaling. This can lead to the activation of transcription factors like YAP/TAZ, which regulate genes responsible for cell proliferation and differentiation, ultimately dictating cell fate [30] [29]. The following diagram illustrates this general pathway.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagent Solutions for Surface Roughness and Cell Response Studies
| Item | Function in Research | Example Application |
|---|---|---|
| Polydimethylsiloxane (PDMS) | A versatile polymer used to fabricate substrates with tunable surface energy and nano-scale roughness for 2D cell culture studies. | Studying the combined effect of surface energy and roughness on cancer cell lines (HeLa, MDA-MB-231) [29]. |
| Hydroxyapatite (HA) Discs | A bioactive calcium phosphate ceramic used as a model substrate for bone regeneration studies; its roughness can be controlled by sintering and grinding. | Evaluating adhesion, proliferation, and differentiation of human bone marrow cells [28]. |
| Titanium Implants (Blasted & Etched) | Standard substrates for dental and orthopedic research, providing moderately rough surfaces for osseointegration studies. | Comparative analysis of commercial implant surfaces using interferometry and SEM [27]. |
| Dulbecco's Modified Eagle Medium (DMEM) | A standard cell culture medium supplemented with fetal bovine serum (FBS) and antibiotics to support cell growth in vitro. | Maintenance and culture of various cell lines, including osteoblasts and fibroblasts, on test substrates [28] [29]. |
| MTT Assay Kit | A colorimetric assay that measures the reduction of a tetrazolium salt by metabolically active cells, used to quantify cell viability and proliferation. | Assessing proliferation of osteoblast-like cells or fibroblasts on different surfaces over time [31] [28]. |
| Scanning Electron Microscope (SEM) | An essential instrument for high-resolution imaging of surface topography and cell morphology attached to the substrate. | Visualizing the micro- and nano-structure of treated surfaces and the morphology of adhered cells [32] [31] [28]. |
| Light/White Light Interferometer | A non-contact optical profiler used for the precise 3D quantitative measurement of surface roughness parameters (Sa, Sds, Sdr). | Characterizing the micrometric roughness of screw-shaped dental implants [27]. |
Advanced Analytical Techniques for Surface Characterization
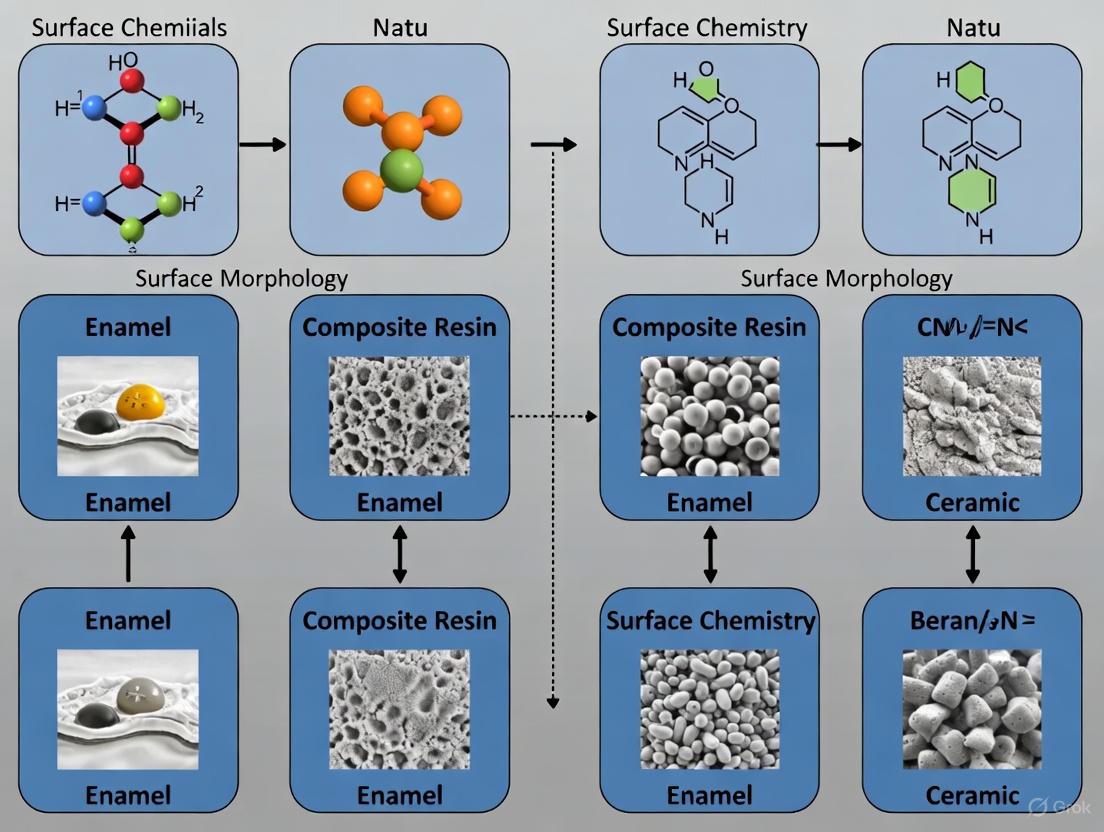
The development and performance of modern dental materials are deeply influenced by their surface properties and chemical composition. Spectroscopic methods have emerged as powerful tools for characterizing these properties, providing insights that are crucial for ensuring the biocompatibility, durability, and clinical success of dental biomaterials. As the field of dental materials science advances toward more complex and miniaturized components, the need for precise analytical techniques becomes increasingly important. Spectroscopic analysis enables researchers to probe materials at the micro- and nanoscale, revealing critical information about molecular structures, elemental composition, and surface interactions that directly impact clinical performance [33]. This guide provides a comprehensive comparison of three fundamental spectroscopic techniques—Fourier-Transform Infrared (FT-IR) spectroscopy, Raman spectroscopy, and X-ray spectroscopy—focusing on their principles, applications, and experimental protocols in dental materials research.
The significance of these techniques lies in their ability to provide non-destructive analysis of a wide range of dental materials, including ceramics, calcium silicate cements, titanium alloys, and resin-based composites [34]. For instance, modern resin-based composites for dental restoration are complex multi-compound materials, and understanding their molecular composition and morphology is essential for material improvement [33]. Similarly, the biocompatibility and osseointegration potential of dental implants are significantly influenced by their surface chemistry and topography, properties that can be thoroughly characterized using spectroscopic methods [35] [36].
Fundamental Principles of Spectroscopy
Spectroscopy encompasses a range of analytical techniques based on the interaction between electromagnetic radiation and matter. When materials are exposed to specific wavelengths of light, they absorb, emit, or scatter energy in characteristic ways that provide information about their chemical composition and molecular structure. The major advantages of spectroscopic techniques include their non-destructive nature and the requirement for only small sample amounts [34].
The electromagnetic spectrum spans a wide range of wavelengths and energies, with different spectroscopic techniques utilizing specific regions to probe various material properties. The table below summarizes the primary regions relevant to the spectroscopic methods discussed in this guide:
Table 1: Regions of the Electromagnetic Spectrum Relevant to Material Characterization
| Region of Electromagnetic Spectrum | Wavelength Range λ (m) | Spectroscopic Technique |
|---|---|---|
| Infrared | 10⁻³–10⁻⁶ | Infrared spectroscopy, Raman spectroscopy |
| Ultraviolet and visible | 10⁻⁶–10⁻⁸ | UV-Visible spectroscopy |
| X-ray | 10⁻⁹–10⁻¹² | X-ray diffraction, X-ray fluorescence |
Based on the radiation measurement process, spectroscopy is categorized into three main types: absorption spectra (transitions from lower to higher energy levels), emission spectra (transitions from higher to lower energy levels), and Raman spectra (frequency changes in scattered radiation) [34]. Each type provides complementary information about the material being analyzed.
Comparative Analysis of Spectroscopic Techniques
FT-IR Spectroscopy
FT-IR spectroscopy operates on the principle that molecules absorb specific frequencies of infrared light that correspond to the natural frequencies of their chemical bonds. The absorption of infrared radiation causes molecular bonds to vibrate through stretching and bending motions, producing a spectrum that serves as a molecular "fingerprint" [34]. Modern FT-IR instruments use an interferometer and Fourier transform mathematics to simultaneously collect spectral data across the entire infrared range, resulting in faster acquisition and enhanced signal-to-noise ratio compared to traditional dispersive instruments [34].
A significant advancement in FT-IR technology is the Attenuated Total Reflectance (ATR) accessory, which allows direct analysis of solid and liquid samples without extensive preparation. ATR-FTIR works by measuring the changes that occur in a totally internally reflected infrared beam when it comes into contact with a sample [37]. This technique is particularly valuable for analyzing dental materials that may be difficult to prepare using traditional transmission methods.
Table 2: Key Applications of FT-IR Spectroscopy in Dental Research
| Application Area | Specific Use Cases | References |
|---|---|---|
| Dental Composite Analysis | Chemical composition assessment, interface study between composite and adhesive, porosity determination | [33] |
| Biopharmaceutical Testing | In-line monitoring of protein formulations, stability assessment under various conditions | [38] |
| Implant Surface Characterization | Analysis of surface chemistry and coating composition | [34] |
| Mineral Content Assessment | Evaluation of enamel mineral changes after bleaching treatments | [39] |
Raman Spectroscopy
Raman spectroscopy complements FT-IR by providing information about molecular vibrations based on the inelastic scattering of light. When monochromatic light interacts with a sample, most photons are elastically scattered (Rayleigh scattering), but a small fraction undergoes energy shifts corresponding to vibrational transitions in the molecules (Raman scattering) [34]. This technique is particularly sensitive to symmetrical covalent bonds and can provide detailed information about crystal structure and polymorphism in dental materials.
The Raman effect occurs when incident photons interact with molecular dipoles, resulting in energy transfer that produces shifted frequencies in the scattered light. These frequency shifts correspond to specific vibrational modes within the molecule, creating a spectrum that can be used for material identification and characterization [40]. Raman spectroscopy excels in analyzing aqueous systems and is minimally affected by water interference, making it suitable for studying biological interfaces and hydrated dental materials.
Table 3: Characteristic Raman Spectral Ranges for Dental Materials
| Material Class | Characteristic Bands (cm⁻¹) | Molecular Information |
|---|---|---|
| Calcium Silicate Cements | 800-1200 | Silicate and carbonate phases |
| Hydroxyapatite | 430-450, 580-620, 950-970 | Phosphate vibrations |
| Titanium Oxides | 140-150, 440-450, 610-620 | Crystalline phase identification |
| Dental Polymers | 1600-1700, 2800-3000 | C=C, C-H vibrations |
X-ray Spectroscopy
X-ray spectroscopy encompasses several techniques that utilize the interaction between X-rays and matter to determine elemental composition and crystal structure. The two primary methods used in dental materials research are X-ray Diffraction (XRD) and X-ray Fluorescence (XRF). XRD measures the diffraction patterns produced when X-rays interact with crystalline materials, providing information about crystal structure, phase composition, and structural properties [34]. XRF analyzes the characteristic secondary X-rays emitted from a material when excited by a primary X-ray source, enabling qualitative and quantitative elemental analysis.
These techniques are particularly valuable for characterizing the inorganic components of dental materials, such as the crystalline phases in ceramics, the oxide layers on metallic implants, and the mineral content of tooth structures. X-ray techniques can detect elements from sodium to uranium, with detection limits ranging from parts per million to percent levels, depending on the element and the sample matrix [34].
Experimental Protocols and Methodologies
Sample Preparation Guidelines
Proper sample preparation is critical for obtaining reliable spectroscopic data. For FT-IR analysis of dental materials using ATR accessories, samples should have smooth, flat surfaces to ensure good contact with the crystal. Powdered samples can be analyzed directly with consistent pressure application. For Raman spectroscopy, samples should be free of fluorescent contaminants that can interfere with signal detection. Dental implant surfaces may require minimal preparation beyond cleaning with solvents to remove organic residues [36].
For X-ray spectroscopy, sample preparation varies significantly based on the technique. XRD typically requires flat, homogeneous samples, while XRF can accommodate various sample forms including powders, solids, and liquids. Metallic dental implant samples are often mounted in resin and polished to create a flat surface for analysis [35].
FT-IR Experimental Protocol
A standard protocol for ATR-FTIR analysis of dental materials involves the following steps:
- Instrument Calibration: Perform background scans without the sample present using the same experimental parameters.
- Sample Placement: Position the dental material sample to ensure complete contact with the ATR crystal.
- Data Acquisition: Collect spectra typically over the range of 4000-400 cm⁻¹ with a resolution of 4 cm⁻¹, accumulating 32-64 scans to improve signal-to-noise ratio.
- Data Processing: Apply atmospheric suppression (for CO₂ and H₂O vapor), baseline correction, and normalization algorithms as needed.
For microspectroscopic imaging, as demonstrated in dental filling analysis, an FT-IR microscope or quantum cascade laser-based IR microscope can be used for microscale analysis, while scattering-type scanning near-field optical microscopy (s-SNOM) enables nanoscale resolution [33].
Raman Experimental Protocol
A typical Raman spectroscopy protocol for dental materials includes:
- Wavelength Selection: Choose an appropriate excitation laser (e.g., 532 nm or 785 nm) to minimize fluorescence while maintaining sufficient signal intensity.
- Sample Alignment: Focus the laser on the area of interest using the microscope objective.
- Parameter Optimization: Adjust laser power, acquisition time, and number of accumulations to maximize signal while preventing sample damage.
- Spectral Collection: Acquire spectra with appropriate grating to achieve the desired spectral resolution.
For dental implant surface analysis, a 532 nm He-Ne laser with a scanning range of 200-2000 cm⁻¹ and constant power output of 10 mW has been effectively utilized [35].
X-ray Spectroscopy Experimental Protocol
Standard protocols for X-ray analysis of dental materials:
- XRD Analysis:
- Mount the sample in the X-ray diffractometer
- Set the appropriate voltage and current for the X-ray tube (typically 40 kV, 40 mA for Cu Kα radiation)
- Scan through a 2θ range of 5-80° with a step size of 0.02° and counting time of 1-2 seconds per step
- XRF Analysis:
- Place the sample in the spectrometer chamber
- Select appropriate collimators and filters for the elements of interest
- Acquire spectra using optimized conditions for the specific sample matrix
Data Interpretation and Analysis
Interpreting spectroscopic data from dental materials requires understanding characteristic spectral features and their correlation with material properties. In FT-IR spectra, the position, intensity, and shape of absorption bands provide information about specific functional groups and molecular environments. For example, the carbonate band at 870-880 cm⁻¹ can indicate the presence of carbonated hydroxyapatite in bone grafts or dental restoratives [39].
Raman spectra of dental materials often show distinct bands that can be assigned to specific molecular vibrations. The phosphate ν1 band at approximately 960 cm⁻¹ is characteristic of hydroxyapatite in natural teeth and bioactive coatings, while the spectral region between 800-1200 cm⁻¹ can reveal silicate structures in ceramic materials [40].
XRD patterns are analyzed by identifying characteristic peak positions and intensities, which are compared to reference patterns in databases such as the International Centre for Diffraction Data (ICDD). For titanium dental implants, XRD can identify the crystalline phases of surface oxides (e.g., anatase vs. rutile TiO₂), which influence biocompatibility and corrosion resistance [35].
Advanced Applications in Dental Materials Research
Dental Composite Characterization
Advanced IR spectroscopic imaging has been applied to characterize the complex multi-compound structure of modern resin-based dental composites. Using FT-IR microscopy and quantum cascade laser-based IR microscopy at the microscale, combined with scattering-type scanning near-field optical microscopy (s-SNOM) at the nanoscale, researchers can assess the distribution of glass cluster structures within their organically modified ceramic (ORMOCER) matrix and evaluate the interface between composite and adhesive materials [33]. This level of analysis provides insights into material homogeneity, interface quality, and potential failure mechanisms in dental restorations.
Implant Surface Modification Analysis
Spectroscopic techniques play a crucial role in evaluating surface modifications designed to enhance the biocompatibility and osseointegration of dental implants. Studies on Ti-6Al-4V alloy surfaces modified through techniques such as sandblasting, shot peening, and electrochemical corrosion have utilized Raman spectroscopy to analyze surface chemistry changes following treatment [35]. Similarly, EDX analysis has revealed how photofunctionalization through UV treatment alters the elemental composition of zirconia implants, reducing surface carbon content from 45.41% to 34.34% while increasing oxygen concentration from 29.09% to 42.8%, thereby enhancing surface hydrophilicity [36].
Mineral Content Assessment in Enamel
Spectroscopic methods enable quantitative assessment of mineral changes in dental enamel following various treatments. Research on bleaching agents has employed colorimetric spectrophotometry to quantify calcium and phosphorus levels extracted from enamel substrates through microbiopsy techniques [39]. These studies have demonstrated that all bleaching protocols induce varying degrees of mineral loss, with traditional McInnes solution causing the most significant effects, while modified McInnes solution shows relatively milder alterations. Post-bleaching remineralization in artificial saliva shows beneficial recovery effects that can be monitored spectroscopically.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Essential Research Reagents and Materials for Spectroscopic Analysis of Dental Materials
| Reagent/Material | Function/Application | Example Uses |
|---|---|---|
| ATR Crystals (diamond, ZnSe) | Enables sample contact for FT-IR measurement | Analysis of solid dental materials without extensive preparation |
| Reference Materials | Instrument calibration and method validation | Quality control protocols for spectroscopic analysis |
| Ultrapure Acids | Sample preparation and etching | Enamel microbiopsy for mineral content analysis [39] |
| Inert Atmosphere Equipment | Protection of air-sensitive samples | Analysis of reactive dental materials [37] |
| Polishing Supplies | Sample surface preparation | Creating flat, uniform surfaces for spectroscopic analysis |
| Calibration Standards | Quantitative analysis verification | Elemental quantification in XRF and EDX analysis |
FT-IR, Raman, and X-ray spectroscopy provide complementary approaches for characterizing the chemical composition and surface properties of dental materials. FT-IR excels in identifying organic functional groups and is advancing through microspectroscopic imaging techniques. Raman spectroscopy offers detailed molecular structure information, particularly for inorganic phases and crystalline structures. X-ray techniques provide essential data on elemental composition and crystal structure. Together, these methods form a comprehensive analytical toolkit for developing and evaluating next-generation dental materials with enhanced performance and biocompatibility.
The continuing evolution of these techniques, including the integration of machine learning for data analysis and the development of hybrid instrumentation, promises to further expand their capabilities in dental materials research. As spectroscopic technologies advance toward higher resolution and faster acquisition times, they will enable more detailed characterization of complex dental material systems and their interactions with the oral environment.
Scanning Electron Microscopy (SEM) has become an indispensable tool in dental materials research, providing critical insights into the micro- and nano-scale morphology of dental biomaterials. When coupled with Energy Dispersive X-ray Spectroscopy (EDX), SEM enables comprehensive characterization of both surface topography and chemical composition, offering researchers a powerful method for evaluating material performance and biological interactions. The principal advantage of SEM lies in its exceptional depth of field and high resolution, which allows for the detailed visualization of surface features that are beyond the capabilities of conventional optical microscopy [41]. This capability is particularly valuable in dentistry, where surface characteristics directly influence biological responses such as osseointegration around dental implants, adhesive bonding in restorative procedures, and cellular interactions with pulp-capping materials.
The application of SEM in dental research spans multiple domains, from evaluating the effects of root canal instrumentation on dentinal structure to assessing the surface properties of emerging biomaterials. For instance, SEM analysis has revealed how different endodontic file systems create varying patterns of smear layer formation and dentinal microcracks during root canal preparation [42]. Similarly, SEM-EDX has been instrumental in characterizing the elemental composition of hydraulic calcium silicate cements used in vital pulp therapy, confirming the presence of key elements like calcium, oxygen, and carbon that contribute to their bioactivity [43]. As dental materials continue to evolve toward more complex formulations and nanostructured surfaces, SEM remains at the forefront of qualitative morphology assessment, providing the critical data needed to correlate material structure with clinical performance.
Comparative Performance of Microscopy Techniques
SEM Versus Alternative Microscopy Methods
While SEM represents the gold standard for high-resolution surface characterization in dental materials research, several alternative microscopy techniques offer complementary capabilities. Understanding the relative strengths and limitations of each method is essential for selecting the appropriate analytical tool for specific research questions.
Digital Optical Microscopy (DOM) has emerged as a viable alternative for certain applications, particularly marginal adaptation analysis of dental adhesive interfaces. A 2021 validation study comparing DOM to SEM demonstrated no statistically significant difference between the two methods when measuring noncontinuous margins (NCM) in resin composite restorations [44]. The Bland-Altman analysis revealed a minimal bias of 0.86%, confirming DOM's acceptable agreement with the SEM gold standard. DOM offered significant advantages in analysis time efficiency, requiring almost half the time compared to SEM for complete margin assessment. However, this efficiency comes at the cost of spatial resolution, with SEM maintaining superior capability for nano-morphological assessment [44].
Traditional Optical Microscopy (OM) faced limitations in dental microwear studies due to restricted depth of field and problems with shiny enamel surfaces, leading to its replacement by SEM for most high-precision applications [41]. However, recent technological improvements in OM equipment and software have revitalized its potential for specific applications. A 2020 comparative study found that OM recorded a 17.50% increase in the mean number of striations compared to SEM when analyzing buccal dental microwear, suggesting potentially greater sensitivity for certain feature types [41]. Despite this advantage, SEM maintains superiority for comprehensive surface characterization due to its greater depth of field and ability to resolve nanoscale features critical for understanding material-tissue interactions.
Table 1: Comparison of Microscopy Techniques in Dental Materials Research
| Technique | Best Applications | Resolution Range | Key Advantages | Principal Limitations |
|---|---|---|---|---|
| Scanning Electron Microscopy (SEM) | Surface morphology, fracture analysis, coating integrity | 1 nm to 1 μm | Exceptional depth of field, high resolution, elemental analysis with EDX | Expensive equipment, requires conductive coatings, vacuum environment |
| Digital Optical Microscopy (DOM) | Marginal adaptation, dimensional measurements | 500 nm to 10 μm | Rapid analysis, non-destructive, minimal sample preparation | Limited magnification, reduced resolution compared to SEM |
| Optical Microscopy (OM) | Microwear analysis, initial screening | 1 μm to 100 μm | Accessible, cost-effective, minimal sample preparation | Limited depth of field, lower resolution, reflective surfaces problematic |
Performance Metrics and Analytical Capabilities
The quantitative performance of SEM in dental materials research extends beyond basic imaging to comprehensive surface characterization. When evaluating marginal adaptation of dental adhesives, SEM demonstrated exceptional measurement repeatability with no statistically significant differences between repeated measurements (p = 0.523) [44]. This reliability makes SEM particularly valuable for longitudinal studies assessing material degradation or interface stability under simulated clinical conditions.
For chemical composition analysis, the integration of EDX with SEM provides unique capabilities for elemental mapping and quantitative composition assessment. In the evaluation of titanium dental implants, EDX analysis revealed significant variations in surface composition depending on implant design and manufacturing process. Single-piece compressive implants showed titanium (Ti) as the major component (71.55-89.71 weight%), while spiral implants exhibited iron (Fe) as the predominant element (66.60 weight%) with significant chromium (Cr) content (18.34 weight%) [45]. These compositional differences directly influence corrosion resistance and biocompatibility, highlighting the importance of combined morphological and chemical analysis.
For surface roughness assessment, SEM complements quantitative techniques like optical profilometry by providing visual context for numerical roughness parameters. Studies on additively manufactured Ti-6Al-4V implants demonstrated how SEM imaging revealed partially unfused particles that contributed to increased roughness and decreased corrosion resistance [46]. Subsequent acid etching treatment improved both surface characteristics and corrosion behavior, with changes clearly documented through SEM analysis.
Table 2: SEM-EDX Applications in Dental Biomaterials Characterization
| Material Category | Research Application | Key SEM Findings | EDX Elemental Data |
|---|---|---|---|
| Calcium Silicate Cements [43] | Surface morphology and cytocompatibility | Adequate human dental pulp cell attachment; uniform microstructure | ProRoot MTA: Ca, C, O; Biodentine: Ca, C, O; Bio-C Repair: High Zr, low Ca |
| Titanium Implants [45] | Surface topography and composition | Irregularities (10-20 μm); grinding marks; amorphous structures | Single-piece: Ti (71.55-89.71%); Spiral: Fe (66.60%), Cr (18.34%) |
| Resin Composites [44] | Marginal adaptation | Noncontinuous margins at tooth-restoration interface | Not typically analyzed for marginal adaptation studies |
| Nickel-Titanium Files [42] | Dentinal surface effects | Smear layer removal; dentinal cracks in coronal/apical thirds | Ca, P, O distribution from remnant dentinal products |
Experimental Protocols for SEM Analysis in Dental Research
Sample Preparation and Imaging Protocols
Proper sample preparation is fundamental to obtaining reliable SEM data in dental materials research. The specific protocol varies depending on material properties and research objectives, but several common steps ensure optimal imaging conditions.
For hard tissue studies, such as evaluating dentinal surfaces after root canal instrumentation, researchers typically extract human teeth and subject them to the experimental procedures. Following treatment, samples are sectioned longitudinally to expose the region of interest. The critical preparation step involves sputter coating with a conductive material (typically gold-palladium or carbon) to prevent charging effects under electron bombardment. A representative study on radicular dentine effects after instrumentation employed metallization of the active surface before SEM analysis at magnifications of 750× and 800× to identify dentinal cracks and assess smear layer removal [42].
For biomaterial characterization, including hydraulic cements and titanium implants, sample preparation follows different pathways. In evaluating vital pulp materials like Biodentine, ProRoot MTA, and Bio-C Repair, researchers prepared samples using silicone molds with 2mm depth and 5mm diameter wells, allowing them to set for one week in an incubator at 37°C and 95% humidity to simulate oral conditions [43]. These samples were then visualized under various magnifications (500× to 50,000×) at accelerating voltages of 5-10 kV. For non-conductive materials, similar conductive coating procedures are essential to obtain clear images without charging artifacts.
The replica technique provides a valuable approach for analyzing surface features without destroying original samples. In marginal adaptation studies, researchers create polyvinylsiloxane impressions of restored teeth, followed by epoxy resin replicas that are gold-coated before SEM examination [44]. This method preserves the original specimens for additional testing or clinical use while providing accurate representations of surface topography.
SEM-EDX Analytical Methodology
Combining SEM with Energy Dispersive X-ray Spectroscopy (EDX) enables comprehensive morphological and chemical characterization. The standard EDX protocol involves scanning the electron beam across the region of interest while detecting characteristic X-rays emitted from the sample surface.
In dental implant characterization, EDX analysis is typically performed at multiple locations to account for surface heterogeneity. A recent study on commercially available titanium implants conducted EDX analysis on both plain areas and top land areas, with three measurements per area to ensure representative sampling [45]. The analysis utilized an acceleration voltage of 20 kV with a beam current of 200 pA, parameters optimized for exciting characteristic X-rays from elements present in titanium alloys. The resulting data provided weight percentages of detected elements, revealing significant differences between implant types that would remain undetected through morphological analysis alone.
For hydraulic cement characterization, researchers employed similar EDX methodologies with particular attention to calcium and silicon distribution, key elements contributing to bioactivity. Studies on ProRoot MTA, Biodentine, and Bio-C Repair revealed distinct elemental profiles despite similar clinical applications, with Bio-C Repair showing unexpectedly low calcium concentration and high zirconium content [43]. These compositional differences potentially influence biological responses including cell viability and attachment, demonstrating the importance of combined morphological and chemical analysis.
Research Reagent Solutions for SEM Studies
Successful SEM analysis in dental materials research requires specific reagents and materials at various stages, from sample preparation to final imaging. The following table details essential research reagent solutions and their functions in SEM-based studies.
Table 3: Essential Research Reagents and Materials for SEM Dental Studies
| Reagent/Material | Specific Function | Application Example | Technical Notes |
|---|---|---|---|
| Gold-Palladium Coating | Creates conductive surface to prevent charging | Sputter coating of non-conductive samples | 20-30 nm thickness typically used for high-resolution imaging |
| Epoxy Resin (EpoFix) | Creates high-resolution replicas from impressions | Marginal adaptation studies [44] | Slow-curing transparent resin preserves surface detail |
| Polyvinylsiloxane Impression Material | Captures negative imprint of surface topography | Replica technique for marginal analysis [44] | High precision materials (e.g., President light body) recommended |
| Hydrofluoric Acid Etchant | Surface treatment of titanium alloys | Improving corrosion resistance of SLM Ti-6Al-4V [46] | 20-minute treatment enhanced surface properties |
| Glutaraldehyde (4%) | Fixation of biological specimens | Cell attachment studies on material surfaces [43] | Preserves cellular structures during dehydration process |
| Ethanol Series (30%-90%) | Gradual dehydration of biological samples | Preparing cell-seeded specimens for SEM [43] | Prevents tissue distortion that occurs with rapid drying |
Advanced Applications and Current Research Frontiers
Emerging Applications in Dental Materials Development
SEM analysis continues to drive innovation across multiple domains of dental materials science through its ability to characterize novel material structures and surface modifications. In the field of additively manufactured implants, SEM has revealed critical differences in surface morphology between traditionally machined and selective laser melted (SLM) Ti-6Al-4V samples. Researchers discovered that SLM-produced implants exhibited partially unfused particles and increased surface roughness that compromised corrosion resistance until optimized post-processing treatments were applied [46]. These findings directly inform manufacturing protocols for improved clinical outcomes.
In endodontic research, SEM analysis has provided crucial insights into tissue-instrument interactions that directly impact clinical practice. A comprehensive 2025 study compared five different endodontic instrumentation systems and revealed significant differences in dentinal crack formation depending on the file system employed [42]. While Kerr files and the Orodeka system produced minimal cracks, ProTaper Flydent instruments generated multiple cracks in the coronal third, and when combined with an ultrasonic scaler, produced cracks in both coronal and apical thirds. These morphological findings directly correlate with clinical concerns about root fracture susceptibility and inform instrument selection criteria.
The biological evaluation of dental materials represents another advancing frontier for SEM application. Studies examining human gingival fibroblast adhesion to zirconia surfaces with different treatments demonstrated that polished and glazed surfaces promoted superior cell spreading compared to untreated or sandblasted surfaces [47]. Similarly, SEM analysis of human dental pulp cells cultured on vital pulp materials confirmed adequate cell attachment and revealed cytoskeletal organization when exposed to material eluates [43]. These biological interactions at the material-tissue interface fundamentally influence clinical success and can be optimally characterized through SEM.
Methodological Innovations and Future Directions
The evolving methodology of SEM application in dental research includes several promising innovations that enhance analytical capabilities. The integration of elemental mapping with EDX provides spatial distribution of elements critical to bioactivity, such as calcium and phosphorus in hydraulic cements. Advanced imaging techniques, including environmental SEM (ESEM) that eliminates the need for conductive coatings, are expanding application possibilities for moisture-containing biological samples.
The correlative microscopy approach, combining SEM with other techniques like confocal microscopy, offers comprehensive datasets that link surface morphology with subsurface structures and compositional data. As noted in dental microwear studies, "the problems that initially led to the replacement of OM for dental microwear studies are no longer the obstacle they once were" due to technological improvements [41], suggesting potential for multimodal approaches that leverage the respective strengths of different microscopy platforms.
Future applications will likely focus on dynamic processes such as material degradation, protein adsorption, and early biofilm formation, utilizing specialized SEM stages that allow for time-lapse observations under controlled conditions. These advances will further solidify SEM's position as an essential tool for qualifying and quantifying the micro- and nano-morphological features that determine the clinical performance of dental materials.
In dental materials research, the surface morphology and chemical composition of implants and restorative materials directly influence critical biological processes such as osseointegration, cellular response, and long-term stability [45] [43]. Quantitative topographical analysis provides the methodological foundation for characterizing these surface properties with high precision. Among the available techniques, White Light Interferometry (WLI) and Laser Profilometry (specifically, Laser Scanning Confocal Microscopy - LSCM) represent two advanced optical methods for non-contact surface measurement [48]. This guide objectively compares the performance, capabilities, and limitations of these two technologies within the context of dental materials research, providing researchers with experimental data and protocols to inform their methodological selections.
Technical Principles and Measurement Methodologies
Understanding the fundamental operational principles of WLI and LSCM is essential for selecting the appropriate technique for specific research scenarios in dental material characterization.
White Light Interferometry (WLI)
Fundamental Principle: WLI operates on the principle of interference. Light from a white-light source is split, directed simultaneously toward a reference surface and the sample surface, and then recombined [49] [48]. Where the sample is in focus, the recombined light creates a pattern of interference fringes. The system vertically scans the objective, and the precise position of maximum fringe contrast for each pixel is determined to be the surface height at that location [48]. This process generates a detailed three-dimensional surface map.
Key Metrology Advantage: WLI was developed specifically for industrial metrology applications, which is reflected in its design and performance characteristics [48].
Laser Scanning Confocal Microscopy (LSCM)
Fundamental Principle: Confocal microscopy, originally developed for biological imaging, uses a focused laser spot scanned across the sample (raster scanning in X and Y) [48]. A small pinhole aperture in front of the detector eliminates out-of-focus light. To build a 3D profile, the sample or objective is advanced in discrete steps along the Z-axis, and the intensity at each point is recorded at every vertical position. The height for each pixel is found by detecting the Z-position of the peak intensity or calculating the center of mass of the intensity distribution [48].
Inherent Limitation: Because each point on the surface must be scanned in both the X-Y plane and the Z-axis, data acquisition over large fields of view can be time-consuming, though spinning-disk confocal systems can mitigate this by imaging multiple points simultaneously [48].
Experimental Workflow for Surface Topography Measurement
The following diagram illustrates the generalized workflow for conducting surface topographical analysis using these non-contact methods, from sample preparation to data interpretation.
Performance Comparison and Experimental Data
The selection between WLI and LSCM involves trade-offs between speed, resolution, and suitability for different surface types. The following data, drawn from comparative studies, quantifies these performance characteristics.
Quantitative Performance Metrics
Table 1: Direct Performance Comparison between WLI and Confocal Microscopy [48]
| Performance Characteristic | White Light Interferometry (WLI) | Laser Scanning Confocal Microscopy (LSCM) |
|---|---|---|
| Vertical Resolution | Sub-nanometer at all magnifications | Degrades significantly with lower magnification objectives (e.g., 5x, 10x) |
| Vertical Scan Speed | Up to 100 µm/sec | Approximately 0.3 Megapixels/sec |
| Measurement Principle | Phase-shifting interferometry; measures fringe contrast | Axial response; measures intensity peak |
| Ideal Application | High-speed, high-accuracy areal surface metrology | High-resolution imaging, can be less ideal for metrology |
Measurement Accuracy and Algorithm Performance
The accuracy of WLI is further influenced by the algorithm used to reconstruct surface topography from interferograms. A comparative analysis of common envelope detection algorithms reveals critical performance differences [49].
Table 2: Performance of WLI Surface Reconstruction Algorithms [49]
| Algorithm | Measurement Speed | Noise Immunity | Measurement Accuracy | Best Use Case |
|---|---|---|---|---|
| Centroid Method | High (computationally simple) | Low | Low to Moderate (acceptable only at low noise levels) | Rapid screening in controlled environments |
| Fast Fourier Transform (FFT) | Low (time-consuming) | High | High | Applications demanding high accuracy, even with noisy data |
| Hilbert Transform (HT) | Moderate to High | High | High (similar to FFT) | A balanced choice for speed and accuracy |
Experimental studies measuring step height standards have confirmed that while the centroid method is fastest, the FFT and HT methods provide superior noise immunity and measurement accuracy, with the HT method offering a favorable compromise between speed and precision [49].
Applications in Dental Materials Research
The comparative performance of WLI and LSCM has direct implications for their effectiveness in specific dental research applications.
Surface Morphology of Dental Implants
The surface topography of titanium implants is a critical factor for osseointegration [45]. Scanning Electron Microscopy (SEM) is a gold standard for visualizing surface ultrastructure, revealing features like grinding marks, surface irregularities, and amorphous structures at high magnifications [45] [43]. However, SEM provides primarily qualitative 2D images.
WLI and LSCM complement SEM by providing quantitative 3D areal data. For example:
- WLI excels at measuring the complex micro-roughness (Sa, Sz) of implant surfaces, which often includes steep flanks and undercuts, in a high-speed manner [48].
- LSCM can effectively measure smoother, prepared implant surfaces, though its vertical performance is best with high-magnification objectives, limiting the field of view [48].
Studies on additively manufactured Ti-6Al-4V implants have successfully used optical profilometry to correlate surface roughness parameters with corrosion resistance, demonstrating that lower roughness, achievable through processes like hydrofluoric acid etching, leads to improved corrosion performance [46].
Chemical Composition and Surface Chemistry
While WLI and LSCM are topographical tools, their data is often correlated with chemical composition analysis to provide a comprehensive material characterization. Energy Dispersive X-ray Spectroscopy (EDX), typically coupled with an SEM, is the standard technique for determining elemental composition on material surfaces [45] [43].
For instance, an EDX study of commercial titanium implants revealed that their surfaces are predominantly composed of titanium, but also contained other elements like oxygen, aluminum, iron, and chromium, depending on the implant type and manufacturing process [45]. Similarly, EDX analysis of vital pulp materials (e.g., MTA, Biodentine) confirms the presence of key ions like calcium, which are essential for their bioactivity [43]. The combination of 3D topography from WLI and elemental data from EDX allows researchers to form robust structure-property relationships in novel dental materials.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagents and Materials for Surface Analysis
| Item | Function/Application |
|---|---|
| Titanium Alloy (Ti-6Al-4V) Samples | Representative substrate for dental implant material research [45] [46]. |
| Calcium Silicate-based Cements (e.g., MTA, Biodentine) | Vital pulp materials for studying bioactivity and surface properties [43]. |
| Hydrofluoric Acid (HF) Etching Solution | Surface treatment for titanium to increase corrosion resistance and alter topography [46]. |
| Sodium Chloride (NaCl) Electrolyte (0.9%) | Simulates physiological conditions for corrosion resistance testing [46]. |
| Reference Step Height Standards | Calibrated samples for validating the vertical accuracy of WLI and LSCM systems [49]. |
Integrated Analytical Workflow
A robust analysis of dental materials often requires an integrated approach, combining multiple analytical techniques to correlate surface form with function. The following diagram outlines a logical workflow for a comprehensive study.
This integrated methodology was effectively employed in a study comparing vital pulp materials, where SEM visualized the surface morphology, EDX confirmed the elemental composition, and cell culture assays (using human dental pulp cells) linked these physical characteristics to biological outcomes like cell viability and attachment [43].
In dental materials research, the surface properties of implants and restorative materials directly influence biological interactions such as osseointegration and biofilm formation. Energy-Dispersive X-ray Spectroscopy (EDX) and X-ray Photoelectron Spectroscopy (XPS) are two cornerstone techniques for elemental and chemical state analysis. This guide provides a comparative analysis of EDX and XPS, supporting material selection for specific research goals in dental science.
Technical Comparison: EDX vs. XPS
The following table outlines the fundamental operational differences between EDX and XPS, which dictate their respective applications.
Table 1: Fundamental technical characteristics of EDX and XPS.
| Feature | Energy-Dispersive X-ray Spectroscopy (EDX) | X-ray Photoelectron Spectroscopy (XPS) |
|---|---|---|
| Acronyms | EDS, EDXA | XPS, ESCA (Electron Spectroscopy for Chemical Analysis) |
| Primary Function | Elemental identification and semi-quantitative composition | Elemental identification, chemical state, and empirical formula |
| Underlying Principle | Detects characteristic X-rays emitted from a sample bombarded with electrons [45] [50] | Measures kinetic energy of photoelectons ejected by X-ray irradiation [50] |
| Information Depth | ~1-3 µm; bulk micro-volume analysis [51] [52] | ~1-10 nm; extreme surface sensitivity [50] |
| Spatial Resolution | Lower resolution, suitable for larger areas [50] | High spatial resolution for detailed small-area analysis [50] |
| Chemical State Data | No direct chemical bonding information | Detailed chemical state and oxidation state information [53] |
| Detection Limits | ~0.1 - 1 at.% (1000 - 10,000 ppm) | ~0.1 - 1 at.% |
| Typical Sample Environment | High vacuum | Ultra-high vacuum (UHV) |
| Key Output | Elemental spectrum and weight% (wt.%) | Elemental spectrum and atomic% (at.%), high-resolution peaks for bonding |
Analysis of Experimental Data in Dental Research
The application of EDX and XPS in dental research provides complementary data, as demonstrated by studies on implant surfaces and dental materials.
Table 2: Experimental data from dental materials research using EDX and XPS.
| Research Focus | Material / Sample | Key EDX Findings | Key XPS Findings | Reference |
|---|---|---|---|---|
| Implant Surface | Nanograting patterns on silicon | Predominantly silicon; no significant oxygen or carbon contaminants [54]. | Clear Si spectrum; no metal oxide or organic contamination; valence band offsets quantified [54]. | [54] |
| Commercial Implants | Eight implant models from different manufacturers | Identified Al in 5/8 models (4-11 wt.%); N and Zr in specific models; Ti content ranged from 67-100 wt.% [55]. | Not concurrently applied in this study. | [55] |
| Surface Characterization | TiUnite, OsseoSpeed, Osseotite, SLA implants | Not the primary focus in this comparative study. | TiUnite surface incorporated P (6-10 at.%); others were primarily TiO2 with varying carbon contamination [56]. | [56] |
| Failed Implant Analysis | Retrieved fractured tissue-level implant | Revealed Au (10.99 wt.%) and Ti (14.03 wt.%) in bone-attached middle region; high Ca/P ratio (1.82) indicated calcified tissue [51]. | Not concurrently applied in this study. | [51] |
Experimental Protocols for Dental Materials
EDX Analysis Protocol for Dental Implants
The following protocol is adapted from methodologies used to characterize commercial dental implants [45] [55].
- Sample Preparation: Remove new dental implants from sterile packaging using titanium forceps and nitrile gloves to prevent surface contamination. Do not wash or rinse the samples. Mount implants on a sample holder (stub) using double-sided conductive carbon tape [55].
- Instrumental Setup: Use a Variable Pressure Scanning Electron Microscope (VP-SEM) or standard SEM equipped with an EDX detector. Set the acceleration voltage to 20 kV [45]. The beam current can be set to 200 pA [45], and the working distance should typically be between 9-16 mm [45].
- Data Acquisition: Perform the analysis on multiple random areas of the implant surface (e.g., coronal, middle, apical regions) to ensure representative sampling [45]. Acquire the EDX spectrum.
- Data Analysis: Identify elements present based on their characteristic X-ray peaks. The software typically provides semi-quantitative data as weight percentage (wt.%) for each detected element [55].
XPS Analysis Protocol for Implant Surfaces
This protocol is informed by studies analyzing the surface chemistry of titanium implants and patterned silicon [54] [56].
- Sample Preparation: For intrinsic surface analysis, implants may be used as-received. To remove native oxides and contaminants, a pre-treatment using Reactive Ion Etching (RIE) can be employed. For example, use a gas mixture of SF₆ and O₂ at a pressure of 50 mTorr and power of 100 W for 60 seconds [54]. Transfer samples to the XPS instrument immediately after pre-treatment to minimize air exposure [54].
- Instrumental Setup: Use an XPS system with a monochromatic Al Kα X-ray source (1486.6 eV). Set the analyzer pass energy to 160 eV for wide/survey scans and 20 eV for high-resolution regional scans [56].
- Data Acquisition: Perform analysis under ultra-high vacuum (UHV). Acquire a wide survey scan (e.g., 0-1100 eV binding energy) to identify all elements present. Follow with high-resolution scans for key elements of interest (e.g., Ti 2p, O 1s, C 1s, P 2p, Ca 2p) to determine chemical states [56].
- Data Analysis: Use the wide scan to determine atomic concentrations (at.%) after applying relative sensitivity factors (RSFs). Analyze high-resolution spectra by calibrating to the C 1s peak (adventitious carbon at 284.8 eV) and performing curve-fitting to identify different chemical species (e.g., TiO₂, Ti⁰, CaCO₃, HA) [56].
Decision Workflow and Technical Pathways
The following diagram illustrates the decision-making process for selecting and applying these analytical techniques in a dental research context.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key research reagents and materials used in EDX and XPS analysis of dental materials.
| Item | Function in Analysis | Example Use Case |
|---|---|---|
| Conductive Carbon Tape | Provides stable electrical and physical connection between sample and stub, preventing charging. | Mounting dental implants for SEM/EDX analysis [55]. |
| Reactive Ion Etch (RIE) Gases | Removes surface contaminants and native oxide layers to reveal the intrinsic material surface. | Pre-treatment of silicon nanograting patterns before XPS analysis (e.g., SF₆/O₂ mixture) [54]. |
| Monochromated Al Kα X-ray Source | High-energy photon source for ejecting core-level photoelectrons from the sample surface. | Standard excitation source in XPS for probing surface chemistry of titanium implants [56]. |
| High-Purity Reference Materials | Calibrates instrument response function and binding energy scale for quantitative analysis. | Gold (Au) or copper (Cu) foils for XPS calibration; pure element standards for EDX [53]. |
| Ultra-High Vacuum (UHV) System | Creates a clean environment for electron analysis without interference from gas molecules. | Essential for XPS measurement to ensure long electron mean free paths and avoid surface contamination [56]. |
Surface wettability is a fundamental property governing the interaction between dental materials and their biological environment. Quantified through contact angle (CA) measurements and surface free energy (SFE) calculations, it directly influences critical outcomes such as bacterial adhesion, cell viability, and the durability of adhesive interfaces [57]. The performance of a dental material is not an intrinsic property but is profoundly affected by its surface characteristics, which can be engineered through various treatment methods.
This guide provides a comparative analysis of surface properties across major dental material classes, including zirconia, resin-based composites (RBCs), orthodontic aligners, and acrylic resins. By presenting structured experimental data and detailed protocols, it aims to equip researchers with the knowledge to select appropriate materials and characterization techniques for specific applications, from promoting osseointegration to reducing plaque accumulation.
Comparative Surface Performance of Dental Materials
The following tables consolidate key wettability and surface energy data for diverse dental materials, providing a reference for comparing their theoretical interaction potential with the biological milieu.
Table 1: Contact Angle and Surface Free Energy of Various Dental Materials
| Material Class | Specific Material / Treatment | Contact Angle (°) | Surface Free Energy (mJ/m²) | Key Findings |
|---|---|---|---|---|
| Zirconia [23] | Polished (NT) | Not Specified | Lowest | Poor MG-63 cell attachment and viability. |
| Airborne-particle abrasion (AB) | Not Specified | Increased vs. NT | Improved surface properties. | |
| Glass-ceramic spray deposition (GC) | Significantly reduced | Significantly increased (p<0.05) | Enhanced hydrophilicity and MG-63 cell adhesion. | |
| Orthodontic Aligners [58] | Spark | 70.5 | 60.8 | Lowest metabolic activity for most bacteria; higher polar SFE component (31.9 mJ/m²). |
| Invisalign | 80.6 | 66.7 | Intermediate performance. | |
| Smile | 91.2 | 74.2 | Lowest metabolic activity for S. gordonii and E. faecalis; low polar SFE. | |
| Polymer-Based Materials [57] | Acrylic Resins | 65.5 - 84.7 | High Critical SFE | Zone of "good adhesion" (critical SFE >40 mJ/m²). |
| BIS-GMA Composites | 49.1 - 58.8 | Low Critical SFE | Zone of "poor adhesion" (critical SFE <30 mJ/m²). | |
| PET-G Retainers (as delivered) | 81.9 | Not Specified | Hydrophobic surface. |
Table 2: Surface Roughness and Adhesive Wettability of Resin-Based Composites (RBCs) [59]
| Composite Type | Surface Treatment | Surface Free Energy (mJ/m²) | Contact Angle of Dental Adhesives | Key Findings |
|---|---|---|---|---|
| Nanohybrid (TEC) | Polished | 45.65 - 49.07 | 16.01° - 35.10° | Adhesive parameters, not SFE, primarily govern wettability. |
| Sandblasted | 45.65 - 49.07 (No significant change) | Reduced for solvent-free adhesive | Sandblasting increases micromechanical retention without altering SFE. | |
| Bulk-Fill (TECBF) | Polished | 45.65 - 49.07 | 16.01° - 35.10° | Similar SFE across different RBC types. |
| Sandblasted | 45.65 - 49.07 (No significant change) | Reduced for solvent-free adhesive | Surface tension of the adhesive is critical for penetration. | |
| Bulk-Fill (SDR) | Polished | 45.65 - 49.07 | 16.01° - 35.10° | Similar SFE across different RBC types. |
| Sandblasted | 45.65 - 49.07 (No significant change) | Reduced for solvent-free adhesive |
Experimental Protocols for Wettability Assessment
Standard Contact Angle Measurement Protocol
The sessile drop method is the most prevalent technique for assessing surface wettability [57]. The standard protocol involves:
- Sample Preparation: Materials are fabricated into disk-shaped specimens (e.g., 10-30 mm diameter) and polished to a standardized finish using a sequence of abrasive papers (e.g., #320 to #600 grit) to minimize initial roughness variations [23] [57]. Subsequent surface treatments (e.g., sandblasting, acid-etching, coating) are applied to experimental groups.
- Liquid Selection: Drops (typically 0.5 µL) of standard test liquids with well-known surface tension components are deposited on the sample surface. Commonly used liquids include [57] [60]:
- Distilled water: A polar liquid representing the biological environment.
- Diiodomethane: A non-polar (dispersive) liquid.
- Glycerol or Ethylene glycol: Polar liquids with different surface tension properties.
- Measurement: An optical goniometer and corresponding software are used to capture the droplet image and automatically calculate the contact angle from the droplet profile [57]. To ensure statistical reliability, a minimum of 10 measurements per sample are recommended.
- Advanced CA Measurements: For superhydrophobic surfaces or to measure hysteresis, the needle method is recommended. The advancing contact angle is measured as the droplet volume is slowly increased, and the receding angle is measured as liquid is withdrawn back into the needle [61].
Surface Free Energy Calculation Methods
Surface free energy is calculated from contact angle data using thermodynamic models. The most common methods are:
- Owens, Wendt, Rabel, and Kaelble (OWRK) Method: This two-liquid method distinguishes between dispersive and polar components of SFE [59] [60]. It uses the equation:
(1 + cosθ)γ_L = 2[(γ_S^D γ_L^D)^0.5 + (γ_S^P γ_L^P)^0.5]where θ is the contact angle, γ_L is the liquid surface tension, and D and P superscripts refer to dispersion and polar components, respectively [60]. - van Oss-Chaudhury-Good (vOCG) / Lifshitz-van der Waals Acid-Base (LWAB) Method: This more detailed approach subdivides the polar component into Lewis acid (γ+) and Lewis base (γ-) parameters [57] [60]. The equation is:
(1 + cosθ)γ_L = 2(γ_S^LW γ_L^LW)^0.5 + 2(γ_S^+ γ_L^-)^0.5 + 2(γ_S^- γ_L^+)^0.5It requires contact angle data from at least three liquids, including a non-polar one [60]. - Zisman's Method (Critical Surface Tension): This involves plotting the cosine of the contact angle (cos θ) for a series of liquids against their surface tension (γ_L). Extrapolating to cos θ = 1 gives the critical surface tension, which approximates the SFE for low-energy surfaces [57] [60].
Research Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions for Wettability Testing
| Item Name | Function / Application | Examples from Research |
|---|---|---|
| Standard Test Liquids | Used for contact angle measurement and SFE calculation based on their known surface tension components. | Distilled water, Diiodomethane, Glycerol, Ethylene Glycol, Alpha-bromonaphtalene [57] [60]. |
| Abrasive Particles / Sandpapers | Used for surface roughening (e.g., airborne-particle abrasion) or polishing to create defined surface topographies. | 50-μm aluminum oxide particles [23]; Silicon carbide sandpapers of varying grits (P80-P2000) [62]. |
| Bioactive Coatings | Applied to material surfaces to enhance bioactivity, hydrophilicity, and cellular response. | Lithium disilicate-based glass-ceramic spray deposition (GCSD) for zirconia [23]. |
| Chemical Etchants | Used to modify surface chemistry and micro-topography, increasing surface energy and improving adhesion. | 5.0% Hydrofluoric Acid (HF) gel [23] [46]. |
| Dental Adhesives | Used in studies to evaluate the wetting ability and bond strength on material surfaces. | 10-MDP containing universal adhesives, solvent-free resins (e.g., Heliobond) [59]. |
Workflow and Interrelationships in Wettability Analysis
The following diagram illustrates the logical sequence and key decision points in a comprehensive surface wettability assessment workflow for dental materials.
The systematic assessment of surface wettability and free energy is indispensable for advancing dental materials research. Data reveals that surface treatments like glass-ceramic spray deposition for zirconia or specific polishing systems for composites can significantly enhance hydrophilicity and biological response, moving beyond the inherent properties of the base material. Furthermore, a material's performance is dictated by the interplay between its surface energy, roughness, and the surface tension of interacting liquids like adhesives or saliva.
This comparative guide underscores that there is no universally optimal surface. The choice of material and surface treatment must be application-specific: high-energy, hydrophilic surfaces are desirable for osseointegration and cell adhesion, while lower-energy, smoother surfaces may be beneficial for reducing plaque accumulation on orthodontic devices. Researchers are thus equipped to make informed decisions, driving the development of next-generation dental biomaterials with predictable and improved clinical outcomes.
Addressing Clinical Challenges: From Material Degradation to Biofilm Control
The pursuit of minimally invasive dental therapies must be balanced against the potential for iatrogenic damage to dental hard tissues. Two prominent clinical procedures—dental bleaching and laser-assisted decontamination—while highly effective, induce significant surface and compositional alterations in dental substrates. Understanding these changes is critical for optimizing clinical protocols and developing protective strategies. This review synthesizes recent evidence on the surface morphology and chemical composition changes resulting from these procedures, providing a direct comparison of their effects on enamel and dentin. By examining experimental data on mineral content, surface roughness, and microscopic topography, this analysis offers insights into the mechanistic actions of bleaching agents and laser therapies, framing them within the broader context of dental materials research for a scientific audience.
Surface and Elemental Alterations from Dental Bleaching
Mechanisms of Bleaching-Induced Surface Changes
Tooth bleaching relies on the diffusion of hydrogen peroxide (HP) or carbamide peroxide (CP) through enamel, where reactive oxygen species oxidize organic chromogens. However, this process also interacts with the inorganic component of enamel, primarily hydroxyapatite. The mineral dissolution potential is influenced by multiple factors, including peroxide concentration, application time, and crucially, the pH of the bleaching gel. Acidic formulations enhance oxidative action but simultaneously increase mineral solubility, leading to demineralization through calcium (Ca) and phosphate (P) loss [63]. Additionally, light/heat activation may accelerate free radical generation, though its added benefit remains inconsistent across studies and may potentially exacerbate surface alterations in some systems [63].
Comparative Evidence of Bleaching Effects
Table 1: Summary of In-Vitro Studies on Bleaching-Induced Surface and Elemental Changes
| Bleaching Agent | Key Surface Findings (SEM) | Elemental Changes (EDS) | Statistical Significance |
|---|---|---|---|
| BlancOne Ultra+ (35% HP, light-activated) [63] | Diffuse erosion, loss of prismatic clarity | Marked demineralization: Ca 1.42–7.85 at%, P 1.22–6.71 at%; Local C rise to 46.61 at% | Ca and P significantly reduced (p<0.001) |
| Opalescence Quick (45% CP) [63] | Surface roughness, fissures, microcracks | Ca reduced from 11.5-12.5 to 9.53–11.73 at%; P from 7.5-8.9 to 7.41–8.59 at% | Ca and P significantly reduced (p<0.001) |
| Opalescence Boost (40% HP) [63] | Mild superficial restructuring, granular deposits | Variable Ca (13.80–27.94 at%) and P (7.32–14.65 at%) | Ca and P significantly reduced (p<0.001) |
| McInnes Solution [39] | Pronounced surface alterations | Greatest decrease in calcium concentration at T2 (P=0.001) and T4 (P=0.04) | Statistically significant mineral loss |
| Pola Office [39] | Less severe alterations compared to McInnes | Measurable mineral loss | - |
| At-Home Bleaching (10% CP) [63] | - | May not meaningfully reduce enamel microhardness | - |
The severity of surface alterations follows a distinct gradient, with light-activated, high-concentration hydrogen peroxide causing the most pronounced damage. BlancOne Ultra+ (35% HP with light) resulted in the most significant demineralization, with calcium levels plummeting to as low as 1.42 at% [63]. This was accompanied by a substantial local increase in carbon (C), indicating organic residue deposition or possibly the exposure of the organic enamel matrix following mineral loss. Opalescence Quick (45% CP) induced microcracks and fissures, while Opalescence Boost (40% HP) showed milder, more superficial changes with granular deposits [63].
Independent studies on McInnes solution (a potentially more aggressive formulation) confirmed the most significant calcium loss, particularly at specific time points, with SEM revealing pronounced surface damage compared to modified McInnes and Pola Office [39]. This confirms that the gel's formulation, not just peroxide concentration, is a critical determinant of surface safety.
Experimental Protocols in Bleaching Research
Standardized in-vitro methodologies are crucial for comparing outcomes across studies. A typical protocol involves:
- Specimen Preparation: Extracted human molars/premolars are sectioned, with one half serving as the internal control and the other as the experimental specimen. Teeth are embedded in acrylic resin and polished sequentially to create a standardized surface [63] [39].
- Bleaching Application: Gels are applied according to manufacturers' instructions, varying in concentration, application time, and number of sessions. Light-activated systems use specific LED or halogen curing lights for designated periods [63].
- Post-Treatment Analysis: Specimens are stored in saline or artificial saliva to prevent dehydration before analysis. Surface topography is assessed using Scanning Electron Microscopy (SEM) at magnifications from ×500 to ×1100. Elemental composition is quantified via Energy-Dispersive X-ray Spectroscopy (EDS), which measures the atomic percentage (at%) of calcium (Ca), phosphorus (P), oxygen (O), and carbon (C) [63]. Alternatively, colorimetric spectrophotometry following an enamel microbiopsy can be used to quantify extracted calcium and phosphorus ions [39].
Surface and Compositional Changes from Laser Decontamination
Laser-Tissue Interaction Mechanisms
Laser decontamination, used in procedures like peri-implantitis treatment, removes biofilm and alters the surface through photothermal and photomechanical interactions. The primary effects include:
- Ablation and Vaporization: Erbium lasers (Er:YAG) are highly absorbed by water and hydroxyapatite, causing micro-explosions that remove calcified tissues with minimal thermal effect [64].
- Surface Sterilization: Diode lasers (e.g., 810 nm) exert bactericidal effects, eliminating pathogenic bacteria from implant surfaces and periodontal pockets [65] [66].
- Surface Roughening: Laser irradiation increases surface area by creating micro-retentive patterns, which can enhance bonding for restorative materials but may also potentially facilitate biofilm retention if overdone [64].
Table 2: Comparative Analysis of Laser Surface Treatments on Dentin
| Laser Parameter | Surface Morphology (SEM/AFM) | Elemental Composition (EDX) | Bond Strength Outcome |
|---|---|---|---|
| Er:YAG Laser (2940 nm, 200 mJ, 20 Hz) [64] | Rough surfaces, opened dentin tubules | Increased mineral content (Ca/P) vs. control (p<0.05) | Unaffected by surface treatment or bonding type (P>0.05) |
| Femtosecond Laser (800 nm, 90 fs, 400 mW) [64] | Precise ablation with negligible heat loss | Increased mineral content (Ca/P) vs. control (p<0.05) | Unaffected by surface treatment or bonding type (P>0.05) |
| Diode Laser (810 nm, 2.5 W, 50 Hz) [65] | - | - | - |
Unlike bleaching procedures, laser irradiation of dentin surfaces consistently increases the relative mineral content [64]. This occurs because lasers effectively ablate the superficial, more organic and hydrated dentin layer, leaving behind a surface enriched in the underlying mineral phase. Both Er:YAG and femtosecond lasers produce this effect, creating rough surfaces with open dentin tubules ideal for resin cementation, without compromising bond strength [64].
Clinical Efficacy and Surface Outcomes in Peri-Implantitis
Table 3: Clinical Trial Outcomes: Laser vs. Surgical Decontamination
| Treatment Outcome | Test (Diode Laser + Debridement) | Control (Surgical Debridement) |
|---|---|---|
| 12-Month Survival Rate [65] | 76.5% (4 failures in 17 patients) | 100% (0 failures in 19 patients) |
| Prob. Depth (PD) 3 Months [65] | 4.5 mm [3.8, 4.8] | 3.7 mm [3.2, 4.0] |
| Prob. Depth (PD) 12 Months [65] | 4.3 mm [3.7, 4.8] | 3.3 mm [3.1, 3.9] |
| Mucosal Recession 3 Months [65] | 0 mm [0.0, 0.3] | 0.5 mm [0.3, 1.2] |
| Patient Discomfort (VAS) [65] | Significantly lower | Higher |
| Disease Resolution [65] | No significant difference between groups | No significant difference between groups |
A 2025 randomized clinical trial compared non-surgical debridement with repeated diode laser application against surgical treatment for peri-implantitis. While the laser group reported significantly lower patient discomfort, it showed a lower survival rate (76.5% vs. 100%) and less reduction in probing depths compared to surgery [65]. This indicates that while lasers offer a minimally invasive approach, their efficacy in advanced cases may be limited. A separate systematic review confirmed that adjunctive laser therapy (including photodynamic and low-level laser therapy) improves clinical outcomes like probing depth and bleeding on probing compared to mechanical debridement alone, highlighting its value as an adjunctive, rather than standalone, treatment [66].
Experimental Protocols in Laser Research
Standardized laser research protocols ensure reproducible results:
- Laser Application: Specimens are irradiated under water cooling to prevent thermal damage. Parameters (wavelength, power, pulse duration, repetition rate) are meticulously controlled. For example, Er:YAG lasers may be applied at 2940 nm, 200 mJ, and 20 Hz for 20 seconds, while femtosecond lasers use ultra-short pulses (90 fs) at 800 nm [64].
- Clinical Decontamination: In peri-implantitis studies, laser fibers are moved vertically and horizontally in a scanning motion along contaminated implant surfaces. Protocols often include multiple irradiation cycles (e.g., 3 times for 30 seconds) [65].
- Surface Analysis: Post-treatment, surfaces are analyzed using Atomic Force Microscopy (AFM) for nanoscale topography and roughness (Ra), and SEM for micromorphology. EDX quantifies elemental changes, focusing on Ca and P weight% and Ca/P ratios [64].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 4: Essential Materials for Enamel and Dentin Surface Research
| Research Material / Reagent | Function in Experimental Protocols |
|---|---|
| Scanning Electron Microscope (SEM) [63] [39] | High-resolution imaging of surface topography (e.g., microcracks, erosion, porosity). |
| Energy-Dispersive X-ray Spectroscopy (EDS/EDX) [63] [64] | Quantitative elemental analysis of dental hard tissues (e.g., Ca, P, O, C weight% or atomic%). |
| Atomic Force Microscope (AFM) [64] | Nanoscale 3D profiling and surface roughness (Ra) measurements. |
| Colorimetric Spectrophotometer [39] | Quantification of calcium and phosphorus ions dissolved from enamel via microbiopsy. |
| Simulated Body Fluid (SBF) [67] | In-vitro solution mimicking ion composition of human blood plasma, used for demineralization/remineralization cycling studies. |
| Self-curing Acrylic Resin [63] [64] | For embedding and stabilizing tooth specimens during sectioning and polishing. |
| Vanish β-TCP-F Varnish [68] | Commercial remineralizing agent containing β-tricalcium phosphate and fluoride; used in preventive/restorative studies. |
| CPP-ACP-F Paste [68] | Commercial remineralizing agent containing casein phosphopeptide-amorphous calcium phosphate and fluoride. |
Visualization of Experimental Workflows
The following diagrams illustrate the core experimental pathways and logical relationships in studying surface alterations from bleaching and laser procedures.
Bleaching Surface Alteration Pathway
Laser-Tissue Interaction Pathway
Surface Analysis Methodology
The evidence demonstrates that both bleaching and laser decontamination procedures significantly alter dental substrate surfaces, though through fundamentally different mechanisms and with distinct outcomes. Bleaching agents, particularly acidic, high-concentration, light-activated hydrogen peroxide gels, primarily cause surface demineralization, manifesting as increased roughness, porosity, and measurable loss of calcium and phosphorus [63] [39]. In contrast, laser decontamination typically results in surface ablation and increased relative mineral content by removing the superficial organic layer, creating micro-rough surfaces beneficial for adhesion but requiring careful parameter control to avoid excessive roughness that could promote biofilm accumulation [64].
From a clinical materials perspective, these findings necessitate procedure-specific mitigation strategies. For bleaching, the use of neutral-pH formulations and immediate post-treatment remineralization therapies with agents like CPP-ACP-F or β-TCP-F are strongly supported by the evidence to counteract mineral loss [63] [68]. For laser applications, precision in parameter selection is crucial to balance decontamination efficacy with surface preservation.
Future research should focus on optimizing combination therapies that maximize clinical outcomes while minimizing structural damage. The development of "smart" bleaching gels with built-in remineralizing components and standardized laser protocols for specific clinical indications represents promising avenues for advancing minimally invasive dentistry.
Strategies to Mitigate Biofilm Formation on Different Dental Material Surfaces
Biofilm formation on dental material surfaces is a primary cause of restorative failure, secondary caries, and periodontal complications. The complex oral environment, characterized by diverse microbial communities and constant exposure to saliva, creates ideal conditions for bacterial adhesion and proliferation on dental materials. The surface properties of these materials—including roughness, chemical composition, and bioactivity—significantly influence the initial attachment of microorganisms and subsequent biofilm development. This guide provides a comparative analysis of biofilm formation across different dental material classes, supported by experimental data and detailed methodologies, to inform the development of next-generation, biofilm-resistant dental biomaterials.
Comparative Analysis of Biofilm Formation on Dental Materials
Restorative Materials
The inherent properties of restorative materials significantly influence bacterial adhesion and biofilm accumulation. The table below summarizes quantitative findings from comparative studies.
Table 1: Bacterial Adhesion and Surface Roughness of Restorative Materials
| Material Type | Specific Product | Bacterial Adhesion (CFUs) | Surface Roughness | Key Findings | Source |
|---|---|---|---|---|---|
| Alkasite | Cention N | Lowest (p<0.001) | Higher than nanohybrid composite | Significantly impedes bacterial adhesion despite higher roughness | [69] |
| Nanohybrid Resin Composite | Tetric N-Ceram | Highest | Smoothest (p=0.002) | Smoothest surface but highest bacterial adhesion | [69] |
| Glass Hybrid Restorative | Equia Forte HT Fil | Intermediate | Higher than nanohybrid composite | Lower mean bacterial cell count than nanohybrid composite | [69] |
Analysis: The data reveals a complex relationship between surface roughness and bacterial adhesion. While nanohybrid composite achieved the smoothest surface, it exhibited the highest bacterial adhesion, suggesting that chemical composition and ion release may be more critical factors than roughness alone for biofilm mitigation [69]. Alkasite (Cention N) demonstrated superior resistance to bacterial adhesion, attributed to its bioactive ion release (calcium, fluoride, hydroxide), which creates an environment less conducive to bacterial survival [69].
Orthodontic Brackets
Orthodontic appliances, with their complex geometries, are particularly susceptible to biofilm accumulation, increasing the risk of white spot lesions.
Table 2: Biofilm Formation on Different Orthodontic Bracket Types
| Bracket Material | Relative Biofilm Formation | Key Findings | Source |
|---|---|---|---|
| Plastic | Highest | Greatest risk of inducing white spot lesions due to high biofilm accumulation | [70] |
| Ceramic | Intermediate | -- | [70] |
| Metal | Lowest | -- | [70] |
Analysis: The study employed a novel bioluminescence method using S. mutans UA159.renG, which allows for accurate quantification of active biofilm formation on complex structures. The significantly higher biofilm formation on plastic brackets underscores the importance of material selection in orthodontic treatment to mitigate caries risk [70].
Advanced Material Strategies for Biofilm Resistance
Material Modification and Functionalization
Innovative approaches to modify existing materials have shown significant promise in enhancing biofilm resistance.
- Zwitterionic Additives: Incorporating zwitterionic materials like 2-methacryloyloxyethyl phosphorylcholine (MPC) or sulfobetaine methacrylate (SB) into 3D-printed PMMA dental base polymers demonstrated a significant reduction in bacterial and biofilm adhesion due to their potent protein-repellent properties. This resistance was maintained even after hydrothermal fatigue, without significant deterioration of mechanical properties [71].
- Imidazolium Salt Modification: Modifying glass ionomer cement (GIC) with the imidazolium salt C16MImCl conferred strong antibiofilm activity against multidrug-resistant Candida species without compromising the material's micromechanical properties or hypoallergenicity. This presents a promising strategy for combating fungal biofilm formation [72].
- Bioactive Hydrogels: Hydrogels based on natural polymers like chitosan, alginate, and agarose are being engineered as delivery systems for antibacterial agents (e.g., peptides, nanoparticles) or remineralization promoters. Their 3D network structure provides prolonged retention and release of active ingredients at the target site, offering a synergistic approach to caries management [73].
Targeting the Biofilm Lifecycle
Novel therapeutic strategies focus on disrupting key stages of biofilm development rather than merely killing planktonic bacteria.
- Enzymatic Disruption: Enzymes such as Dispersin B, DNAases, and glucanohydrolases (mutanase, dextranase) target the structural integrity of the biofilm matrix. By degrading extracellular polymeric substances (EPS), proteins, and e-DNA, these enzymes disrupt the biofilm scaffold, facilitating its removal and enhancing the efficacy of antimicrobial agents [74].
- Quorum Sensing Inhibitors: These compounds interfere with the bacterial cell-to-cell communication system (quorum sensing), which regulates biofilm formation and virulence factor expression. By disrupting this process, they can prevent biofilm maturation without inducing bacterial death, thereby reducing selective pressure for resistance [74].
Essential Experimental Workflows
Standardized Protocol for Evaluating Bacterial Adhesion
The following methodology is commonly used for in vitro assessment of bacterial adhesion to dental materials [69].
Diagram 1: Bacterial Adhesion Assessment Workflow
Detailed Experimental Protocol:
Specimen Preparation:
- Prepare material discs (e.g., 8 mm diameter × 2 mm height) using metal molds according to manufacturers' instructions [69].
- Polish all specimens to a standardized finish using a series of polishing discs (e.g., Sof-Lex system) to control for surface roughness [69].
- Sterilize specimens prior to testing (e.g., using ethylene oxide or UV light).
Microbial Adhesion Assay:
- Grow a standard cariogenic strain like Streptococcus mutans in a suitable broth (e.g., Tryptic Soy Broth) for 24 hours at 37°C.
- Inoculate specimens with bacterial suspension and incubate for a set period (e.g., 24 hours for initial adhesion, 96 hours for mature biofilm) under anaerobic conditions at 37°C [69].
- Quantify adhered bacteria using:
- Optical Density (OD): Measure turbidity of suspension after dislodging bacteria.
- Colony Forming Units (CFUs): Plate serial dilutions of dislodged bacteria onto agar plates and count colonies [69].
- Scanning Electron Microscopy (SEM): Fix biofilms and image to visualize and count bacterial cells on the surface [69].
Surface Roughness Measurement:
- Assess surface roughness (Ra) using a contact or non-contact profilometer.
- Take multiple measurements across different areas of the specimen to obtain an average Ra value [69].
Statistical Analysis:
- Perform statistical tests (e.g., ANOVA, Tukey's test) to compare bacterial adhesion and surface roughness between material groups.
- Conduct correlation analysis to determine the relationship between surface roughness and bacterial adhesion metrics [69].
Mechanism of Bacterial Adhesion to Dental Surfaces
Understanding the fundamental process of adhesion is key to developing inhibition strategies.
Diagram 2: Bacterial Adhesion Mechanism
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Reagents for Biofilm Research on Dental Materials
| Reagent/Material | Function in Experiment | Application Example |
|---|---|---|
| Streptococcus mutans UA159 | Model cariogenic bacterium for initial adhesion and biofilm studies. | Evaluating bacterial adhesion to restorative materials and orthodontic brackets [69] [70]. |
| renG-expressing S. mutans | Engineered strain expressing Renilla luciferase for bioluminescence-based quantification of live biofilm. | Accurately assessing active biofilm formation on complex structures like brackets [70]. |
| Sof-Lex Polishing System | Aluminum oxide and diamond-impregnated discs for standardized surface finishing of material specimens. | Controlling for surface roughness variable across different material groups [69]. |
| Artificial Saliva | Simulates the ionic composition and pH of human saliva for in vitro pellicle formation and biofilm growth. | Creating clinically relevant conditions for microbial adhesion assays [72]. |
| Zwitterionic Monomers (MPC, SB) | Additives that impart potent protein-repellent properties to polymeric materials like PMMA. | Developing 3D-printed dental resins with durable biofilm resistance [71]. |
| Imidazolium Salts (C16MImCl) | Organic salts with strong antimicrobial and antibiofilm activity against bacteria and fungi. | Modifying glass ionomer cements to prevent Candida biofilm formation [72]. |
| Dispersin B, DNAases, Glucanohydrolases | Enzymes that degrade key components (e.g., polysaccharides, e-DNA) of the biofilm EPS matrix. | Disrupting pre-formed mature biofilms as a non-biocidal strategy [74]. |
The mitigation of biofilm formation on dental materials requires a multifaceted approach that considers material composition, surface characteristics, and bioactive functionality. Evidence indicates that while surface roughness is a critical factor, the bioactive potential of a material—such as ion release in alkasites or the protein-repellent nature of zwitterionic polymers—can be a more powerful determinant of its biofilm resistance. The future of antimicrobial dental materials lies in smart, multifunctional strategies that combine optimal physical properties with biological activity, such as enzymatic biofilm disruption, targeted drug delivery via hydrogels, and non-biocidal quorum sensing inhibition. These approaches, validated through robust and standardized experimental protocols, hold the key to developing longer-lasting restorations and improving overall oral health outcomes.
Impact of Oral Environment and Aging on Material Surface Stability
The long-term success of dental restorative materials depends on their ability to maintain surface stability in the harsh oral environment, which subjects materials to thermal fluctuations, moisture, pH changes, and mechanical stresses. This complex interplay of factors can trigger degradation processes that compromise material integrity, leading to clinical failure. Understanding how different material classes respond to these challenges is fundamental to advancing restorative dentistry. This guide provides a comparative analysis of the surface stability of contemporary dental materials, including ceramics, resins, and metals, under simulated aging conditions, offering researchers objective data on their performance characteristics.
Comparative Performance of Dental Materials
The surface stability of dental materials is evaluated through key properties such as phase composition, hardness, color stability, and resistance to water sorption and solubility. The following tables synthesize experimental data from recent in-vitro studies, providing a direct comparison of material performance before and after accelerated aging.
Table 1: Surface Stability of CAD/CAM Ceramics After Surface Treatments and Aging
| Material Type | Key Surface Treatment | Aging Protocol | Primary Findings Post-Aging | Reference |
|---|---|---|---|---|
| 3Y-TZP Infrastructure Zirconia | Sandblasting (50 µm Al₂O₃) | Hydrothermal aging (EN ISO 13356:2015) | - Phase Transformation (t→m): ↑↑- Compressive Strength: ↓↓- Surface Damage: Severe | [75] |
| Super-Translucent Zirconia | Sandblasting (50 µm Al₂O₃) | Hydrothermal aging (EN ISO 13356:2015) | - Phase Transformation (t→m): ↑↑↑- Compressive Strength: ↓↓↓- Surface Damage: Most Severe | [75] |
| 3Y-TZP Infrastructure Zirconia | Polishing | Hydrothermal aging (EN ISO 13356:2015) | - Phase Transformation (t→m): Minimal- Compressive Strength: Least reduction- Surface Quality: Best retained | [75] |
| Zirconia-Reinforced Lithium Silicate | Milling (CAD/CAM) | N/A (Initial characterization) | - Mean Roughness (Ra): 1.97 µm- Contact Angle (Water): 42.48°- Surface Skewness (Rsk): Negative (smoothest) | [76] |
| Lithium Disilicate | Milling (CAD/CAM) | N/A (Initial characterization) | - Mean Roughness (Ra): 2.86 µm- Contact Angle (Water): 31.41° (most hydrophilic) | [76] |
Table 2: Stability of Resin-Based Denture and Restorative Materials
| Material Type | Key Surface Treatment | Aging Protocol | Primary Findings Post-Aging | Reference |
|---|---|---|---|---|
| 3D-Printed Denture Base | Glaze application | 7-day water immersion; 2000 thermocycles | - Water Sorption: Lowest values- Water Solubility: Highest after thermocycling- ISO 4049 Compliance: Within limits | [77] |
| Conventional Heat-Cured PMMA | Glaze application | 7-day water immersion; 2000 thermocycles | - Water Sorption: Highest values- Water Solubility: Generally lowest | [77] |
| 3D-Printed Experimental Soft Liner | Post-curing (10 min, 395 nm) | 3-month coffee immersion | - Shore A Hardness: Highest- Color Stability (ΔE): Moderate- Surface Roughness (Ra): Highest | [5] |
| Silicone-Based Soft Liner | Manufacturer's instructions | 3-month coffee immersion | - Shore A Hardness: Lowest- Color Stability (ΔE): Best (most stable)- Surface Roughness (Ra): Lowest | [5] |
| 3D-Printed Restorative Resin | Printing at 0° and 90° angles | Artificial weathering (light, temperature, humidity) | - Color Change (ΔE00): Unacceptable (> 1.8 AT00)- Optical Properties: Significant change in R%, T%, S, K | [78] |
Experimental Protocols for Assessing Surface Stability
Phase Transformation Analysis in Zirconia Ceramics
Objective: To quantify the tetragonal-to-monoclinic (t→m) phase transformation in zirconia after surface treatments and hydrothermal aging, a key indicator of material degradation and potential strength reduction [75].
Methodology:
- Specimen Preparation: CAD/CAM-milled zirconia cubes (8x8x8 mm) are sintered according to manufacturer specifications. Groups include infrastructure zirconia (3Y-TZP) and super-translucent multilayered monolithic zirconia.
- Surface Treatment: Specimens are divided into subgroups: untreated control, sandblasted with 30 µm or 50 µm Al₂O₃ particles at a standard pressure and distance, and polished with sequential abrasive grits.
- Hydrothermal Aging: Accelerated aging is performed in a steam environment following EN ISO 13356:2015 standards, typically at 134°C and 2 bar pressure for several hours to simulate years of in-vivo service.
- Phase Analysis: X-ray Diffraction (XRD) with Rietveld refinement is used to quantify the percentage of monoclinic phase content. Raman spectroscopy provides complementary data on crystalline phases and local stresses.
Water Sorption and Solubility Testing for Polymers
Objective: To determine the susceptibility of denture base polymers to water uptake and mass dissolution, which can lead to plasticization, dimensional changes, and reduced mechanical properties [77] [79].
Methodology:
- Specimen Fabrication: Disc-shaped specimens (Ø15 x 2 mm) are fabricated via conventional heat-curing, milling, or 3D printing. 3D-printed specimens undergo varied post-curing (e.g., 10-60 minutes, in air or glycerin).
- Conditioning: Specimens are dried in a desiccator with silica gel at 37°C until constant mass (m₁) is achieved, weighed on a microbalance (±0.0001 g).
- Water Immersion: Specimens are immersed in distilled water at 37°C for 7 days, then patted dry and weighed to obtain saturated mass (m₂).
- Re-drying: Specimens are returned to the desiccator until constant mass (m₃) is reached again.
- Calculation: Water sorption (Wsp) and solubility (Wsl) in µg/mm³ are calculated as:
- ( W{sp} = (m2 - m3) / V )
- ( W{sl} = (m1 - m3) / V ) where V is the specimen volume.
- Additional Aging: Protocols may include immersion in denture cleanser solutions or thermocycling (e.g., 2000 cycles between 5°C and 55°C) to simulate clinical use.
Color and Optical Property Assessment
Objective: To evaluate the color stability and changes in fundamental optical properties of resin-based restorative materials after artificial aging [78].
Methodology:
- Specimen Preparation: Square-shaped specimens (10x10 mm) are 3D-printed at different build orientations (e.g., 0° and 90°).
- Baseline Measurement: Initial color coordinates (L, a, b*) are measured with a spectrophotometer against white, black, and grey backgrounds. Spectral reflectance (R%) and transmittance (T%) are recorded.
- Optical Calculation: The Kubelka-Munk (K-M) theory is applied to calculate:
- Scattering (S) and absorption (K) coefficients
- Light reflectivity (RI)
- Infinite optical thickness (X∞)
- Artificial Aging: Specimens undergo artificial weathering in a climate chamber, simulating oral conditions with controlled cycles of light exposure, temperature (e.g., 4-60°C), and relative humidity (e.g., 0-100%).
- Post-Aging Measurement: Color and optical measurements are repeated. The CIEDE2000 (ΔE₀₀) color difference formula is used with 50:50% perceptibility (PT₀₀=0.8) and acceptability (AT₀₀=1.8) thresholds.
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for Dental Material Aging Studies
| Reagent/Material | Function in Experimental Protocol | Typical Application/Concentration |
|---|---|---|
| Aluminum Oxide (Al₂O₃) Particles | Surface abrasion to simulate clinical adjustments and study adhesion/phase stability. | Sandblasting at 0.1-0.3 MPa pressure with 30-50 µm particle size [75]. |
| Distilled Water | Medium for water sorption/solubility tests and hydrothermal aging; simulates oral moisture. | Immersion at 37°C for 7 days to 3 months; hydrothermal aging at 134°C, 2 bar pressure [77] [75]. |
| Coffee Solution | Staining agent for accelerated color stability testing of polymeric materials. | 3.6 g instant coffee in 300 mL boiling distilled water; daily renewal [5] [78]. |
| Instant Coffee (e.g., Nescafé Classic) | Standardized chromogenic challenge for color stability assays. | 3.6 g instant coffee in 300 mL boiling distilled water; daily renewal [5]. |
| Arsenazo III Reagent | Colorimetric quantification of calcium ions in mineral loss studies (e.g., enamel biopsies). | Spectrophotometric analysis at 630 nm absorbance [39]. |
| Hydrochloric Acid (HCl) in Glycerol | Enamel microbiopsy etchant for controlled mineral sampling from tooth surfaces. | 1.6 M HCl in 70% glycerol (v/v); 5 µL applied for 20 seconds [39]. |
| Silver Vanadate (AgVO₃) Nanoparticles | Bio-functionalization additive to impart antimicrobial properties to impression materials. | Incorporated into alginate at 2.5%, 5%, and 10% concentrations by weight [8]. |
The comparative data presented in this guide reveals a critical trade-off in dental material selection. Zirconia ceramics offer superior strength but are susceptible to phase transformation from abrasive treatments, whereas highly translucent formulations are less stable. 3D-printed resins provide fabrication efficiency but show significant vulnerability to color change and water solubility. Polishing, over sandblasting, and glaze application are effective surface treatments for enhancing stability. Future research should focus on nano-engineered material compositions and hybrid surface functionalization to develop next-generation dental materials capable of withstanding the complex oral environment over extended service lifetimes.
Fatigue Strength and Long-Term Durability Under Cyclic Loading
Fatigue failure is a predominant mode of mechanical failure in dental restorations, accounting for an estimated 80% to 90% of mechanical service failures [80]. It occurs when a material undergoes progressive, localized structural damage due to fluctuating stresses that are below the material's ultimate tensile strength. In the oral environment, restorations are subjected to relentless cyclic loading from mastication, swallowing, and parafunctional habits, combined with challenging environmental conditions such as moisture, temperature fluctuations, and varying pH levels [81]. Understanding fatigue strength—the maximum stress a material can withstand for a given number of cycles—and long-term durability is therefore paramount for predicting clinical longevity and ensuring patient safety.
This guide objectively compares the fatigue performance of major dental material classes, including ceramics, resin composites, and metals, by synthesizing current experimental data. The analysis is framed within a broader research context examining how surface morphology and chemical composition dictate performance under cyclic loading. The objective is to provide researchers, material scientists, and dental professionals with a data-driven foundation for material selection and future development.
Fundamental Principles of Fatigue Testing
Definition and Key Metrics
Fatigue testing evaluates a material's behavior under repeated (cyclic) stress and loading [82]. Key properties measured include:
- Fatigue Life (N): The number of cycles a material can endure before failure occurs [82].
- Endurance Limit: The maximum stress level below which a material can theoretically endure an infinite number of cycles without failing. Some materials, like certain steels, exhibit a clear endurance limit, while others, like aluminum and many polymers, do not [82].
- Stress-Life (S-N) Curve: A plot of the applied stress amplitude (S) against the logarithm of the number of cycles to failure (N). This curve is fundamental for determining a material's fatigue characteristics [80].
- Fatigue Strength: The stress value at which a material fails after a specific number of cycles (e.g., 50,000 or 1 million cycles) [81].
Standardized Testing Methodologies
Standardized experimental protocols are critical for generating comparable and reliable data. The following are core methodologies:
- High-Cycle Fatigue (HCF) Testing: Characterized by low stress amplitudes (below the material's yield strength) and a high number of cycles (typically > 10,000). It leads to elastic deformation and is governed by ASTM E466 standards. This test is relevant for components experiencing high-frequency, low-strain vibrations [80].
- Low-Cycle Fatigue (LCF) Testing: Characterized by high stress amplitudes (exceeding the yield strength) and a low number of cycles (typically < 10,000). It results in plastic deformation and is conducted according to ASTM E606. This test simulates high-strain environments [80].
- Mouth-Motion Step-Stress Fatigue Test: A clinically relevant protocol that simulates the oral environment. It typically involves submerging specimens in water baths at body temperature and applying cyclic loads in a stair-step manner, increasing the load after a set number of cycles until failure. This method allows for efficient prediction of failure probabilities and often reveals failure modes not seen in static tests [81].
Table 1: Key Fatigue Testing Standards and Conditions
| Test Type | Standard Protocol | Frequency Range | Stress/Strain Control | Primary Output |
|---|---|---|---|---|
| High-Cycle Fatigue (HCF) | ASTM E466 | 20 Hz - 100 Hz | Stress-controlled (Load) | S-N Curve (Stress vs. Cycles) |
| Low-Cycle Fatigue (LCF) | ASTM E606 | 0.25 Hz - 5 Hz | Strain-controlled | ε-N Curve (Strain vs. Cycles) |
| Mouth-Motion Fatigue | Customized protocols | ~1-2 Hz (simulating mastication) | Load or Displacement | Failure Load & Cycle Count |
Experimental Workflow for Dental Material Fatigue Analysis
The following diagram outlines a generalized experimental workflow for evaluating the fatigue resistance of dental materials, integrating key steps from specimen preparation to data analysis.
Diagram 1: Experimental workflow for dental material fatigue analysis.
Comparative Fatigue Performance of Dental Materials
CAD/CAM Ceramic Materials
Lithium Disilicate Glass-Ceramic (e.g., IPS e.max CAD) This material is renowned for its combination of mechanical strength and aesthetic qualities, making it ideal for anterior and posterior restorations [81].
- Flexural Strength: Ranges from 360 to 400 MPa [81].
- Fatigue Resistance: Its durability stems from an interlocking crystal structure that strongly resists crack propagation. The slow crack growth parameter (n) is approximately 14.2, indicating good resistance to subcritical crack extension under cyclic loads [81].
- Design Constraint: Requires a minimum thickness of 1.5 mm to ensure a longevity of over five years under a moderate 100 N load. Thinner designs are prone to premature failure [81].
- Performance Data: The ratio of fatigue strength to flexural strength is about 0.58 at 50,000 cycles, demonstrating consistent performance under stress [81].
Zirconia (e.g., 3-5 mol% Yttria-stabilized) Zirconia offers exceptional fracture resistance and is a game-changer for minimally invasive dentistry, particularly in the posterior region [81].
- Key Mechanism: Its polycrystalline structure undergoes transformation toughening, a stress-induced phase change that compresses the tip of an advancing crack, effectively halting its propagation [81].
- Fatigue Resistance: Zirconia's exceptional strength allows for ultra-thin designs. A 0.7 mm thick zirconia crown can withstand a 50 N load for up to 24 years in simulation models. Even under a high 120 N load, it can last about 1.9 years, outperforming many alternatives at that thickness [81].
- Surface Treatment Consideration: Airborne-particle abrasion, used to improve adhesion, increases surface roughness, which can lead to pronounced bacterial adhesion (Streptococcus mutans). However, applying polyelectrolyte multilayer coatings can reduce this bacterial adhesion by up to 50% without compromising the core mechanical properties [83].
CAD/CAM and Direct Resin Composites
This category includes materials like Brilliant Crios, Cerasmart 270, and direct restorative composites. They combine a resin matrix with ceramic fillers, offering unique mechanical properties [81].
- Flexural Strength: Generally moderate compared to high-strength ceramics.
- Fatigue Resistance: These materials exhibit high damage tolerance. Their fatigue-to-flexural strength ratios range from 0.57 to 0.65 at 50,000 cycles, similar to lithium disilicate [81]. In mouth-motion tests, monolithic resin composite crowns can withstand loads as high as 1,700 N with only minor surface damage, whereas glass-ceramics often fracture catastrophically at loads as low as 450 N [81].
- Failure Mode: The resin matrix allows for energy absorption, reducing the likelihood of sudden, catastrophic fracture. Instead, they may exhibit time-dependent creep (gradual deformation) after prolonged cyclic testing (e.g., 3 million cycles) [81].
Acrylic Denture Base Resins
Fatigue failure is a common clinical occurrence for dentures, often resulting from repeated small flexural stresses rather than a single overload [84].
- Comparative Performance: A study comparing five acrylic resins found that high-impact heat-polymerized resin (Lucitone 199) exhibited the highest fatigue resistance. Its performance was statistically superior to self-cured and light-cured resins [84].
- Material Variation: The study concluded that denture base materials have widely varying fatigue resistance, underscoring the importance of material selection based on clinical needs [84].
Table 2: Comparative Summary of Dental Material Fatigue Performance
| Material Class | Example Product | Flexural Strength (MPa) | Key Fatigue Performance Indicator | Clinical Consideration |
|---|---|---|---|---|
| Lithium Disilicate | IPS e.max CAD | 360-400 [81] | Fatigue strength ratio: 0.58 @ 50k cycles [81] | Requires adequate thickness (≥1.5mm) [81] |
| Zirconia | 3-5 mol% Yttria | Very High [81] | 0.7mm crown lasts ~24 years @ 50N load [81] | Excellent for thin, strong crowns; limited aesthetics [81] |
| CAD/CAM Composite | Cerasmart 270 | Moderate [81] | Fatigue strength ratio: 0.57-0.65 @ 50k cycles [81] | High damage tolerance; susceptible to creep [81] |
| High-Impact Acrylic | Lucitone 199 | Not Specified | Highest fatigue resistance among denture resins [84] | Superior for withstanding long-term flexural fatigue [84] |
| Direct Resin Composite | Various | Varies | Can endure up to 1,700 N with minor damage [81] | Fails by deformation, not catastrophic fracture [81] |
Advanced Characterization and Analysis Techniques
Multi-Modal Damage Monitoring
To comprehensively characterize damage evolution, researchers employ complementary techniques that capture both macro- and micro-scale events.
- Acoustic Emission (AE) Monitoring: AE parameters are highly sensitive to the initiation and growth of micro-cracks. Each micro-fracture event releases a stress wave, which is detected by sensors. This allows for real-time tracking of internal damage and localization of failure origins [85].
- Resistivity Measuring: The electrical resistivity of a material changes as macro-cracks form and connect, altering the conduction path. Resistivity-based damage variables can effectively indicate the connectivity state of cracks within the material, providing a macroscopic view of damage [85].
- Combined Approach: Using both AE and resistivity monitoring provides a more accurate characterization of the damage state, as micro-crack initiation and macro-crack connection occur simultaneously during loading [85].
The Researcher's Toolkit: Essential Reagents and Materials
The following table details key materials and reagents used in the preparation, testing, and analysis of dental materials for fatigue research.
Table 3: Essential Research Reagent Solutions and Materials
| Item Name | Function / Application | Research Context |
|---|---|---|
| Polyelectrolyte Multilayer (e.g., Chitosan, Poly(acrylic acid)) | Surface coating to reduce bacterial adhesion on abraded zirconia [83]. | Studying biofilm-material interactions and surface modifications. |
| Acoustic Emission (AE) Sensors | To monitor and record high-frequency stress waves generated by micro-crack formation within a material under load [85]. | Essential for real-time, in-situ monitoring of damage evolution during fatigue testing. |
| Resistivity Measurement Setup | To measure changes in electrical resistance of a material specimen, which correlates with the development of internal cracks [85]. | Used to establish macroscopic damage variables based on crack connectivity. |
| Coffee Solution | A standardized staining agent (e.g., according to ISO) used in discoloration studies to simulate real-world dietary exposure [86]. | Evaluating the aesthetic longevity and stain resistance of composite resins. |
| Aluminum Oxide Abrasive | For controlled airborne-particle abrasion of material surfaces (e.g., zirconia) to modify surface roughness and improve adhesion [83]. | Surface pretreatment studies and investigating the effects of surface morphology on properties. |
The fatigue strength and long-term durability of dental materials are dictated by a complex interplay of factors, including chemical composition, microstructure, and resulting surface morphology. The data synthesized in this guide demonstrates a clear performance spectrum:
- Zirconia excels in high-strength, thin-design applications due to its transformation toughening mechanism.
- Lithium Disilicate provides an optimal balance of strength and aesthetics but requires careful attention to minimum thickness.
- CAD/CAM Composites offer superior damage tolerance and resistance to catastrophic fracture, though they may be susceptible to long-term deformation.
- Material-specific performance varies significantly, as seen in acrylic resins, where high-impact formulations vastly outperform conventional and self-cured types.
Future research should focus on standardizing fatigue testing protocols that more accurately mimic the complex oral environment, including simultaneous mechanical, thermal, and chemical challenges. Furthermore, the development of novel composite materials and surface modifications, such as anti-bacterial polyelectrolyte coatings, holds promise for enhancing the functional longevity of dental restorations. A deep understanding of the relationships between material structure, properties, and clinical performance remains the foundation for advancing restorative dentistry.
Dental implantology has revolutionized oral rehabilitation, yet infections related to biomedical implants remain a significant challenge. Biomaterials inherently risk forming bacterial adhesions and biofilms, which can lead to peri-implantitis and subsequent implant failure [87]. The global threat of antimicrobial resistance (AMR) further complicates this picture, with bacterial infections causing millions of deaths annually and antibiotic-resistant strains becoming increasingly prevalent [88] [89].
Innovative surface modifications and advanced drug delivery systems present promising strategies to overcome these challenges. These approaches aim to enhance osseointegration while providing robust antimicrobial protection through localized, controlled release mechanisms. This review comprehensively compares current technologies—evaluating surface morphology alterations, chemical nanocoatings, and sophisticated drug-carrier systems—to provide researchers and clinicians with evidence-based insights for selecting optimal antimicrobial strategies in dental implantology.
Surface Morphology Modifications for Enhanced Biocompatibility and Antimicrobial Properties
Surface morphology plays a crucial role in determining biological responses to dental implants, influencing everything from bacterial adhesion to osseointegration. Researchers have developed various strategies to engineer implant surfaces that favor tissue integration while discouraging microbial colonization.
Micro/Nano Hierarchical Structures
Bionic micro/nano hierarchy structures represent a significant advancement in implant surface engineering. These structures typically consist of titanium surface microstructures with internal TiO₂ nanotubes (TNTs) that provide drug-carrying capacity. Research demonstrates that these textured surfaces markedly reduce wear volume and depth compared to untextured samples, primarily due to reduced contact area of the microstructure. The microstructure also protects TNTs from damage during friction, maintaining their drug-delivery functionality. Critically, these hydrophilic biomimetic anti-wear hierarchies promote MC3T3-E1 cell adhesion and proliferation while exhibiting no cytotoxic effects [90].
Table 1: Comparison of Surface Modification Techniques for Dental Implants
| Technique | Key Features | Biological Effects | Limitations |
|---|---|---|---|
| Bionic Micro/Nano Hierarchy [90] | Microstructures with internal TiO₂ nanotubes (TNTs); drug-carrying capacity | Enhanced wear resistance; promotes cell adhesion/proliferation; no cytotoxicity | Complex fabrication process |
| Laser Irradiation [91] | Alters chemical composition and topography; double-wavelength approach | Efficient decontamination; preserves microstructure with proper parameters | Potential for surface alteration with incorrect parameters |
| CAD/CAM Post-Processing [92] | Sandblasting with alumina particles followed by polishing | Low surface roughness (Sa); reduced monoclinic phase; better mechanical properties | Sequence-dependent results; improper sequencing degrades properties |
| Nanotopography [93] | Physically hostile surface with nanoprotrusions | Kills bacterial cells via biomechanical damage; prevents adhesion | Manufacturing consistency challenges |
Laser-Based Surface Treatments
Laser irradiation techniques have emerged as precise methods for implant surface modification and decontamination. Studies evaluating double-wavelength laser irradiation (Er,Cr:YSGG 2780 nm/diode 940 nm) on commercial titanium implants have demonstrated significant alterations in surface chemical composition and topography while efficiently removing artificial biofilm. When performed under controlled parameters with water spray, these treatments avoid adverse thermal effects, maintaining temperatures close to the applied water-spray temperature (20–24°C) without damaging implant microstructure [91].
The critical importance of surface characterization in evaluating these modifications cannot be overstated. Advanced techniques like white light laser profilometry (interferometry) provide three-dimensional characterization through spatial parameters (Sₐ, Sds, Sdr), offering more comprehensive data than simple profile measurements. Research indicates that areal parameters Sₐ and Sq provide better readability than 2-D parameters Ra and Rq, enabling more accurate correlation between surface topography and biological response [91] [92].
Surface Modification Strategies Diagram
Antimicrobial Nanocoatings: Composition and Efficacy
Nanocoatings represent a paradigm shift in implant protection, utilizing engineered nanomaterials (ENMs) with intrinsic antimicrobial properties. These coatings can be categorized based on their composition and mechanism of action.
Metal and Metal Oxide Nanocoatings
Silver nanoparticles demonstrate potent broad-spectrum antimicrobial activity through multiple mechanisms, including induction of hydroxyl radical formation in both Gram-positive and Gram-negative bacteria. Studies show that AgNPs (38 nm) applied to surgical nylon threads create zones of inhibition >15 mm against pathogens including Pseudomonas aeruginosa, Bacillus subtilis, and Staphylococcus aureus [93]. The concentration-dependent behavior is crucial—while 0.5% (w/v) AgNPs showed little effect on cell viability, 4% (w/v) demonstrated 80% reduced cell viability with several-fold higher silver release, highlighting the importance of dosage optimization.
Zinc oxide and titanium oxide nanocoatings provide alternative antimicrobial mechanisms, often involving reactive oxygen species (ROS) production. ZnO nanoparticles (12–27 nm) applied to TiZrNb alloy via plasma electrolytic oxidation achieved >2 log reduction in Staphylococcus aureus biofilm after just 2 hours. Dual-layer ZnO nanospheres and nanorods on Ti or Ti–Zr surfaces demonstrated interesting release kinetics: rapid release from nanospheres provided up to 2-fold higher short-term antibacterial activity, while slower release from nanorods offered longer-term protection [93].
Polymeric and Composite Nanocoatings
Polymer-based systems offer versatile platforms for antimicrobial delivery. Quaternary ammonium-modified gold nanoclusters applied to orthodontic devices reduced Streptococcus mutans biofilm mass by 85% and viability by 95%, with excellent stability and no hemolysis or significant cellular toxicity [93]. Similarly, nanocomposite dental resins incorporating TiO₂ or MgO nanoparticles demonstrated concentration-dependent antibacterial effects, with 4% MgO NP reducing Streptococcus mutans viability by 99.4% [93].
Table 2: Efficacy of Antimicrobial Nanocoatings Against Pathogenic Microorganisms
| Coating Type | Target Organisms | Key Results | Source |
|---|---|---|---|
| Ag NPs (38 nm) on nylon | 6 Gram+, 4 Gram- bacteria, 3 molds, 1 yeast | >15 mm ZOIs against P. aeruginosa, B. subtilis, M. luteus; >12 mm against fungi | [93] |
| ZnO NPs on TiZrNb alloy | Staphylococcus aureus | >2 log reduction in biofilm after 2 hours | [93] |
| Dual-layer ZnO nanospheres/nanorods | Escherichia coli, Staphylococcus aureus | 30-70% S. aureus biofilm inhibition; 60-80% in vivo inhibition over 2 weeks | [93] |
| Quaternary ammonium-modified gold nanoclusters | Streptococcus mutans | 85% reduced biofilm mass; 95% reduced viability | [93] |
| MgO NPs in dental resin | Streptococcus mutans | 1% MgO reduced viability by 67.7%; 4% MgO by 99.4% | [93] |
Advanced Drug Delivery Systems for Localized Antimicrobial Therapy
Conventional antibiotic administration often fails to achieve therapeutic concentrations at implant-tissue interfaces while causing systemic side effects. Advanced drug delivery systems address these limitations through localized, controlled release mechanisms.
Hydrogel Microspheres (HMS)
Hydrogel microspheres represent a significant advancement in antimicrobial delivery, overcoming limitations of conventional carriers like liposomes (low drug-loading capacity, poor stability) and dendrimers (cytotoxicity, high production costs) [89]. HMSs offer distinct advantages including small dimensions, high drug-loading capacity, deep-targeted delivery capability, and adjustable physicochemical properties. Their "shear-thinning" or "solid-like" rheological properties enable injection without additional chemical modifications, enhancing clinical applicability [89].
Preparation methods significantly influence HMS performance. The batch emulsion method offers simple processing and suitability for large-scale production but typically yields polydisperse microspheres with potential batch-to-batch variation. Microfluidic methods provide excellent monodispersity and reduced variation through precise regulation of the production process. Combined with electrospray technology, microfluidics can produce hundreds of HMSs per minute, enabling efficient large-scale production [89].
Polymer-Based Delivery Systems
Biodegradable polymeric carriers like poly lactic-co-glycolic acid (PLGA) offer considerable entrapment capacity and controlled biodegradability, making them ideal for local antibiotic delivery. PLGA-vancomycin systems have shown particular promise for treating osteomyelitis, with hollow particles providing flexibility in performance and application [94].
An innovative evidence-based design-of-experiments (DoE) approach has been developed to optimize such drug delivery systems without requiring new experiments. This methodology uses historical release data from literature to create regression models, analyzes factor interactions and correlations, and numerically optimizes factors by linking release data with the drug's therapeutic window. When applied to PLGA-VAN systems, this approach identified optimal parameters including molecular weight of PLGA, lactic acid to glycolic acid (LA/GA) molar ratio, polymer-to-drug mass ratio (P/D), and particle size [94].
DoE Optimization Workflow
Liposomal and Targeted Systems
Liposomal delivery systems provide stable formulation, improved pharmacokinetics, and passive targeting to tumor tissue through the "enhanced permeability and retention" effect. Liposomal anthracyclines achieve efficient drug encapsulation with reduced cardiotoxicity, while next-generation immunoliposomes enable molecular targeting of specific cells [95].
Advanced targeting strategies conjugate antibody fragments to liposomes, creating systems with efficient cellular binding and internalization. Anti-HER2 immunoliposomes loaded with doxorubicin displayed potent and selective activity against HER2-overexpressing tumors, with significantly superior efficacy compared to free doxorubicin, liposomal doxorubicin, or antibody-drug combinations [95].
Experimental Protocols and Methodologies
Surface Characterization Protocols
Comprehensive surface analysis requires multiple complementary techniques. Standard protocols include:
Scanning Electron Microscopy (SEM) for qualitative evaluation of micro- and nano-texture morphology. Typical parameters include high vacuum, 5 kV voltage, and 60 µA current intensity [92].
Energy-Dispersive X-ray Analysis (EDX) for semi-quantitative chemical and elemental composition analysis of titanium surfaces [91].
White Light Laser Profilometry (Interferometry) for accurate three-dimensional characterization of quantitative surface roughness, providing spatial parameters (Sₐ, Sds, Sdr) that offer more comprehensive data than 2-D measurements [91].
X-ray Diffraction (XRD) for phase transformation analysis, typically using Cu K-α radiation with wavelength of 0.15406 nm in the 2θ range of 10–80° with scanning step of 0.02° [92].
Antimicrobial Efficacy Testing
Standardized methodologies for evaluating antimicrobial activity include:
Zone of Inhibition (ZOI) Tests using agar diffusion to assess antimicrobial activity against various pathogens [93].
Biofilm Assay Protocols involving sample immersion in bacterial suspension with subsequent enumeration of adherent cells after specified periods (typically 24-48 hours) [93].
Minimum Inhibitory/Bactericidal Concentration (MIC/MBC) Determination through broth dilution methods to establish effective antibiotic concentrations [94].
Confocal Laser Scanning Microscopy (CLSM) with live/dead staining for real-time in situ imaging of bacterial viability on treated surfaces [93].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Antimicrobial Implant Research
| Category | Specific Materials | Research Applications | Key Functions |
|---|---|---|---|
| Metal Nanoparticles | Silver, Copper, Gold, Zinc oxide, Titanium oxide | Antimicrobial nanocoatings | ROS generation; ionic release; membrane disruption |
| Polymeric Carriers | PLGA, Chitosan, Gelatin, Hyaluronic acid | Drug delivery systems | Controlled biodegradability; drug encapsulation |
| Characterization Tools | SEM, EDX, XRD, White Light Profilometry | Surface analysis | Morphological, chemical, and topographic characterization |
| Biological Assays | Crystal Violet, MTT, Live/Dead staining, CLSM | Efficacy and biocompatibility testing | Biofilm quantification; cell viability assessment |
| Crosslinking Agents | Glutaraldehyde, Genipin, EDC/NHS | Hydrogel fabrication | Polymer network formation; stability enhancement |
Comparative Analysis and Future Perspectives
When comparing antimicrobial strategies, each approach demonstrates distinct advantages and limitations. Surface morphology modifications offer permanent physical antimicrobial properties without drug-related resistance concerns but may have limited potency against established biofilms. Nanocoatings provide potent broad-spectrum activity but raise potential toxicity concerns at higher concentrations. Advanced drug delivery systems enable precise temporal control over antimicrobial release but involve more complex fabrication processes.
Future developments will likely focus on multifunctional systems that combine physical and chemical antimicrobial approaches. The integration of growth factors with antimicrobial agents represents a promising direction, as seen in systems combining BMP-2 and VEGF delivery for simultaneous infection control and enhanced bone regeneration [87]. Smart responsive systems that release antimicrobials only in the presence of pathogens or specific environmental cues (pH, enzymes) also show considerable potential for minimizing resistance development while maximizing therapeutic efficacy.
The application of artificial intelligence and meta-analytic approaches to optimize delivery systems without extensive experimentation will accelerate development timelines and improve reproducibility. As regulatory frameworks evolve to better address nanomaterial safety concerns, these innovative antimicrobial strategies will increasingly translate from laboratory research to clinical practice, ultimately improving patient outcomes through reduced implant-associated infections and enhanced osseointegration.
Systematic Comparison of Dental Biomaterials: Performance and Clinical Implications
The long-term success of dental implants is fundamentally governed by their surface properties, which directly influence biological responses at the implant-bone interface. Surface morphology and chemical composition serve as critical determinants for osseointegration—the direct structural and functional connection between living bone and the implant surface [96]. Titanium and its alloys have represented the gold standard in implant dentistry for decades, leveraging their exceptional biocompatibility and mechanical properties [97]. However, zirconia implants have emerged as a promising metal-free alternative, offering superior aesthetics and unique biological advantages [98].
This scientific review provides a comprehensive surface-level comparison between these two biomaterials, analyzing how their distinct surface characteristics influence cellular behavior, bacterial response, and clinical outcomes. The analysis extends beyond macroscopic performance to molecular-level interactions, exploring how surface modifications manipulate the biological environment to enhance bone integration and prevent complications. For researchers and biomaterial scientists, understanding these surface-biology relationships is paramount for driving innovation in implant design and surface engineering strategies that push the boundaries of clinical success.
Material Properties and Surface Characteristics
The fundamental differences between titanium and zirconia extend beyond their metallic versus ceramic classification to their inherent surface properties and interactions with the biological environment.
Titanium implants depend on a spontaneously forming titanium dioxide (TiO₂) layer that provides corrosion resistance and biocompatibility [97]. This native oxide layer, however, is thin and can be compromised under mechanical loading, potentially leading to corrosion and ion release [97]. Titanium surfaces are naturally hydrophobic but can be modified to become superhydrophilic through various treatments, significantly enhancing their biological performance [99]. For instance, premium titanium implants like Straumann Roxolid SLActive and Nobel TiUltra demonstrate superior hydrophilicity with contact angles significantly below 90°, accelerating early bone healing [99].
Zirconia exists in multiple crystalline phases, with yttria-stabilized tetragonal zirconia polycrystals (Y-TZP) being the predominant form for dental implants due to its excellent mechanical properties and transformation toughening mechanism [98] [100]. This ceramic material is naturally bio-inert and exhibits minimal ion release in biological environments [1]. Unlike titanium, zirconia's tooth-like color provides inherent aesthetic advantages, particularly in the anterior region and for patients with thin gingival biotypes [101] [98]. However, zirconia surfaces are typically more hydrophobic than treated titanium surfaces, which may initially slow biological interactions [99].
Table 1: Fundamental Properties of Titanium and Zirconia Implant Materials
| Property | Titanium Implants | Zirconia Implants |
|---|---|---|
| Material Type | Metal (Commercially Pure or alloy) | Ceramic (Y-TZP) |
| Atomic/Crystal Structure | Hexagonal close-packed (α), Body-centered cubic (β) | Tetragonal (stabilized), Monoclinic, Cubic |
| Native Surface Layer | Titanium dioxide (TiO₂) [97] | Zirconium dioxide (ZrO₂) [1] |
| Base Surface Hydrophilicity | Hydrophobic (can be modified to superhydrophilic) [99] | Hydrophobic [99] |
| Aesthetic Characteristics | Grayish color, may show through thin gums [101] | Tooth-like white color, superior aesthetics [98] |
| Corrosion Behavior | Prone to electrochemical and pitting corrosion in biological fluids [97] | Excellent corrosion resistance, protective oxide layer [1] |
| * Bacterial Affinity* | Higher bacterial adhesion potential [101] | Low plaque affinity, reduced bacterial adhesion [98] |
Surface Modification Techniques and Experimental Outcomes
Surface modification technologies represent a rapidly advancing frontier in biomaterials research, aiming to overcome inherent material limitations by engineering the implant-tissue interface at micro- and nanoscales.
Titanium Surface Modification Strategies
Titanium surface modifications focus on enhancing osseointegration and imparting antibacterial properties through physical, chemical, and biological strategies:
Physical Modifications: Techniques including sandblasting, acid etching, laser etching, and plasma spraying primarily alter surface topography and roughness [96]. Sandblasting with large grits followed by acid etching creates micro-rough surfaces that enhance mechanical interlocking with bone [96]. Laser etching offers precision in creating controlled microgroove patterns that guide cellular response [96].
Chemical Modifications: Methods such as anodic oxidation, alkali treatment, and acid etching alter surface chemistry and oxide layer properties [96]. Anodic oxidation can create nanotubular structures that significantly increase surface area for bone attachment [97].
Biological Modifications: These involve applying bioactive coatings to the titanium surface. Hydroxyapatite coatings provide osteoconductive properties, while ion doping with zinc, magnesium, or copper enhances both osteogenesis and antibacterial efficacy [102]. Zinc-doped coatings have demonstrated 25% increased osteoblast proliferation and 40% enhanced cell adhesion [102].
Zirconia Surface Modification Strategies
Zirconia surface modifications address its biological inertness and hydrophobic nature:
Physical Modifications: Sandblasting with alumina particles increases surface roughness but may introduce micro-cracks that compromise mechanical integrity [100]. Laser treatment creates precise micro-patterns without structural damage, enhancing osseointegration and reducing biofilm formation [100].
Chemical Modifications: Acid etching with hydrofluoric acid creates micro-retentive surfaces and incorporates fluoride ions that enhance osteoblastic differentiation [100]. Studies report bone-implant contact of approximately 81% with HF-etched zirconia [100].
Physicochemical Modifications: Ultraviolet light treatment effectively removes surface hydrocarbons, creating a super-hydrophilic surface that promotes protein adsorption and osteoblast attachment [100]. UV-treated zirconia demonstrates significantly higher bone-implant contact and removal torque values [100].
Table 2: Comparative Analysis of Surface Modification Techniques and Outcomes
| Modification Type | Technical Approach | Key Experimental Findings | Biological Impact |
|---|---|---|---|
| Titanium: Ion Doping | Incorporation of Zn, Mg, Cu, or Sr ions into surface layer | • Zn: 25% ↑ osteoblast proliferation, 40% ↑ cell adhesion [102]• Cu: 99.45% antibacterial efficacy against S. aureus [102] | Enhanced osseogenesis and antibacterial activity |
| Titanium: Nanotubular Structures | Anodic oxidation to create TiO₂ nanotubes | Increased osteoblastic activity and gene expression for bone formation [97] | Mimics natural cellular environment, promotes rapid bone growth |
| Zirconia: UV Light Treatment | UV exposure to remove surface hydrocarbons | Creates super-hydrophilic surface, significantly higher BIC and RTQ values [100] | Enhanced protein adsorption, cell adhesion, and bone formation |
| Zirconia: Laser Etching | Precision surface patterning with laser systems | Microgroove patterns promote cell adhesion and orientation [100] | Guides cellular response, reduces biofilm formation |
| Both: Sandblasting & Acid Etching | Sequential abrasive and chemical etching | Significantly increases surface roughness and RTQ values [96] [100] | Enhanced mechanical interlocking with bone tissue |
Quantitative Performance Comparison
Rigorous evaluation of implant performance requires assessment across multiple parameters, from cellular interactions to long-term clinical outcomes.
Biological and Clinical Performance Metrics
Table 3: Performance Metrics of Titanium vs. Zirconia Implants
| Performance Parameter | Titanium Implants | Zirconia Implants | Research Context |
|---|---|---|---|
| 10-Year Survival Rate | 94-98% [101] | 94-98% [101] | Clinical studies |
| Hydrophilicity (Contact Angle) | As low as 0° (superhydrophilic) for SLActive [99] | Generally higher, material-dependent [99] | In vitro measurement |
| Bone-to-Implant Contact (BIC) | ~53% in submerged healing [1] | ~53% in submerged healing [1] | Minipig model, 8 weeks |
| Removal Torque Values | High, material- and surface-dependent | Significantly higher for modified vs. machined surfaces [100] | Animal studies |
| Fracture Resistance | High, excellent long-term data | Lower BMF vs. titanium, concerns in high-load areas [98] | In vitro mechanical testing |
| Bacterial Adhesion | Higher affinity for plaque accumulation [101] | Low bacterial affinity, reduces peri-implantitis risk [98] | In vitro and clinical studies |
| Aesthetic Performance | Risk of gray show-through in thin gingiva [101] | Superior, natural tooth color [103] | Clinical assessment in anterior region |
Advanced Surface Characterization Techniques
Surface analysis employs sophisticated methodologies to quantify implant-topography interactions:
Contact Angle Measurement: Quantifies surface hydrophilicity/hydrophobicity using the sessile drop method, where a smaller contact angle indicates greater hydrophilicity [99]. Premium titanium implants demonstrate contact angles approaching 0° (superhydrophilic), while zirconia typically shows higher contact angles [99].
Surface Roughness Analysis: Optical profilometry measures arithmetic mean height (Sa) over a specified area, typically using 640,000 data points across a 10.24 mm² region for statistical significance [104]. Measurements are performed on multiple specimens (n≥3) to ensure reliability.
Removal Torque Analysis: Evaluates bone-implant integration strength in animal models by measuring the torque required to remove osseointegrated implants, with higher values indicating stronger integration [100].
Bone-Implant Contact Quantification: Histomorphometric analysis of undecalcified stained sections under light microscopy to calculate the percentage of implant surface in direct contact with mineralized bone [1].
The following diagram illustrates the methodological workflow for comprehensive implant surface characterization:
Surface Characterization Workflow: Comprehensive methodology for analyzing implant surface properties and biological performance.
The Scientist's Toolkit: Essential Research Reagents and Materials
Advancing implant surface research requires specific reagents, equipment, and methodologies. The following toolkit outlines critical resources for conducting rigorous surface science investigations in dental biomaterials.
Table 4: Essential Research Toolkit for Implant Surface Studies
| Category | Specific Reagents/Equipment | Research Application | Function in Experimental Protocols |
|---|---|---|---|
| Surface Modification | Alumina particles (110 μm), Hydrofluoric acid, UV light source [100] | Surface roughening, etching, and functionalization | Creates micro-rough surfaces, enhances surface energy and hydrophilicity |
| Coating Materials | Hyaluronic acid, Chitosan, Polyacrylic acid, Polyallylamine hydrochloride [104] | Polyelectrolyte multilayer coatings | Enhances surface biocompatibility, protein adsorption, and cell adhesion |
| Characterization Tools | Optical Profilometer, Scanning Electron Microscope, Contact Angle Goniometer [99] [104] | Surface topography and wettability analysis | Quantifies surface roughness, morphology, and hydrophilicity |
| Biological Assays | Lactate dehydrogenase assay, Bradford protein assay [104] | Cell adhesion and protein adsorption quantification | Measures osteoblast adhesion and human serum albumin adsorption |
| Test Organisms/Cells | Normal Human Osteoblasts (NHOst), S. aureus, E. coli [102] [104] | In vitro biocompatibility and antibacterial testing | Evaluates cellular response and bacterial adhesion on modified surfaces |
This surface-level analysis demonstrates that both titanium and zirconia implants offer distinct advantages rooted in their material-specific surface properties. Titanium provides superior hydrophilicity and adaptability through extensive surface modification options, including ion doping that enhances both osteogenesis (25-40% increase in cell proliferation/adhesion with Zn doping) and antibacterial efficacy (up to 99.45% against S. aureus with Cu doping) [102]. Zirconia delivers inherent aesthetic superiority and reduced bacterial affinity while offering excellent biocompatibility, though its surface modification landscape is less developed [98] [100].
The research trajectory points toward increasingly sophisticated multi-functional surface engineering that combines physical, chemical, and biological strategies. Future developments will likely focus on smart coatings with controlled release kinetics, personalized surface topographies optimized for specific patient populations, and hybrid material systems that combine the advantages of both titanium and zirconia. The scientific community faces the challenge of translating promising in vitro findings into predictable clinical outcomes through more sophisticated animal models and well-controlled long-term clinical trials.
For researchers, the key lies in deepening our understanding of the molecular-level interactions between implant surfaces and biological systems, enabling the rational design of next-generation implants with enhanced osseointegration capacity and reduced complication profiles.
Comparative Analysis of Biofilm Formation Across Different Material Types
The formation of microbial biofilms on material surfaces is a critical concern across medical, industrial, and environmental domains. Biofilms are structured communities of microbial cells enclosed in a self-produced matrix of extracellular polymeric substances (EPS) that adhere to biotic or abiotic surfaces [105]. The material composition and surface characteristics of substrates significantly influence microbial adhesion and subsequent biofilm development, affecting outcomes ranging from medical device-related infections to the performance of dental restorations [106] [107]. This comparative analysis examines biofilm formation across diverse material types, synthesizing experimental data on how surface properties and chemical composition affect microbial colonization, with particular relevance to dental materials research. Understanding these relationships enables the development of advanced materials with enhanced resistance to biofilm formation, potentially reducing device-related infections and improving clinical outcomes.
Theoretical Framework: Biofilm Development and Material Interactions
Biofilm Formation Process
Biofilm formation is a multi-stage process that begins with the reversible attachment of planktonic microorganisms to surfaces, followed by irreversible adhesion, microcolony formation, maturation into complex three-dimensional structures, and eventual dispersal of cells to new sites [108] [105]. The initial attachment phase is governed by physical forces including van der Waals forces, electrostatic interactions, and hydrophobic effects, while subsequent stages involve complex genetic regulation and cell-to-cell communication through quorum sensing [105].
The extracellular polymeric substance matrix, comprising exopolysaccharides, proteins, extracellular DNA, and lipids, provides structural stability and protection to embedded microorganisms [109]. This matrix facilitates nutrient trapping, waste product removal, and intercellular communication while acting as a barrier against antimicrobial agents and host immune responses [109] [105].
Material Properties Influencing Biofilm Formation
Surface Roughness: Microscopic irregularities on material surfaces provide increased surface area and protective niches that enhance microbial attachment and retention. Surface roughness is typically quantified by the Ra value (arithmetic average of surface deviations) [107]. Studies suggest a threshold Ra value of approximately 0.2 μm, below which bacterial attachment is considered clinically negligible [107].
Surface Chemistry and Hydrophobicity: Material composition affects surface energy and hydrophobicity, which significantly influence initial microbial adhesion. Hydrophobic surfaces generally promote greater bacterial attachment than hydrophilic surfaces due to reduced repulsion between microbial cells and the substrate [106] [105]. The surface charge of materials also plays a crucial role, with positively charged surfaces typically attracting more microorganisms due to the generally negative charge of bacterial cells [106].
Environmental Factors: Conditions such as temperature, pH, oxygen availability, and nutrient concentration modulate biofilm development on material surfaces [106]. In dental applications, frequent exposure to dietary compounds and pH fluctuations further complicate these interactions [107].
Figure 1: Biofilm formation process on material surfaces, highlighting critical stages where material properties influence microbial attachment and development.
Experimental Approaches for Assessing Biofilm Formation
Standardized Methodologies for Biofilm Quantification
Static Models: The 96-well microtiter plate assay represents the most widely utilized static model for biofilm assessment. In this method, planktonic cultures with standardized concentrations are added to microtiter plates and incubated to allow adhesion to polystyrene surfaces. Non-adherent microorganisms are removed through rinsing, leaving mature biofilms for analysis [106]. Crystal violet staining is commonly employed to quantify total biofilm biomass by staining both bacterial cells and polysaccharides within the extracellular matrix [106]. While this approach offers high throughput and reproducibility, it may not accurately simulate fluid dynamics present in natural environments.
Dynamic Flow Models: Flow-cell systems provide more physiologically relevant conditions by simulating shear forces and nutrient flow encountered in natural environments. These systems consist of polystyrene chambers mounted on microscope slides, connected via tubing to medium reservoirs and waste containers, with peristaltic pumps regulating fluid flow [106]. Specific devices include chemostats, drip flow reactors, rotating biofilm reactors, constant-depth film fermenters, and the modified Robbins device [106]. The Calgary Biofilm Device (CBD) represents a particularly advanced flow model that enables high-throughput determination of minimum inhibitory concentrations (MIC) against biofilms [106].
Advanced Microfluidic Approaches: Recent technological advances have introduced 3D-printed microfluidic devices for dynamic biofilm studies. These "H-type" structured devices feature dual input channels that enable cultivation of polymicrobial biofilms and testing of anti-biofilm substances [108]. With an estimated cost of approximately €0.1285 per device, this approach offers an accessible alternative to conventional methods while providing superior simulation of in vivo conditions compared to static models [108].
Analytical Techniques for Biofilm Characterization
Microscopy Methods: Scanning electron microscopy (SEM) provides high-resolution visualization of biofilm morphology and spatial distribution on material surfaces [108] [107]. Optical coherence tomography (OCT) enables non-destructive, real-time monitoring of biofilm development and three-dimensional structure [108].
Spectroscopic Analysis: Fourier Transform Infrared (FTIR) spectroscopy identifies chemical functional groups and compositional elements within the biofilm matrix, providing insights into EPS composition and material-biofilm interactions [108].
Surface Analysis: Mechanical profilometers and atomic force microscopy (AFM) quantitatively assess surface roughness parameters (Ra values) that influence microbial adhesion [86] [107]. These instruments measure topographical variations at micro- and nanoscales, correlating surface features with biofilm formation potential.
Figure 2: Comprehensive workflow for assessing biofilm formation on material surfaces, integrating multiple analytical approaches for quantitative and qualitative characterization.
Comparative Analysis of Biofilm Formation on Dental Materials
Surface Roughness and Bacterial Adhesion Across Restorative Materials
Table 1: Surface Roughness and Bacterial Adhesion of Tooth-Colored Restorative Materials After Exposure to Food-Simulating Liquids
| Material Type | Representative Product | Mean Surface Roughness (Ra, μm) | Bacterial Adhesion (CFU/mL × 10⁵) | Key Findings |
|---|---|---|---|---|
| Alkasite | Cention N | 0.92 ± 0.11 | 8.7 ± 1.2 | Significantly higher roughness and bacterial adhesion after citric acid exposure [107] |
| Giomer | Beautifil II | 0.85 ± 0.09 | 7.9 ± 1.1 | High surface roughness promoting substantial S. mutans adhesion [107] |
| Ormocer | Admira Fusion | 0.48 ± 0.07 | 4.3 ± 0.8 | Moderate resistance to biofilm formation [107] |
| Direct Composite | G-ænial A'Chord | 0.39 ± 0.05 | 3.8 ± 0.6 | Lower bacterial adhesion compared to giomer and alkasite [107] |
| Indirect Composite | Gradia Plus | 0.31 ± 0.04 | 2.9 ± 0.5 | Smoothest surface with minimal bacterial retention [107] |
Experimental protocol: 220 disc-shaped specimens (8mm diameter × 2mm depth) were prepared from five restorative materials (n=44 per material). Each group was divided into four subgroups (n=11) and immersed in food-simulating liquids (heptane, ethanol, 10% citric acid, or artificial saliva) for 7 days. Surface roughness was measured using mechanical profilometry before and after immersion. Bacterial adhesion was assessed using Streptococcus mutans with colony-forming unit enumeration. One specimen per subgroup was analyzed via scanning electron microscopy for qualitative assessment [107].
Time-Dependent Biofilm Formation on Orthodontic Aligners
Table 2: Comparative Biofilm Formation on Clear Aligner Materials Over Time
| Aligner Material | 24h Biofilm Formation | 168h Biofilm Formation | Statistical Significance | Key Comparisons |
|---|---|---|---|---|
| Smartee | Low | Low | Reference | Demonstrated lowest overall biofilm formation [110] |
| ClearCorrect | Moderate | High | p < 0.05 vs Smartee at 120, 168, 240h | Significantly higher bacterial accumulation [110] |
| Graphy | Moderate | High | p < 0.05 vs Smartee at 168h | Enhanced S. mutans + L. acidophilus biofilm at 120-168h [110] |
| Invisalign | Low | Moderate | p < 0.05 vs Graphy at 120-168h | Lower biofilm formation than Graphy for mixed species [110] |
Experimental protocol: Six different clear aligner systems were evaluated for biofilm formation by Streptococcus mutans ATCC 25175 and Lactobacillus acidophilus ATCC 4356 at multiple time points (0, 24, 48, 72, 96, 120, 168, and 240 hours). Biofilm formation was quantified using standardized microbiological methods, with statistical analysis performed using two-way repeated measures ANOVA and post-hoc Bonferroni T2 tests. Significance was set at p < 0.05 [110].
Surface Modification and Bacterial Adhesion on Zirconia
Table 3: Impact of Surface Treatments on Bacterial Adhesion to Zirconia Materials
| Surface Treatment | Surface Roughness Change | Bacterial Adhesion Reduction | Key Mechanisms |
|---|---|---|---|
| As-sintered Zirconia | Reference | Reference | Baseline characteristics [83] |
| Airborne-Particle Abrasion | Significant increase | 70% increase vs as-sintered | Roughness-induced adhesion enhancement [83] |
| PEM Coating (PAA-terminated) | Minimal change | 50% reduction vs abraded | Negative surface charge repelling bacteria [83] |
| PEM Coating (Chitosan-based) | Minimal change | 70% reduction vs abraded | Hydrophobic modification and charge effects [83] |
Experimental protocol: Zirconia specimens with yttria content between 3-5 mol% were isostatically pressed into disks and sintered at 1450°C for 2 hours. Airborne-particle abrasion was applied to selected specimens, followed by polyelectrolyte multilayer coating with chitosan and poly(acrylic acid). Surface characterization included profilometry, atomic force microscopy, tensiometry, and electrokinetic analysis. Bacterial adhesion of Streptococcus mutans was quantified using spectrophotometry and scanning electron microscopy [83].
The Researcher's Toolkit: Essential Reagents and Materials
Table 4: Essential Research Reagents and Materials for Biofilm Studies
| Category | Specific Items | Function/Application | Experimental Context |
|---|---|---|---|
| Microbiological Culture | Columbia agar + 5% sheep blood, Mueller Hinton broth, MacConkey agar, Mannitol salt agar | Bacterial cultivation and standardization | Used for preparing bacterial inoculum (e.g., McFarland standard) for adhesion assays [108] [111] |
| Biofilm Quantification | Crystal violet (1%), Microtiter plates, Spectrophotometer | Total biofilm biomass assessment | Stains bacterial cells and EPS; absorbance measured at 590nm [108] [106] |
| Surface Characterization | Mechanical profilometer, Atomic force microscope, Scanning electron microscope | Surface roughness and topography analysis | Quantifies Ra values and visualizes surface-biofilm interactions [86] [107] [83] |
| Food-Simulating Liquids | 10% Citric acid, Ethanol absolute, n-Heptane, Artificial saliva | Simulating oral environment challenges | Testing material degradation and biofilm formation under clinically relevant conditions [107] |
| Microfluidic Systems | 3D-printed H-type devices, Syringe pumps, Polylactic acid filament | Dynamic biofilm cultivation | Enables flow-condition studies with controlled shear forces [108] |
Discussion: Implications for Material Selection and Design
The comparative analysis of biofilm formation across material types reveals several significant patterns with important implications for clinical practice and material development.
Material Composition and Surface Properties
The relationship between surface roughness and bacterial adhesion demonstrates a clear correlation across all material categories. Materials with Ra values exceeding 0.8 μm, such as alkasites and giomers, supported substantially greater biofilm formation compared to smoother surfaces like indirect composites (Ra ≈ 0.31 μm) [107]. This finding aligns with the established threshold of 0.2 μm, below which bacterial attachment becomes clinically negligible [107]. Beyond roughness, surface chemistry significantly influences microbial adhesion, as evidenced by the effectiveness of polyelectrolyte multilayer coatings in reducing bacterial attachment to zirconia by up to 70% despite minimal changes in surface topography [83].
Methodological Considerations in Biofilm Research
The comparison between static and dynamic biofilm models reveals substantial differences in biofilm architecture and development kinetics. Flow-condition models demonstrate accelerated biofilm formation, with mature structures developing within 18-24 hours compared to 48-72 hours in static systems [112]. Microfluidic approaches better simulate in vivo conditions by incorporating shear forces that influence biofilm morphology and gene expression [108] [112]. These methodological differences highlight the importance of selecting appropriate experimental models that reflect the intended application environment of the material being tested.
Clinical Implications and Future Directions
The substantial variation in biofilm susceptibility among different material classes underscores the importance of evidence-based material selection in clinical applications. In dental practice, indirect composites demonstrate superior performance regarding biofilm resistance, while alkasites and giomers, despite their therapeutic ion release capabilities, may require additional surface modifications to mitigate microbial adhesion [107]. Future material development should focus on optimizing both surface topography and chemical composition to disrupt microbial attachment mechanisms without compromising mechanical properties or biocompatibility. Emerging strategies include surface charge modification, antimicrobial functionalization, and smart materials that respond to microbial challenges.
The advent of digital manufacturing has revolutionized dental materials research, presenting clinicians and researchers with two dominant methodologies: subtractive manufacturing (milling) and additive manufacturing (3D printing). This comparison guide objectively evaluates the efficacy of these techniques based on mechanical properties, accuracy, surface characteristics, and biochemical performance. Mounting evidence from systematic reviews and controlled in vitro studies indicates that milling consistently delivers superior mechanical strength and surface finish for definitive restorations, particularly in high-stress applications. In contrast, 3D printing demonstrates exceptional capabilities in material efficiency, complex geometry fabrication, and marginal adaptation for interim restorations. This analysis synthesizes current experimental data to provide researchers and dental professionals with evidence-based protocols and insights for selecting appropriate manufacturing techniques based on specific clinical and research requirements.
Digital workflows in restorative dentistry and dental materials research rely primarily on two computer-aided manufacturing (CAM) techniques: subtractive (milling) and additive (3D printing) manufacturing. The fundamental distinction lies in their production methodologies. Milling is a subtractive process that carves restorations from pre-fabricated solid blocks of materials like polymethyl methacrylate (PMMA) or zirconia, achieving high precision through material removal [113] [114]. Conversely, 3D printing is an additive process that builds components layer-by-layer from liquid resins or powders, enabling unparalleled geometric freedom and minimal material waste [113] [114].
The selection between these techniques significantly impacts the ultimate cost, development cycle, and functional performance of dental devices and research models. For researchers investigating surface morphology, material properties, or long-term durability, understanding the inherent strengths and limitations of each manufacturing method is paramount. This guide provides a comprehensive, data-driven comparison to inform experimental design and material selection in dental materials science.
Comparative Analysis of Mechanical Properties and Durability
Mechanical performance is a critical determinant for the clinical success of dental restorations and the validity of in vitro research models. Extensive comparative analyses reveal significant differences between milled and 3D-printed components.
Key Experimental Findings on Strength and Durability
A systematic review and network meta-analysis published in Dental Materials (2025) analyzing 63 in vitro studies concluded that milled PMMA denture bases demonstrated significantly superior mechanical properties across multiple outcomes, including flexural strength, flexural modulus, and surface roughness, compared to 3D-printed counterparts [115] [116]. The review consistently ranked milling first or second across all measured mechanical properties, while 3D-printed bases demonstrated the lowest overall mechanical performance [115].
A controlled chewing simulation study (2025) evaluated the wear of interim resin materials, providing quantitative data on volumetric loss and wear depth after 30,000 cycles (approximately 1.5 months of clinical function) [117]. The results indicated no statistically significant differences in wear volume loss between manufacturing techniques, suggesting comparable short-term wear resistance [117].
Table 1: Mechanical Properties and Wear Resistance Comparison
| Property | Milling Technique Performance | 3D Printing Technique Performance | Research Implications |
|---|---|---|---|
| Flexural Strength | Superior [115] [116] | Lower [115] [116] | Milling preferred for high-stress, long-term definitive restorations and fatigue testing models. |
| Surface Hardness | Superior (Vickers Hardness) [115] [116] | Lower [115] [116] | Milled parts better resist indentation in biomimetic testing scenarios. |
| Impact Strength | Superior [115] [116] | Lower [115] [116] | Milling recommended for removable prostheses research where drop-impact resistance is relevant. |
| Wear Volume Loss | 0.1099 ± 0.0873 mm³ [117] | 0.0934 ± 0.0788 mm³ to 0.0980 ± 0.1021 mm³ [117] | Both techniques show statistically comparable wear resistance in interim applications. |
| Material Isotropy | Isotropic (uniform in all directions) [113] | Anisotropic (weak interlayer adhesion) [113] | Milled parts provide predictable mechanical behavior; printed parts show direction-dependent properties. |
Experimental Protocol: Chewing Simulation for Wear Analysis
The chewing simulation study provides a validated methodology for evaluating the durability of dental materials [117]:
- Sample Preparation: Specimens are fabricated (n=8 per group) and divided into four groups: (1) LCD 3D-printed, (2) DLP 3D-printed, (3) conventional autopolymerizing bis-acrylic, and (4) CAD/CAM milled interim resin materials.
- Dynamic Loading: Specimens undergo 30,000 cycles in a chewing simulator, applying physiological occlusal forces at a specified frequency and angle.
- Wear Measurement: Volumetric loss (mm³) and wear depth (mm) are calculated using high-resolution 3D scanning or profilometry before and after cycling.
- Statistical Analysis: Non-parametric tests (e.g., Kruskal-Wallis) are used to identify intergroup differences (α=0.05).
Accuracy, Trueness, and Fitness in Restorative Applications
Accuracy encompasses both trueness (deviation from original design) and fitness (marginal and internal adaptation). These parameters directly influence the clinical longevity of restorations and the precision of research prototypes.
Controlled Comparison of Trueness and Fitness
A pivotal 2025 in vitro study eliminated material composition as a confounding variable by using the same printable hybrid ceramic resin for both 3D-printed crowns and milled blocks [118]. This design allowed for a direct comparison of the manufacturing techniques' inherent capabilities.
The findings revealed a significant trade-off:
- Milling produced restorations with significantly higher trueness (closer to the digital design) [118].
- 3D Printing resulted in restorations with superior internal and marginal adaptation (better fit) [118].
This inverse relationship highlights that a more precise fabrication process (milling) does not automatically guarantee a better-fitting restoration, a crucial consideration for research on cement thickness, microleakage, and periodontal health.
Table 2: Accuracy and Fitness Metrics in Fixed Prosthodontics
| Parameter | Milling Technique Performance | 3D Printing Technique Performance | Clinical/Research Significance |
|---|---|---|---|
| Trueness | Higher [118] | Lower [118] | Milling better replicates the exact digital design geometry. |
| Marginal Gap | Zirconia: 123.89 ± 56.89 µm [114] | Interim Crowns: 123.87 ± 67.42 µm [114] | Both techniques can achieve clinically acceptable marginal gaps (<120 µm). |
| Internal Adaptation | Lower than 3D printing [118] | Superior to milling [118] | 3D printing creates better internal fit, potentially reducing cement space. |
| Geometric Complexity | Limited by bur size and axis [113] [114] | Excellent for complex geometries [113] [114] | 3D printing is superior for research on intricate structures, hollow features. |
| Material Waste | High (>30-40% waste) [114] | Low (<5% waste) [114] | 3D printing is more material-efficient, reducing costs for prototype iterations. |
Experimental Protocol: Trueness and Fitness Measurement
The methodology for assessing accuracy is critical for reproducible research [118]:
- Digital Workflow: Tooth preparations or master models are scanned using an intraoral or laboratory scanner. Crowns are designed with standardized parameters (e.g., 0.1 mm cement space, 1 mm minimum thickness) and exported as STL files.
- Fabrication: Crowns are manufactured via both milling and 3D printing from the same base material.
- Trueness Measurement: Fabricated crowns are 3D-scanned. The scan data is aligned with the original reference design (STL file) in metrology software (e.g., Geomagic Control X). Trueness is quantified using root mean square (RMS) error or color-difference maps.
- Fitness Measurement: The restoration is seated on the die. The marginal and internal gaps are measured at multiple predetermined points using silicone replica techniques or direct cross-sectional analysis.
- Statistical Analysis: Data are analyzed using appropriate tests (e.g., one-way ANOVA, Kruskal-Wallis, Tukey's HSD).
Surface Morphology and Chemical Composition Analysis
Surface characteristics and chemical stability are critical for bacterial adhesion, aesthetic retention, and biocompatibility—key research areas in dental materials science.
Surface Roughness and Stainability
Surface roughness (SR) is a primary factor influencing bacterial colonization and stain accumulation. Research indicates that milled components typically exhibit superior initial surface smoothness [115] [116]. For 3D-printed resins, surface quality is highly dependent on process parameters. A 2025 study on denture base resin found that printing technology and orientation significantly influence outcomes [119]. LCD printers produced lower roughness than DLP at a 45-degree angle, and immersion in coffee for three months led to clinically unacceptable color changes, particularly in DLP-printed specimens [119].
Advanced machine learning (ML) models are now being deployed to predict and optimize the surface roughness of 3D-printed dental appliances. A 2025 study utilized ensemble ML techniques, finding that XGBoost algorithms (R²=0.99858, RMSE=0.00346998) outperformed other models in predicting SR based on printing parameters [120].
Chemical Composition and Biocompatibility
The chemical composition of 3D-printing resins and the potential leaching of unreacted monomers raise important biocompatibility questions. A 2025 materials chemistry study used ultra-high-performance liquid chromatography–high-resolution mass spectrometry (UHPLC–HRMS) to characterize liquid resins [121]. Targeted analysis detected leaching of methacrylate monomers (e.g., BisEMA, UDMA) into artificial saliva, with concentrations peaking after 24 hours of incubation [121]. Untargeted analysis further revealed that at least 67 putative chemical compounds leached from the tested resins over time, highlighting a more complex elution profile than previously understood [121]. This leaching dynamics have direct implications for research on oral inflammatory responses and material cytotoxicity.
Research Reagent Solutions and Essential Materials
The following table details key materials and reagents used in the cited experimental research, providing a reference for protocol development.
Table 3: Research Reagent Solutions for Dental Manufacturing Studies
| Reagent/Material | Composition/Type | Research Function & Application |
|---|---|---|
| PMMA Denture Base Resin | Polymethyl Methacrylate (pre-polymerized block) | Gold-standard material for subtractive manufacturing; control group for mechanical property testing (flexural strength, modulus) [115] [116]. |
| Printable Hybrid Ceramics | e.g., Ceramic Crown (SprintRay), Varseosmile Crown plus (Bego) | Resin-based composites with ceramic fillers; used for comparative studies on the accuracy and mechanical properties of permanent restorations [118]. |
| Artificial Saliva | Standardized inorganic and organic compound solution | Simulates the oral environment for aging studies, monomer leaching analyses, and investigations of chemical degradation [121]. |
| Methacrylate Monomer Standards | e.g., BisEMA, UDMA, TEGDMA | High-purity chemical standards used for quantitative analysis (LC-MS/MS) to identify and quantify leachable compounds from polymerized resins [121]. |
| Acetonitrile / Methanol (HPLC Grade) | High-purity organic solvents | Mobile phase components in chromatographic separation (UHPLC, LC-MS/MS) for identifying and quantifying leachables [121]. |
Visualizing the Research Workflow and Technical Differences
The following diagrams illustrate the core conceptual and experimental workflows discussed in this guide.
Fundamental Manufacturing Principles
Integrated Research Methodology for Dental Device Evaluation
The comparative analysis of milling and 3D printing reveals a clear paradigm: no single manufacturing technique is universally superior. The optimal choice is profoundly application-dependent, dictated by specific research objectives and clinical requirements.
Milling remains the benchmark for applications demanding high mechanical strength, superior surface finish, and isotropic material properties, making it ideal for definitive restorations and functional testing prototypes [115] [116]. 3D printing excels in geometric complexity, material efficiency, and rapid iteration, offering significant advantages for creating anatomically complex models, custom research jigs, and devices with internal features unattainable through subtractive methods [113] [114].
Future research should prioritize the development of hybrid workflows that leverage the strengths of both technologies, advanced material formulations for 3D printing that close the mechanical property gap, and the integration of AI-driven optimization for process parameter control. Furthermore, long-term clinical validation and standardized testing protocols are essential to translate in vitro findings into predictable clinical outcomes and robust research models.
In restorative dentistry, the surface of a dental material is the critical interface that determines its long-term performance, biological integration, and clinical success. Surface modifications—ranging from traditional glazing techniques to advanced bioactive coatings and nanotechnology enhancements—represent powerful strategies to control how materials interact with the biological environment. The validation of these modifications requires sophisticated analytical techniques to assess their effects on surface morphology, chemical composition, and functional performance. This comparison guide examines the current landscape of surface modification technologies, providing researchers and dental scientists with experimental data and methodological frameworks for objective evaluation. By comparing conventional approaches with emerging nanotechnological and biomimetic strategies, this analysis aims to establish a rigorous foundation for assessing the efficacy of surface modifications across multiple parameters, including roughness, hardness, bacterial adhesion, and biological integration.
The significance of surface properties extends beyond mechanical performance to encompass fundamental biological responses. Rough surfaces can promote bacterial adhesion and biofilm formation, while improperly modified surfaces may demonstrate inadequate tissue integration or increased susceptibility to corrosion. Similarly, the chemical composition of surface layers directly influences cellular responses, protein adsorption, and ultimately, the biocompatibility of the material. As dental materials science advances, validation methodologies must evolve to characterize these complex surface interactions at multiple scales—from macroscopic topographic features to nanoscale molecular arrangements.
Comparative Analysis of Surface Modification Technologies
Table 1: Performance Comparison of Major Surface Modification Approaches for Dental Materials
| Modification Type | Key Materials/Agents | Surface Roughness (Ra, µm) | Hardness/Hydrophilicity | Bacterial Adhesion Impact | Key Functional Outcomes |
|---|---|---|---|---|---|
| Traditional Glazing | Vita AKZENT glaze powder/liquid, paste, spray [122] | 0.50 ± 0.26 µm (powder/liquid) [122] | No significant hardness changes post-corrosion (p > 0.05) [122] | Indirectly reduced via smoother surfaces | Powder/liquid method created smoothest surface, particularly after corrosion [122] |
| CAD/CAM Ceramics (Unmodified) | Zirconia-reinforced lithium silicate, Feldspathic ceramic, Lithium disilicate [76] | 1.97 µm (zirconia-reinforced), 1.91 µm (feldspathic), 2.86 µm (lithium disilicate) [76] | Contact angles: 42.48° (zirconia-reinforced), 38.31° (feldspathic), 31.41° (lithium disilicate) [76] | Varies with roughness and chemistry | Zirconia-reinforced lithium silicate showed favorable topography and wettability combination [76] |
| Polyelectrolyte Multilayer Coatings | Chitosan, poly(acrylic acid) [83] | Application on abraded zirconia (increased roughness) [83] | Negatively charged terminating layer [83] | 50-70% reduction in Streptococcus mutans adhesion [83] | Biocompatible coatings effective against biofilm development [83] |
| Biomimetic Antifouling Coatings | Shark skin-inspired patterns, lotus leaf-inspired superhydrophobic surfaces [123] | Micro/nanoscale topological patterning [123] | Extreme hydrophobicity or contact-based bactericidal effects [123] | Significant reduction via topological disruption of bacterial membranes [123] | Nature-inspired physical antifouling without chemical agents [123] |
| Nanotechnology Enhancements | Nano-hydroxyapatite, carbon nanotubes, metal oxide nanoparticles [124] | Can be tailored at nanoscale | Enhanced mechanical properties, bioactive surfaces | Antibacterial properties of metal oxide nanoparticles (e.g., ZnO, TiO₂) [124] | Multi-functionalization: drug delivery, antimicrobial, improved mechanical properties [124] |
Table 2: Analytical Techniques for Surface Modification Validation
| Validation Technique | Key Parameters Measured | Resolution/Sensitivity | Applications in Dental Research |
|---|---|---|---|
| Profilometry | Surface roughness (Ra, Rq, Rsk), skewness [76] | Sub-micron vertical resolution | Quantitative roughness assessment after polishing, glazing, or abrasion [122] [76] |
| Scanning Electron Microscopy (SEM) | Surface morphology, microstructural features, coating integrity [39] | Nanoscale resolution with high-depth field | Qualitative analysis of surface alterations, coating homogeneity, wear patterns [39] |
| Colorimetric Spectrophotometry | Mineral content (calcium, phosphorus) [39] | µg/ml concentration detection | Quantifying mineral loss/gain after bleaching, erosion, or remineralization [39] |
| Contact Angle Measurements | Wettability, surface free energy, hydrophilicity/hydrophobicity [76] | Degree resolution with liquid droplets | Assessing biofilm propensity, tissue integration potential, coating performance [76] |
| Energy-Dispersive X-ray Spectroscopy (EDS) | Elemental composition, coating chemistry [76] | Elemental detection limits ~0.1% | Verifying coating composition, detecting surface contaminants [76] |
Experimental Protocols for Surface Modification Validation
Surface Roughness and Hardness Assessment of Glazed Zirconia
Objective: To evaluate the effects of different glazing methods on the surface roughness and hardness of monolithic translucent zirconia ceramics [122].
Materials: Highly translucent, pre-shaded zirconia samples (XTCERA SHT, Shenzhen Xiangtong Co., Ltd., China) with shade A3, dimensioned at 12 × 10 × 2 mm; Vita AKZENT plus glaze materials (powder/liquid, paste, and spray); 4% acetic acid for corrosion simulation; Vickers diamond indenter for hardness testing; non-contact optical profilometer (U500x Digital Microscope) for roughness measurements [122].
Methodology:
- Sample Preparation: Thirty zirconia samples were divided into three groups (n=10) based on glazing method: Group 1 (powder and liquid), Group 2 (paste), and Group 3 (spray) [122].
- Glazing Application: Each group underwent their respective glazing procedures according to manufacturer instructions, followed by thermal processing.
- Corrosion Simulation: Each group was further divided into subgroups (n=5): Subgroup A (non-corroded, immersed in distilled water) and Subgroup B (corroded, immersed in 4% acetic acid) [122].
- Testing Protocol:
- Surface hardness was measured using a Vickers diamond indenter.
- Surface roughness was assessed using a non-contact optical profilometer with WSxM analysis software.
- Statistical Analysis: Data normality was verified using Shapiro-Wilk test. Comparisons were made using t-test and one-way ANOVA with significance level set at P ≤ 0.05 [122].
Key Findings: The type of glazing method did not significantly affect zirconia hardness, with no significant differences before and after corrosion for any group (Group 1: p = 0.977, Group 2: p = 0.969, Group 3: p = 0.857). However, surface roughness showed significant increases post-corrosion, with Group 1 (powder/liquid) differing significantly from Group 2 (p = 0.034) and Group 3 (p = 0.038), demonstrating that the powder and liquid method produced the smoothest surface, particularly after corrosion [122].
Mineral Content Analysis After Dental Bleaching
Objective: To evaluate alterations in calcium and phosphorus levels and enamel microstructure following application of three different bleaching protocols [39].
Materials: Ninety extracted human premolars; three bleaching agents: McInnes solution (36% HCl + 30% H₂O₂ + anesthetic ether), modified McInnes solution (35% H₂O₂ + 20% NaOH + diethyl ether), and Pola Office bleach (SDI Ltd., Australia); hydrochloric acid (HCl, Merck, India) in 70% glycerol for microbiopsy; Arsenazo III reagent for calcium quantification; molybdic acid solution for phosphorus quantification; Shimadzu UV-1800 Spectrophotometer [39].
Methodology:
- Sample Preparation: Teeth were randomly divided into three groups (n=30) based on bleaching protocol. Each group was further divided into subgroups for calcium, phosphorus, and SEM analyses [39].
- Enamel Microbiopsy: Samples were collected using enamel microbiopsy technique at five time points: baseline (Day 0), Day 7 and Day 14 before bleaching, and again on Day 7 and Day 14 post-bleaching. The sampling site was demarcated using adhesive tape with a 1.6 mm-diameter perforation [39].
- Etching Protocol: Each biopsy used 5 µl of hydrochloric acid (1.6 M) in 70% glycerol for 20 seconds with gentle agitation. The solution was transferred to Eppendorf tubes with 200 µl ultrapure water [39].
- Spectrophotometric Analysis:
- Calcium quantification used Arsenazo III reagent with absorbance measured at 630 nm.
- Phosphorus quantification used molybdic acid solution with absorbance read at 740 nm.
- Concentrations were expressed in µg/ml [39].
- SEM Analysis: Surface changes were qualitatively observed using scanning electron microscopy [39].
Key Findings: All three bleaching agents resulted in measurable mineral loss, with McInnes solution showing the greatest decrease in calcium concentration at T2 (P = 0.001) and T4 (P = 0.04). SEM revealed pronounced surface alterations in the McInnes group compared to modified McInnes and Pola Office groups. Remineralization was observed after storage in artificial saliva, with partial recovery of mineral levels over 14 days [39].
Bacterial Adhesion Assessment on Modified Zirconia Surfaces
Objective: To evaluate whether polyelectrolyte multilayers added to airborne-particle abraded zirconia can minimize biofilm development [83].
Materials: Zirconia powders with yttria content between 3-5 mol%; polyelectrolyte multilayer components (chitosan and poly(acrylic acid)); profilometer; atomic force microscopy; tensiometry; electrokinetic analyzer; spectrophotometry; SEM [83].
Methodology:
- Sample Preparation: Zirconia powders were isostatically pressed into Ø20-mm disks and sintered at 1450°C for 2 hours (n=8). Untreated specimens were compared with airborne-particle abraded ones [83].
- Coating Application: Specimens with 3 mol% yttria were coated with polyelectrolyte multilayers (n=4) using chitosan and poly(acrylic acid) [83].
- Surface Characterization:
- Roughness measured using profilometry and atomic force microscopy
- Hydrophobicity assessed via tensiometry
- Surface charge determined using electrokinetic analyzer [83].
- Bacterial Adhesion Assessment: The extent of Streptococcus mutans adhesion was determined using spectrophotometry and SEM [83].
- Statistical Analysis: Data were analyzed with single-factor ANOVA and F-test for variance (α=.05) [83].
Key Findings: Airborne-particle abrasion of zirconia increased surface roughness, leading to pronounced adhesion of Streptococcus mutans. However, polyelectrolyte multilayer coatings reduced bacterial adhesion by 50-70%, with the greatest effect observed in poly(acrylic acid)-terminating specimens. The study demonstrated that biocompatible polyelectrolyte multilayer coatings with negatively charged terminating layers significantly reduce bacteria adhesion compared to uncoated specimens [83].
Visualization of Methodologies and Relationships
Surface Modification Validation Workflow
Nanotechnology Enhancement Mechanisms
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Materials for Surface Modification Studies
| Research Material | Primary Function | Application Examples | Key Considerations |
|---|---|---|---|
| Vita AKZENT Glaze Systems (powder/liquid, paste, spray) [122] | Surface sealing and gloss enhancement for ceramic restorations | Comparative studies of glazing methods on zirconia roughness and hardness [122] | Powder/liquid system produces smoothest surfaces post-corrosion [122] |
| Polyelectrolyte Multilayers (chitosan, poly(acrylic acid)) [83] | Bacterial adhesion reduction through surface charge modification | Coating zirconia surfaces to minimize Streptococcus mutans biofilm development [83] | Negatively charged terminating layer most effective (50% reduction) [83] |
| Spectrophotometry Reagents (Arsenazo III, molybdic acid) [39] | Quantification of mineral content (calcium, phosphorus) | Measuring enamel mineral loss after bleaching treatments [39] | Enables tracking of demineralization/remineralization dynamics |
| CAD/CAM Ceramic Blocks (zirconia-reinforced lithium silicate, feldspathic ceramic, lithium disilicate) [76] | Standardized substrates for surface modification studies | Comparative analysis of surface properties across material classes [76] | Different materials show distinct roughness and wettability profiles |
| Nanoparticles (metal oxides, nano-hydroxyapatite, carbon nanotubes) [124] | Multifunctional enhancement of surface properties | Creating antibacterial surfaces, improving mechanical properties, drug delivery [124] | Size, concentration, and distribution critical for performance |
The validation of surface modifications in dental materials requires a multidisciplinary approach that correlates morphological characteristics with chemical composition and biological performance. Traditional methods like glazing continue to offer reliable surface optimization, while emerging biomimetic and nanotechnological strategies provide unprecedented control over material-biological interactions. The experimental data presented in this comparison guide demonstrates that no single modification technique excels across all parameters; rather, material selection must be guided by specific clinical requirements and performance priorities.
Future directions in surface modification research point toward intelligent, multi-functional coatings that dynamically respond to the biological environment. The integration of nanotechnology with biomimetic principles offers particular promise for creating surfaces that not only resist biofilm formation but actively promote tissue integration and regeneration. As validation methodologies continue to advance—particularly in the realm of in situ characterization techniques—researchers will gain increasingly sophisticated tools for designing and optimizing dental material surfaces that predictably enhance clinical outcomes and patient satisfaction.
Conclusion
The comparative analysis of dental biomaterials underscores that surface morphology and chemical composition are inextricably linked to clinical performance, influencing outcomes from osseointegration to biofilm susceptibility. The integration of advanced characterization techniques is paramount for validating new materials and manufacturing methods, such as 3D printing and nanotechnology-enhanced composites. Future directions must focus on developing smart biomaterials with tailored surface properties that actively promote healing and resist infection. Bridging the gap between in vitro characterization and long-term clinical validation remains crucial, requiring collaborative efforts across material science, microbiology, and clinical dentistry to usher in the next era of predictive and personalized dental restorations.
References
- 1. A comprehensive review: surface modification strategies to ... [https://jmsg.springeropen.com/articles/10.1186/s40712-025-00294-9]
- 2. Surface Testing of Dental Biomaterials—Determination of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8196709/]
- 3. The 3D theory of osseointegration: material, topography, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12317956/]
- 4. Comparative Evaluation of Surface Roughness, Wettability ... [https://pubmed.ncbi.nlm.nih.gov/40585711/]
- 5. Comparative evaluation of the basic physical properties of a ... [https://bmcoralhealth.biomedcentral.com/articles/10.1186/s12903-025-07259-6]
- 6. Evaluation of biological functionality of biomaterial surface ... [https://pubmed.ncbi.nlm.nih.gov/41085716/]
- 7. Correlation of three wear testing methods and surface ... [https://www.nature.com/articles/s41598-025-21341-7]
- 8. Surface characterization and bio-functionalization of ... [https://www.sciencedirect.com/science/article/pii/S2949829525000762]
- 9. The Effect of Different Surface Modifications on ... [https://www.sciencedirect.com/science/article/abs/pii/S1532338225000855]
- 10. Structure and composition of early biofilms formed on ... [https://www.nature.com/articles/s41522-024-00624-3]
- 11. Surface Properties and Surface Characterization of Dental ... [https://www.mdpi.com/journal/materials/special_issues/53Z0141RJ8]
- 12. The Application of ImageJ Software for Roughness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12092845/]
- 13. Biocompatible Materials in Dentistry: Revolutionizing Oral ... [https://www.grandlakedental.com/blog/2025/9/8/biocompatible-materials-in-dentistry-revolutionizing-oral-healthcare/]
- 14. Dental Material Market Size, Share | Global Report [2025- ... [https://www.fortunebusinessinsights.com/dental-materials-market-103300]
- 15. Advantages of Advanced Ceramics over Metals and Plastics [https://www.ceramtec-group.com/en/high-tech-ceramics/materials-in-comparison]
- 16. Additive Zirconia in Dentistry: Techniques, Trends, and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12511785/]
- 17. Comparison of Additively Manufactured Polymer-Ceramic ... [https://www.mdpi.com/1996-1944/17/1/240]
- 18. Tribological performance and surface morphological ... [https://www.sciencedirect.com/science/article/abs/pii/S0301679X25001987]
- 19. Quantitatively Predicting Bacterial Adhesion Using Surface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4854535/]
- 20. Interaction between microorganisms and dental material ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10078137/]
- 21. Bacteria Adhesion of Textiles Influenced by Wettability and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7827894/]
- 22. Surface free energy (SFE), surface energy [https://www.kruss-scientific.com/en/know-how/glossary/surface-free-energy]
- 23. Promoting Surface Energy and Osteoblast Viability on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12387143/]
- 24. Analysis of different approaches for evaluation of surface ... [https://www.sciencedirect.com/science/article/abs/pii/S0001868602000040]
- 25. Effectiveness of Surface Treatments on the Bond Strength ... [https://www.mdpi.com/2673-1592/7/3/56]
- 26. Validation of a Novel Technique and Evaluation of the Surface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5409319/]
- 27. The influence of surface treatment on the implant ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3881788/]
- 28. Effect of surface roughness of hydroxyapatite on human ... [https://www.sciencedirect.com/science/article/abs/pii/S0142961200001745]
- 29. Effect of surface energy and roughness on cell adhesion and ... [https://pubs.rsc.org/en/content/articlehtml/2021/ra/d1ra02402g]
- 30. Roughness and dynamics of proliferating cell fronts as a ... [https://www.nature.com/articles/s41598-021-86684-3]
- 31. The relationship of surface roughness and cell response ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3368253/]
- 32. Terrestrial radiotherapy alters microhardness and surface ... [https://www.frontiersin.org/journals/oral-health/articles/10.3389/froh.2025.1658558/full]
- 33. Probing the micro- and nanoscopic properties of dental ... [https://www.sciencedirect.com/science/article/pii/S1742706123004026]
- 34. Selected Spectroscopic Techniques for Surface Analysis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8156406/]
- 35. Comparative Study of Surface Modification Techniques for ... [https://www.mdpi.com/2076-3417/14/23/10904]
- 36. Analysis of Surface Morphology and Elemental Composition ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8375790/]
- 37. An infrared, Raman, and X-ray database of battery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11711556/]
- 38. The Future of FT-IR Imaging [https://www.spectroscopyonline.com/view/the-future-of-ft-ir-imaging]
- 39. Comparative analysis of enamel mineral content and ... [https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2025.1613733/full]
- 40. Spectroscopic (FT-IR and FT-Raman) and quantum ... [https://www.sciencedirect.com/science/article/pii/S2405844025004840]
- 41. Exploring the utility of optical microscopy versus scanning ... [https://www.sciencedirect.com/science/article/abs/pii/S104061822030255X]
- 42. Evaluation by Scanning Electron Microscopy and Energy ... [https://pubmed.ncbi.nlm.nih.gov/39871384]
- 43. Comparative Surface Morphology, Chemical Composition ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7254305/]
- 44. Evaluation of the Validity of Digital Optical Microscopy in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8747457/]
- 45. Surface morphology and chemical composition analysis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11471401/]
- 46. Surface morphology, roughness, and corrosion resistance ... [https://www.sciencedirect.com/science/article/pii/S2238785422016520]
- 47. Scanning electron microscopy [https://congress.digital-dentistry.org/keyword/scanning-electron-microscopy/]
- 48. Application Note: Comparing 3D Optical Microscopy ... [https://www.bruker.com/en/products-and-solutions/test-and-measurement/3d-optical-profilers/resource-library/an-503-comparing-3d-optical-microscopy-techniques-for-metrology-applications.html]
- 49. Performance Analysis of Surface Reconstruction Algorithms in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7915951/]
- 50. Difference between XPS analysis and EDX analysis? [https://rockymountainlabs.com/difference-between-xps-analysis-and-edx-analysis/]
- 51. Energy-dispersive X-ray spectroscopic investigation of a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9985998/]
- 52. Chemical mapping of teeth in 2D and 3D: X-ray ... [https://www.sciencedirect.com/science/article/abs/pii/S1742706120302051]
- 53. X-ray photoelectron spectroscopy as a useful tool to study ... [https://www.sciencedirect.com/science/article/pii/S0039602824000220]
- 54. SEM, EDX, AFM, and XPS analysis of surface ... [https://link.springer.com/article/10.1007/s11082-025-08106-2]
- 55. Assessment of the Chemical Composition in Different ... [https://www.mdpi.com/2079-6412/10/9/882]
- 56. XPS, AES and SEM analysis of recent dental implants [https://www.sciencedirect.com/science/article/abs/pii/S1742706109000506]
- 57. Surface Testing of Dental Biomaterials—Determination of ... [https://www.mdpi.com/1996-1944/14/11/2716]
- 58. Effect of Surface Wettability and Energy on Bacterial ... [https://pubmed.ncbi.nlm.nih.gov/41007143/]
- 59. The Surface Free Energy of Resin-Based Composite in ... [https://www.mdpi.com/2076-3417/13/21/12061]
- 60. A protocol for determining the surface free energy of dental ... [https://www.sciencedirect.com/science/article/abs/pii/S0109564103001027]
- 61. Comparison of contact angle measurements on ... [https://www.biolinscientific.com/blog/comparison-of-contact-angle-measurements-on-superhydrophobic-surfaces?update_2025=1]
- 62. Quantifying the impact of surface roughness on contact ... [https://www.nature.com/articles/s41598-025-01127-7]
- 63. Enamel Surface and Elemental Changes Following In Vitro ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12468495/]
- 64. Effect of surface treatments and bonding type on elemental ... [https://www.nature.com/articles/s41598-024-75709-2]
- 65. A randomised clinical trial comparing a surgical approach ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11839896/]
- 66. Effectiveness of Laser Therapy as an Adjunctive Treatment ... [https://opendentistryjournal.com/VOLUME/19/ELOCATOR/e18742106400224/FULLTEXT/]
- 67. Surface Micromorphology of Experimental Composites ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12027739/]
- 68. Fluoride Casein Phosphopeptide and Tri-Calcium ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12192112/]
- 69. Comparative evaluation of surface roughness and bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11515379/]
- 70. Assessment of biofilm formation on ceramic, metal, and ... [https://www.sciencedirect.com/science/article/abs/pii/S1349007924002433]
- 71. Durable Oral Biofilm Resistance of 3D-Printed Dental Base ... [https://pubmed.ncbi.nlm.nih.gov/33401545/]
- 72. Glass ionomer cement modified by a imidazolium salt [https://pmc.ncbi.nlm.nih.gov/articles/PMC8324747/]
- 73. Frontiers | From biofilm control to biomimetic remineralization... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2025.1663563/full]
- 74. Comprehensive strategies for overcoming dental : Microbial... biofilms [https://pubmed.ncbi.nlm.nih.gov/40349996/]
- 75. Effects of Different Surface Treatments and Accelerated ... [https://www.mdpi.com/2079-4983/16/7/263]
- 76. Evaluation of the Surface Properties of Three CAD/CAM ... [https://www.mdpi.com/2304-6767/13/12/550]
- 77. Assessing the impact of surface treatment, aging, and post- ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12573946/]
- 78. Effect of aging on optical behavior and color of 3D printing ... [https://www.sciencedirect.com/science/article/abs/pii/S0300571225001782]
- 79. Comparison of 3D printed and conventional denture base ... [https://www.nature.com/articles/s41598-025-01685-w]
- 80. High-Cycle Fatigue vs Low-Cycle Fatigue Testing | ITS [https://its-inc.com/news/high-cycle-fatigue-vs-low-cycle-fatigue-testing/]
- 81. Fatigue Resistance of CAD/CAM Dental Materials [https://completesmilesbv.com.au/fatigue-resistance-of-cadcam-dental-materials/]
- 82. Fatigue Testing: Methods, Materials & Applications - Biopdi [https://biopdi.com/fatigue-testing/]
- 83. Adhesion of Streptococcus mutans on highly translucent ... [https://www.sciencedirect.com/science/article/pii/S0022391325000836]
- 84. Comparative study of the fatigue strength of five acrylic ... [https://www.sciencedirect.com/science/article/abs/pii/S1751616110000810]
- 85. Experimental and theoretical study on cyclic fatigue ... [https://www.sciencedirect.com/science/article/abs/pii/S0263224124025181]
- 86. Evaluation of the effects of different polishing systems on ... [https://pubmed.ncbi.nlm.nih.gov/40598133/]
- 87. The current applications of nano and biomaterials in drug of... delivery [https://bmcoralhealth.biomedcentral.com/articles/10.1186/s12903-024-03911-9]
- 88. Antimicrobial Activity Enhancers: Towards Smart Delivery ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8944422/]
- 89. New Direction in Antimicrobial Delivery System [https://www.mdpi.com/1999-4923/17/4/529]
- 90. Fundamental study on the construction of anti -wear drug ... delivery [https://www.jmst.org/EN/Y2025/V222/I0/215]
- 91. Dental implant surface morphology, chemical composition ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9932231/]
- 92. 3-D Surface Morphological Characterization of CAD/CAM ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9268120/]
- 93. Review of Antimicrobial Nanocoatings in Medicine and Dentistry ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10134505/]
- 94. A new evidence-based design-of-experiments approach ... [https://www.nature.com/articles/s41598-024-82496-3]
- 95. Drug delivery systems: An updated review - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3465154/]
- 96. Research Progress on Surface Modification of Titanium ... [https://www.mdpi.com/2079-6412/15/2/229]
- 97. A narrative review of recent developments in osseointegration ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12123356/]
- 98. Zirconia in Dental Implantology: A Review of the Literature ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12109504/]
- 99. Comparison of Hydrophilic Properties of Titanium and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12028884/]
- 100. Zirconia Materials for Dental Implants: A Literature Review [https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2021.687983/full]
- 101. Zirconia vs Titanium Dental Implants: 2025 Complete ... [https://goodtoothdentalcare.com/zirconia-vs-titanium-dental-implants-2025-complete-comparison-guide-for-choosing-your-best-option/]
- 102. Advanced surface modification techniques for titanium ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1549439/full]
- 103. Clinical effectiveness of Zirconia versus titanium dental ... [https://eurjmedres.biomedcentral.com/articles/10.1186/s40001-025-02488-5]
- 104. Controlling Cellular Behavior by Surface Design of ... [https://iv.iiarjournals.org/content/39/3/1786]
- 105. Understanding bacterial biofilms: From definition to ... [https://www.frontiersin.org/journals/cellular-and-infection-microbiology/articles/10.3389/fcimb.2023.1137947/full]
- 106. Classical and Modern Models for Biofilm Studies [https://www.mdpi.com/2079-6382/13/12/1228]
- 107. Surface roughness and biofilm formation on tooth-colored ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12502266/]
- 108. A New Method for the Studying of Bacterial Biofilm Formation [https://pmc.ncbi.nlm.nih.gov/articles/PMC9777215/]
- 109. What We Still Don't Know About Biofilms—Current ... [https://www.mdpi.com/2036-7481/16/2/46]
- 110. Comparison of microbial adhesion and biofilm formation on ... [https://www.sciencedirect.com/science/article/abs/pii/S0889540624003615]
- 111. Biofilm formation and antibiogram profile of bacteria from ... [https://www.nature.com/articles/s41598-024-78283-9]
- 112. Modern Microbiological Methods to Detect Biofilm ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11205407/]
- 113. CNC Or 3D Printing? The Ultimate Guide To Plastic Part ... [https://www.lsrpf.com/en/blog/cnc-or-3d-printing-the-ultimate-guide-to-plastic-part-manufacturing-in-2025]
- 114. Three-Dimensional Printing and CAD/CAM Milling in ... [https://pubmed.ncbi.nlm.nih.gov/40725530/]
- 115. A systematic review and network meta-analysis [https://pubmed.ncbi.nlm.nih.gov/39779439/]
- 116. Milling has superior mechanical properties to other ... [https://www.sciencedirect.com/science/article/pii/S0109564124003737]
- 117. Wear of interim resin materials manufactured by 3D ... [https://pubmed.ncbi.nlm.nih.gov/41146159/]
- 118. Milling Versus Printing: The Effect of Fabrication Technique ... [https://www.mdpi.com/2673-1592/7/5/107]
- 119. Influence of 3D printing parameters on surface roughness ... [https://www.sciencedirect.com/science/article/abs/pii/S0022391325006468]
- 120. Machine learning based approach for surface roughness ... [https://www.nature.com/articles/s41598-025-17487-z]
- 121. Resin materials for 3D-printing and milling of indirect ... [https://www.sciencedirect.com/science/article/pii/S0109564125008061]
- 122. Effect of different glazing methods on the surface ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12490138/]
- 123. Nature-inspired surface modification strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11925587/]
- 124. Integration of Nanotechnology and Nanomaterials in ... [https://scifiniti.com/3105-0387/2/2025.0019]